Patterned hair loss in men and women, although medically benign, is a common, albeit unwelcome, event that may cause considerable anxiety and concern. Patterned hair loss is progressive and when untreated leads to baldness. The prevalence and severity of this physiologic process both increase with advancing age. Although androgens play a key role in the pathogenesis of male pattern hair loss (MPHL), the role of androgens in female pattern hair loss (FPHL) is less well established. Satisfactory treatment response to antiandrogen therapy supports the involvement of androgens in the pathogenesis of FPHL. Spironolactone has been used for 30 years as a potassium-sparing diuretic. Spironolactone is a synthetic steroid structurally related to aldosterone. Since the serendipitous discovery 20 years ago that spironolactone given to a woman for polycystic ovary syndrome (PCOS) and associated hypertension also improved hirsutism, it has been used as a primary medical treatment for hirsutism. Spironolactone both reduces adrenal androgen production and exerts competitive blockade on androgen receptors in target tissues. Spironolactone has been used off-label in FPHL for over 20 years. It has been shown to arrest hair loss progression with a long-term safety profile. A significant percentage of women also achieve partial hair regrowth. Spironolactone is not used in male androgenetic alopecia because of the risk of feminization.
Female pattern hair loss (FPHL) is the most common cause of diffuse hair loss in women. Ludwig described the hair loss pattern in 1977 and stated it to be the female equivalent of male baldness. It also is referred to as female androgenetic alopecia. Although considerable histologic similarities exist between male pattern hair loss (MPHL) and FPHL, the age of onset, rate of progression, and response to therapy in females differ from that of males.
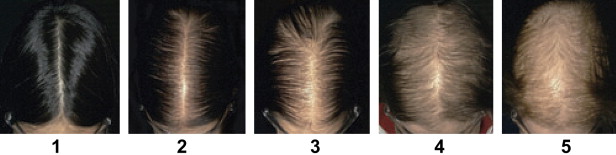
A population survey performed in 2001 indicated that the age-adjusted prevalence of FPHL among Australian women of European descent aged 20 and over is 32.2% (95% confidence interval [CI], 28.8% to 35.6%). Among these women, 10.5% (95% CI, 8.2% to 12.7%) or approximately 800,000 Australian women were assessed as having moderate-to-severe FPHL as defined by stage 3, 4, or 5 on the clinical grading scale shown in Fig. 1 . The prevalence of FPHL increases with advancing age, from approximately 12% among women aged between 20 and 29 years to over 50% of women over the age of 80.
The impact of FPHL is predominately psychological. When compared with control women, FPHL has a significant detrimental effect on self-esteem, psychological well-being and body image that is consistent across different cultures. Affected women report a poorer health-related quality of life (QOL) than nonaffected women.
Topical minoxidil is the only US Food and Drug Administration (FDA)-approved treatment for FPHL. Globally, the antiandrogens cyproterone acetate, spironolactone, and flutamide are widely used for treating FPHL. Prolonged continuous use is required for sustained effect. Poor donor hair density over the occipital scalp precludes many women from hair transplantation. Nonpharmacologic methods such as hair coloring, permanent waves, careful hair styling, and camouflage powders and creams also can provide temporary benefit.
Etiology of FPHL
The hair loss progresses in a highly reproducible pattern. Exposure of genetically susceptible hair follicles to androgen leads to several changes including hair follicle miniaturization and changes in hair cycle dynamics with shortening of anagen duration and prolongation of telogen duration.
Genetics and Androgenetic Alopecia
Twin studies have confirmed the strong genetic predisposition to male pattern baldness. Androgenetic alopecia (AGA) is complex polygenic condition. The first gene associated with AGA was the androgen receptor gene on the X chromosome ; however, this does not explain the high frequency of balding fathers among affected men. Balding risk increases with the number of affected family members. There is racial variation in the age-related prevalence of balding, with Asian men having a lower prevalence than European men.
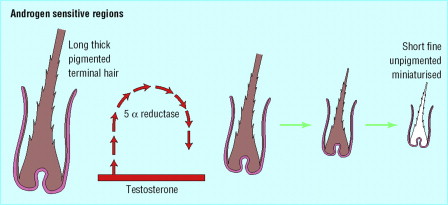
The cytochrome p 450 alpha aromatase enzyme also has been found to contribute to AGA in females. Aromatase is a key enzyme in estrogen biosynthesis. Aromatase catalyzes the conversion of testosterone to estradiol, androstenedione to estrone, and 16- hydroxylated dehydroepiandrosterone to estriol. and thereby diminishes the amount of intrafollicular testosterone available for conversion to dihydrotestosterone (DHT). Young women have higher levels of aromatase compared with the male scalp and much higher levels in the frontal hair line. This may explain the less severe baldness and relative sparing of the frontal hair line in FPHL.
