TABLE 3.4.I.1 CEAP Classification of Chronic Venous Disease | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||
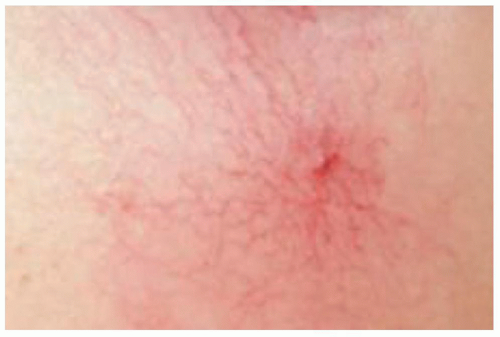
Sinus or simple (linear): A red, linear telangiectasia that occurs on the face, especially the nose, or legs. In addition, a blue linear anastomosing telangiectasia may be found often on the legs.
Arborizing: A treelike appearance of capillary vessels in inflamed condition.
Spider or star: A red, superficial telangiectasia arising from a central filling vessel of arteriolar origin. It
is a focal network of dilated capillaries seen chiefly in pregnancy and hepatic cirrhosis. These are characteristically 0.1 to 1.0 mm in diameter and red to cyanotic in color.
Punctiform (papular): Characterized by small circumscribed, superficial elevation of the skin and are the results of dilated vessels. These are generally less than 2 mm in diameter and frequently present in patients with collagen vascular disease.
Poikiloderma
Solar elastosis
Rosacea
Telangiectasia macularis eruptiva perstans
Discoid lupus erythematosus, systemic lupus erythematosus
Dermatomyositis
Systemic sclerosis/CREST syndrome
Pigmented purpuric eruption
Acroangiodermatitis
Erythema ab igne
Palmar erythema
Radiation dermatitis
Cutaneous collagenous vasculopathy
Generalized essential telangiectasia
TABLE 3.4.I.2 Causes of Spider Veins | ||||
|---|---|---|---|---|
|
with progressive reflux causing secondary changes in the vein wall. Alternatively, or concurrently, the valves may become incompetent secondary to structural abnormalities and focal dilation in vein wall segments near the valve junctions, and the reflux ensues as a secondary event. Acquired telangiectasias are a consequence of the release or activation of vasoactive substances, such as hormones and other chemicals. Insights from histological evaluations have shown dilation of the valvular annulus, bulging valve leaflets, commissural dilation, leaflet stretching, shortening, tearing and perforation, and, ultimately, complete destruction of the valve, whereas the venous wall presents varied areas of either thickening or deterioration.13 Key ultrastructural findings include the infiltration of leukocytes and monocytes, suggesting activation of inflammatory cascades.14,15 The increase in venous pressure causes structural and functional changes in the vein wall that leads to further venous dilation and increases in vein wall tension that trigger the expression/activity of matrix metalloproteinases (MMPs). Engagement of this biochemical cascade induces degradation of the extracellular matrix proteins, thus affecting the structural integrity of the vein wall and engaging inflammatory pathways to scavenge the cellular debris. Recent evidence also suggests an effect of MMPs on the endothelium and smooth muscle components of the vein wall and thereby causing changes in the venous constriction/relaxation properties.16 Endothelial cell injury also triggers further leukocyte infiltration, activation, and inflammation, which leads to additional vein wall damage and release of several growth factors such as vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and platelet-derived growth factor (PDGF). Thus, in the underlying general pathogenesis, vein wall dilation and valve dysfunction, via MMP activation and superimposed inflammation and fibrosis, leads to the appearance of telangiectasias.17
telangiectasias is not curative and that further development of telangiectatic areas is likely.18,19,20
TABLE 3.4.I.3 Sclerosing Agents for Telangiectasia | ||||
|---|---|---|---|---|
|
Inspection and palpation of veins, marking of the treatment area, photography
Skin preparation, cleansing with rubbing alcohol
Syringe preparation: 2.5-mL syringe, 30-gauge needle
Localization, puncture, and injection of telangiectasias
Use magnifying goggles, transillumination, or other visualization devices when needed. Multiple injections of low volumes are advocated and avoidance of blanching areas bigger than 2.5 × 2.5 cm from a single injection point.
After the procedure, injection sites are cleansed and cotton balls are applied, adhesive tape, compression stockings, and/or bandages if necessary. Some recommend wearing compression stockings for up to 3 days post procedure to improve outcomes.
The patient is advised to rest for 5 minutes. Application of nonsteroidal anti-inflammatory cream (ibuprofen ointment or equivalent) can be offered to limit bruising. Patients are advised to avoid hot tubs, hot showers, and hot wax depilation after sclerotherapy treatments because these are known to be responsible for vasodilatation, edema, swelling, and heaviness. Walking is encouraged but not engagement in heavy exercise.
Follow-up evaluation is required several days or weeks after the procedure to assess its outcome, to recognize potential side effects, and to carry out additional injections if necessary (Figure 3.4.i.2).
