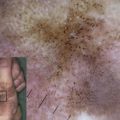
Kindler syndrome is caused by genetic defects in the focal contact–associated protein, fermitin family homologue 1 (FFH1), encoded by the gene FERMT1 (known as KIND1). Defects in FFH1 lead to abnormal integrin activation and loss of keratinocyte epidermal adhesion to the underlying basal lamina, disruption in normal cell cytoskeleton within keratinocytes, and altered signaling pathways, leading to increased extracellular matrix production. Null mutations in FERMT1 result in skin blistering from birth and early childhood progressive poikiloderma, mucosal fragility, and increased risk of cancer. The complete range of FFH1 functions in skin and other epithelia has yet to be determined.
KIND1 gene product biology, structure, and function
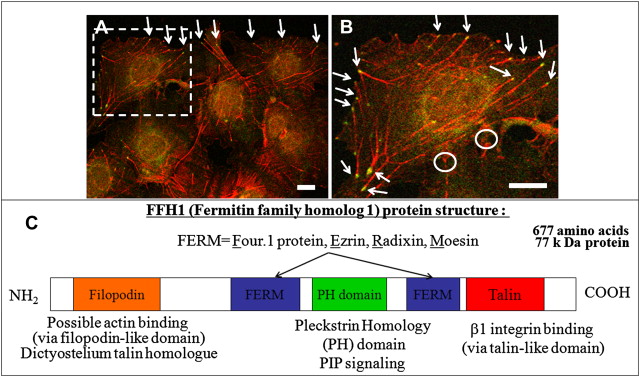
Kindler syndrome (KS) (OMIM173650) is a rare autosomal recessive skin disorder, first described in 1954. The precise mechanism by which FERMT1 gene mutations (encoding fermitin family homolog 1 protein [FFH1]) result in KS symptoms, including blistering, epidermal atrophy, increased risk of cancer, and poor wound healing, remains unclear. Knowledge of some of the more basic FFH1 functions in skin and, in particular, keratinocytes, however, is steadily growing. Cell-matrix interactions are mediated by integrins, heterodimeric transmembrane proteins comprising α and β subunits, and are controlled through integrin activation and cytoskeletal organization. Overall, studies suggest a critical role of FFH1 in cell-matrix interactions via integrin adhesion receptors. An important step in integrin activation is the binding of FERM ( f our point one protein, e zrin, r adixin, and m oesin) domain proteins, such as talin, to the cytoplasmic tail of β integrin subunits. FFH1 (also a FERM or fermitin family homologue protein) can bind to several cytoplasmic motifs within the tail domains of β1 and β3 integrins, colocalizing with vinculin at focal adhesions in cultured keratinocytes. In addition, FFH1 associates with FFH2 (a kindlin-1 homologue, formerly known as kindlin-2) and filamin-binding LIM protein together with other focal adhesions components at sites of cell-matrix contact. Recent cell biologic mutational analysis demonstrated that the second conserved NXXY motif (Tyr795) and the threonine-containing region (Thr788 and Thr789) of the integrin β1A tail, together with the conserved tryptophan in the F3 subdomain of the FFH FERM domain (kindlin-1 Trp612 and kindlin-2 Trp615), are required for direct FFH-integrin interactions. Tissue-specific expression of FFH protein regulates integrin activation, including FFH1 (in epithelia), FFH2 (expressed predominantly in the heart and other tissues), and FFH3 (where expression is restricted to hematopoietic lineages). FFH2/3 proteins are essential regulators of integrin activation, and deficiencies in these focal adhesions proteins leads to cardiac malformation and platelet dysfunction, respectively. These defects make knockout mouse work difficult or prohibitive due to poor survival. From patients with KS disease, it is known that long-term FFH1 deficiency leads to an increased cancer risk; however, the precise mechanisms causing these effects are unknown. Focal adhesions are thought to have important roles in preventing disease by controlling cell survival, growth, signaling, and cell invasion, making them central to the study and treatment of cancer. The role of FFH proteins is thought to be related to stem cell maintenance via its control of important cell cycle processes and also via the regulation of multiple growth factors that are associated with cancer progression and tumor cell survival and, critically, are involved in wounding. FFH1, -2, and -3 form part of a protein family that share a high degree of sequence homology in addition to a bipartite FERM domain interrupted by a pleckstrin homology domain that is important in cell signaling ( Fig. 1 A). Nevertheless, the precise downstream roles of FFH proteins after integrin activation and their involvement in precise signaling pathways controlling specific cell functions remain to be determined.