Androgens and AGA
The role of androgen in MPHL was established by the anatomist James Hamilton, who observed that castrated males did not develop AGA unless treated with testosterone. Testosterone is the main circulating androgen, and the tissue effects of androgens are mediated by biding to the intracellular androgen receptor. In androgen insensitivity syndrome, caused by a mutation in the androgen receptor gene, there is neither baldness nor development of the beard, or axillary and pubic hair, despite the presence of normal circulating androgens.
In most body sites, secondary sexual hair growth is mediated by DHT. The androgen receptor has a several fold higher sensitivity to DHT than its precursor testosterone. Conversion of testosterone to DHT is catalyzed by the enzyme 5 alpha reductase. There are two isoforms of the enzyme (types 1 and 2). Although both are capable of producing DHT from testosterone, they have distinct tissue-specific expression patterns. High concentrations of 5 alpha reductase type 2 are observed in the dermal papillae and androgen dependant sites. Low levels of 5 alpha reductase are seen in the occipital hair, which shows little or no response to androgen. This may explain in part the site-specific behavior within the hair follicle in different body sites.
Itami proposed that the second messenger system determines whether androgen-sensitive follicles will respond to androgens by miniaturization rather than enhancement. Androgen stimulation of cultured beard dermal papilla cells (DPC) lead to increased transcription of insulin-like growth factor-1 and enhanced growth of cocultured keratinocytes. Androgen stimulation of DPC derived from a balding scalp leads to suppression of growth of cocultured keratinocytes. This growth suppression of keratinocytes was mediated by transforming growth factor-beta1 (TGF-beta1) derived from DPC of AGA, suggesting that TGF-beta1 is a paracrine mediator for AGA expressed.
The important role of 5 alpha reductase activity in AGA is supported by absent temporal regression and baldness in men with 5 alpha reductase deficiency caused by mutation in 5 alpha reductase gene, and the proven role of the 5 alpha reductase inhibitors finasteride and dutasteride in treating men with AGA.
Although patterned hair loss and hirsutism are seen in women with hyperandrogenism, most women with FPHL have androgen levels within the normal range. Sawaya and Price reported that the concentration of androgen receptors in women is 40% less compared with men, and also women have low concentrations of 5 alpha reductase levels. A single case report of clinically and histologically proven FPHL in a woman with hypopituitarism and undetectable androgens raises the possibility that this pattern of hair loss also can be induced by events independent of androgens.
Pathophysiology of FPHL
In a normal individual, the hair follicle undergoes a repetitive sequence of growth and rest known as the hair cycle (shown in Fig. 2 ). The period of active hair growth (anagen) determines the final length of hair. At the end of anagen, the hair follicle involutes during the course of a 2-week phase known as catagen. Catagen is followed by telogen, during which time the proximal part of the hair shaft is keratinized to form a club-shaped structure. Telogen lasts approximately 3 months, during which time the club hair lies just below the insertion of arrector pili muscle. Club hair is shed actively in the exogen phase, which usually occurs before the commencement of the next hair cycle with the onset of anagen phase growth.
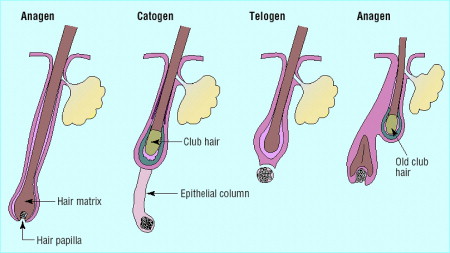
In FPHL, the duration of anagen decreases with each successive cycle, while the duration of telogen remains constant. This leads to progressive shortening of the terminal hairs. Concurrently, the entire size of the hair follicles decreases, leading to a progressive reduction in fiber diameter. Both processes combine to replace long terminal hairs with short fine vellus hairs ( Fig. 3 ). The histologic hallmark of the AGA in both men and women is an increase in the ratio of miniaturized hair follicles; this is appreciated best on horizontally sectioned scalp biopsy.
The histologic decline in mean total follicle count, the reduction in terminal follicle counts, the increase in absolute number of vellus follicles, and terminal/vellus ratio all correlate with clinical severity of FPHL as graded using a five-point scale.
Scalp follicles exist as compound follicles, with a single arrector pili muscle with two or three terminal hairs exiting from a single pore. Histologically this correlates with follicular units (FUs). In FPHL, mean numbers of FUs remain constant, but the mean number of terminal hairs per FU decreases while there is a corresponding increase in the mean number of vellus hairs per FU. This suggests a hierarchy of susceptibility within FUs to AGA. Complete miniaturization of all hairs within the FU is uncommon in women explaining why, unlike men, women rarely progress to complete baldness.
Clinical Features and Diagnosis
The main clinical feature in both men and women is the patterned hair loss over the crown. Women can present early with recurring episodes of increased hair shedding and reduction in the volume of the pony tail, or later with thinning of hair over the crown. Widening of the central part is used to assess the severity of the hair loss. Ludwig first introduced clinical grading of FPHL. He described three stages of severity that range from rarefaction of the hair on the crown (stage 1) to near total baldness (stage 3). One of the authors (RDS) later described a scale with five stages, which the authors have found to be more helpful in assessing the progression of the disease and the treatment response (see Fig. 1 ). Although most women maintain their frontal hair line, about 13% of premenopausal women and 37% of postmenopausal women show bitemporal recession.
The diagnosis of FPHL is largely clinical. Reduction in hair density over the crown and widening of the central part enable a confidant clinical diagnosis. Scalp biopsy is helpful for women who present early with increased hair shedding, reduction in the ponytail volume but no obvious widening of the central part line. Horizontal sectioning is more informative and allows calculation of the vellus hairs to terminal hair ratio. Women with associated hirsutism, acne, menstrual irregularities, or other evidence of virilization should be evaluated for polycystic ovary syndrome or other causes of hyperandrogenism.
Chronic telogen effluvium is an important differential diagnosis for women who present with increased hair shedding, reduction in the ponytail volume but no obvious widening of the central part-line. Miniaturization is not seen on scalp biopsy. This condition tends not to progress to baldness nor respond to oral antiandrogens.
Treatment
Without treatment, FPHL is progressive. The rate of progression has been estimated to be around 10% per year. There are several treatments that are effective in arresting the progression of the hair loss and, in some cases, stimulating partial regrowth of hair. Complete regrowth of hair does not occur. All treatments need to be maintained indefinitely for sustained effect. Medical management for FPHL consists of topical minoxidil, oral antiandrogens, or a combination of these modalities.
Topical minoxidil is effective in arresting hair loss in about 60% of cases. Minoxidil is available in a 2% solution and a 5% solution. The 5% solution has shown greater benefit compared with the 2% solution. A 10% solution is available in India. Minoxidil is a piperidinopyrimidine derivative and a vasodilator and has been used as an oral antihypertensive agent. Minoxidil is thought to act by initiating telogen-to-anagen transition and prolonging anagen duration. Minoxidil is thought to act directly on potassium channel K (ATP) in the follicular dermal papilla.
Oral antiandrogens such as cyproterone acetate (CPA), flutamide, and spironolactone have been available for many years. All are FDA category D drugs, and thus contraindicated in pregnancy. CPA is used in Europe, South America, Asia, Australia, and Canada but is not FDA-approved for use in the United States. Use of Flutamide is limited by its potential severe liver toxicity. The 5 alpha reductase inhibitor finasteride is contraindicated in premenopausal women because of the risk of teratogenesis. A 12-month, randomized, double-blinded placebo-controlled study of postmenopausal women with FPHL failed to demonstrate superiority of finasteride over placebo. Dutasteride, a combined 5 alpha reductase types 1 and 2 inhibitor, has not been systematically tested in women with FPHL, even though beneficial effects have been reported in isolated case studies.
Spironolactone
Spironolactone is an aldosterone antagonist and has been used as a potassium-sparing diuretic for over 50 years. It is structurally a steroid, with basic steroid nuclei with four rings. Its primary metabolite, canrenone, is the active antagonist of aldosterone and contributes to the diuretic action. The ingested drug is absorbed rapidly and metabolized by the liver to canrenone and potassium canrenoate. The drug is available in 25 mg and 100 mg tablets. No dermatologic indications for spironolactone have been approved by the FDA.
Mechanism of Action of Spironolactone in Treatment of FPHL
Spironolactone is an antiandrogen. It acts by decreasing the production and blocking the effect of androgens in the target tissue. Spironolactone decreases testosterone production in the adrenal gland by depleting microsomal cytochrome p450 and by affecting the cytochrome p450-dependant enzyme 17a-hydroxylase and desmolase. The action of spironolactone is limited to tissues with a high microsomal 17a-hydroxylase activity and thereby decreasing steroid 17–hydroxylation. Spironolactone also is a competitive inhibitor of the androgen receptor and thereby blocks the androgen action on the target tissues.
Uses of Spironolactone in Dermatology
Antiandrogenic properties of spironolactone have been exploited for over 30 years in treating hirsutism, acne, and seborrhea. The benefit of spironolactone in hirsutism was discovered when a female with polycystic ovary syndrome was being treated for concurrent hypertension. The patient had noticed improvement in her hirsutism after 3 months of spironolactone therapy. Several studies have shown the clinical efficacy of spironolactone in hirsutism associated with polycystic ovary syndrome (PCOS). When compared, the efficacy of CPA 50 mg/d and ethynylestradiol 35 μg/d with spironolactone 200 mg for idiopathic hirsutism and PCOS, idiopathic hirsutism responded equally while the latter showed better results with CPA-ethynylestradiol combination. Spironolactone has shown superior efficacy when compared with finasteride in treating hirsutism.
Spironolactone also has shown efficacy and long-term safety in treating acne, and it is effective in treating seborrhea. Conflicting results were recorded after using topical spironolactone for seborrhea. Topical 5% spironolactone solution is reported to inhibit dihydrotestosterone receptors in human sebaceous glands. On the other hand, small case studies have failed to demonstrate the effect of topical spironolactone in reducing sebum excretion. Further studies are necessary to determine the antiandrogenic properties of topical spironolactone and the possibility of using it for FPHL.
Spironolactone is the most widely used, albeit off-label, antiandrogen for FPHL in the United States. In Australia, spironolactone has been used for FPHL for more than 20 years, especially in premenopausal women. The standard dose used is 100 to 200 mg daily. Lower doses of 50 to 75 mg also have been shown to stabilize hair loss in some women; however, daily doses in excess of 150 mg have been shown to work better.
Sinclair and colleagues investigated 80 women with biopsy-proven FPHL. Forty women received spironolactone 200 mg daily, and 40 received cyproterone acetate. Premenopausal women received cyproterone acetate 100 mg daily for the first 10 days of each menstrual cycle for 12 months in combination with an oral contraceptive pill. Postmenopausal women received 50 mg daily of cyproterone acetate continuously. Women were followed with serial scalp global photography under standardized conditions. At the end of 12 months, thirty-five (44%) women had clearly visible hair regrowth; 35 (44%) had no change in hair density, and 10 (12%) had reduced hair density at the end of the study, indicating continuing hair loss during the treatment period. There was no significant difference between the two drugs. Most of the women in this study did not have clinical or biochemical evidence of hyperandrogenism. Several other small case studies have shown the beneficial effect of spironolactone in AGA. Additive effects of combination of spironolactone and 5% minoxidil have been noted.
Adverse effects of spironolactone are dose-dependant. Hyperkalemia, although rare in the presence of normal renal function, is a potential adverse effect. Regular monitoring of serum potassium and blood pressure is recommended. Concurrent use of drugs that increase serum potassium should be avoided. Menstrual irregularity, tiredness, and breast tenderness are common, albeit usually mild and rarely necessitating cessation of the medication. As it can feminize the male fetus, spironolactone should be avoided in pregnancy. Chemical hepatitis has been reported, as has chloasma.
Monitoring of Treatment Response
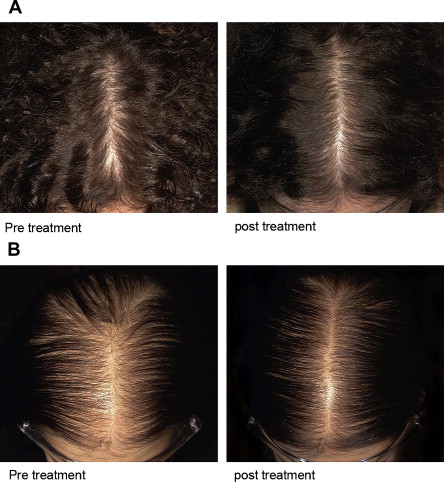
Monitoring clinical response is important for the physician and patient. The response to treatment is slow, and accurate monitoring is facilitated by good-quality photographs ( Fig. 4 ). In the absence of photographs, periodic midscalp clinical grading is the most reliable measure to assess the treatment response. The midscalp clinical grading scale was developed from midscalp photographs and used to assess the severity of hair loss among women undergoing treatment for hair loss. The five-point visual analog grading scale has good interobserver reproducibility and can be used for patient self-assessment. Seasonal fluctuations in hair shedding make counting the number of falling hairs an unreliable method of evaluation.