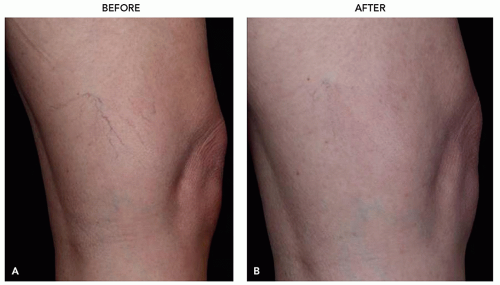
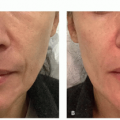
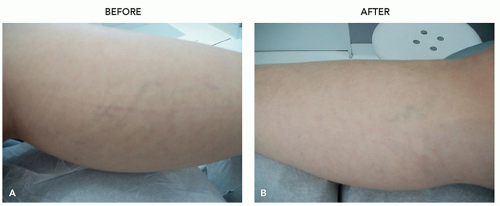 FIGURE 3.4.I.2 A 30-year old woman before (A) and 3 weeks after (B) 1 treatment with 0.5% polidocanol. |
either STS 0.25% to 0.50% or polidocanol 0.75% with a follow-up of 2 to 9 years showed an improvement of 50% to 100%.27 In another study, Weiss et al compared the use of 0.5% polidocanol with 11.7% hypertonic saline (HS) for the treatment of leg telangiectasias. All patients completed 4 visits at 0, 1, 4, and 12 weeks and reported significantly greater pain during treatment with HS than with polidocanol. There were no significant differences in physician-assessed improvement of telangiectasias, but 2 subjects developed ulcerations with HS. Thus, both agents provided effective treatment, but HS caused more pain during injections and resulted in 2 episodes of tissue necrosis.28 In a clinical study by Rabe et al, 160 patients with telangiectasias were treated with 0.5% polidocanol, 1% STS, or placebo. Polidocanol demonstrated a statistically significant superiority versus placebo, and significantly more patients were satisfied with polidocanol at 12 or 26 weeks compared with STS and placebo.29
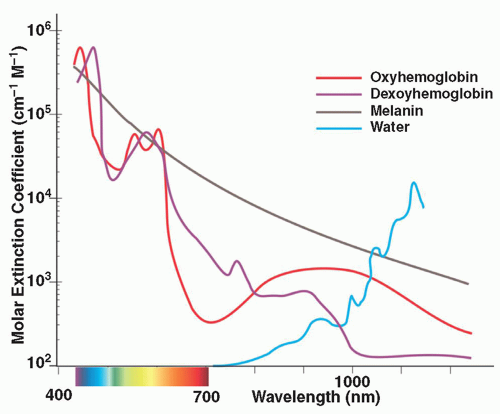
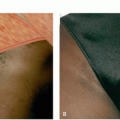
wavelength penetrates to deeper levels and, with long pulse durations, can be used to treat veins from 0.3 to 3 mm in diameter (Figure 3.4.i.5). Because of lower absorption in pigment, Nd:YAG lasers are less likely to cause pigmentary changes and are safer to use in darker skin types.
TABLE 3.4.I.4 Lasers Used to Treat Spider Veins | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||
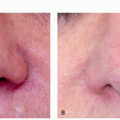
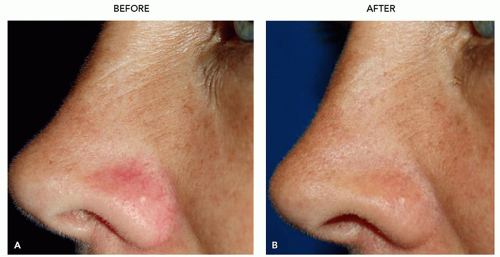 FIGURE 3.4.I.4 A 36-year old woman before (A) and 4 weeks after (B) treatment with 532-nm KTP, 4 mm, 15 milliseconds, 10 J/cm2. |
or VI, and usually develop within 3 weeks after treatment. Hyperpigmentation, which is due to hemosiderin deposition, is usually temporary but may last up to a year or can be permanent as can be hypopigmentation. Thrombosis has been reported in the superficial veins following laser therapy and may take up to 6 weeks to resolve, but it rarely has sequelae.
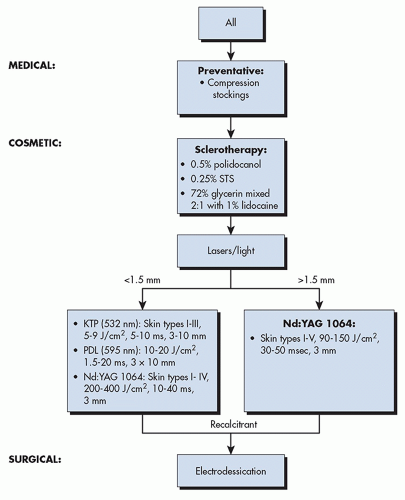
for cosmetic reasons, both sclerotherapy, which is the gold standard, and lasers, which are an appropriate alternative, are excellent options that are effective in a few treatment sessions and can provide long-term results.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree