Keywords
Infliximab, TNF-α, Psoriasis, SPIRIT, EXPRESS, EXPRESS II, IMPACT
Key points
- •
Infliximab is a safe and effective therapy for the treatment of moderate-to-severe psoriasis.
- •
The mechanism of action of infliximab is inhibition of tumor necrosis factor-α, thereby inhibiting the inflammatory cascade leading to psoriatic skin lesions.
- •
Pivotal trials on infliximab therapy for the treatment of psoriasis and psoriatic arthritis include SPIRIT, EXPRESS, EXPRESS II, and IMPACT.
- •
Infliximab is generally safe and well-tolerated. Annual tuberculosis screening is recommended with infliximab use.
Introduction
Standard therapies for the treatment of severe psoriasis have traditionally been limited to methotrexate (MTX), cyclosporine, acitretin, psoralen and UV-A radiation (PUVA), and UV-B phototherapy. Tumor necrosis factor (TNF) inhibitors have been approved for use in severe psoriasis and represent a convenient, targeted systemic agent that can achieve rapid clinical improvement with less potential for drug-related toxicities in both the short and the long term. Infliximab is a chimeric immunoglobulin 1 (IgG1) monoclonal antibody that targets TNF-α. Multiple randomized, controlled trials have evaluated the efficacy and safety of infliximab in psoriasis and psoriatic arthritis (PsA). In 2006, the US Food and Drug Administration approved infliximab for the treatment of chronic, severe plaque psoriasis and PsA. Infliximab is also approved for use in rheumatoid arthritis, Crohn disease, ulcerative colitis, and ankylosing spondylitis and is used off-label in the treatment of neutrophilic and bullous dermatoses, connective tissue diseases such as dermatomyositis and scleroderma, and hidradenitis suppurativa.
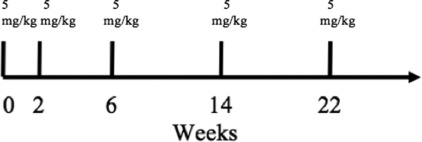
The current recommended dosing of infliximab for chronic, severe psoriasis and PsA is 5 mg/kg given as an intravenous (IV) induction regimen at weeks 0, 2, 6, followed by maintenance dosing of 5 mg/kg at 8-week intervals afterward ( Fig. 9.1 ). Patients who develop neutralizing antibodies to infliximab are less likely to maintain a response than those who are antibody negative, but the formation of antibodies does not necessarily determine clinical responsiveness. When the clinical response to infliximab is waning, the frequency of infusions may need to be increased or low-dose MTX may be prescribed concurrently to decrease the formation of antibodies against infliximab and therefore maintain clinical efficacy ( Table 9.1 ).
| Study | Study Population | Dose and Timing | Results |
|---|---|---|---|
| SPIRIT (phase 2, Gottlieb 2004) |
|
| PASI-75 week 10: 3 mg/kg 72%, 5 mg/kg 88%, placebo 6% ( P <.001) DLQI % improvement week 10: 3 mg/kg 84%, 5 mg/kg 91%, placebo 0% ( P <.001) |
| EXPRESS (phase 3, Reich 2005) |
|
| PASI-75 week 10: infliximab 80%, placebo 3% ( P <.0001) PASI-75 week 24: infliximab 82%, placebo 4% ( P <.0001) PASI-75 week 50: infliximab 61% |
| EXPRESS II (phase 3, Menter 2007) |
|
|
|
| IMPACT (Antoni 2008) |
|
|
|
Mechanism of action
Infliximab is a chimeric monoclonal antibody composed of a constant human IgG1 region and a variable murine region. It has high binding affinity and specificity for TNF-α, forming complexes with both soluble and transmembrane forms of TNF-α, thus preventing its downstream effects. The binding to transmembrane TNF-α mediates complement and antibody-dependent cytotoxicity of cell lines that express TNF-α, namely macrophages and monocytes.
TNF-α is found in high levels in the skin lesions and plasma of psoriasis patients and plays a key role in pathogenesis of psoriasis. TNF-α upregulates vascular endothelial growth factor, induces keratinocyte proliferation, stimulates lymphocyte migration, and increases the expression of proinflammatory cytokines. By inhibiting TNF-α, infliximab inhibits key steps in the pathogenesis of psoriasis and normalizes keratinocyte differentiation.
Efficacy
The efficacy of infliximab in the treatment of psoriasis has been evaluated in multiple randomized, controlled trials. The Study of Psoriasis with Infliximab Induction Therapy (SPIRIT), European Infliximab for Psoriasis Efficacy and Safety Study (EXPRESS), and European Infliximab for Psoriasis Efficacy and Safety Study II (EXPRESS II) trials found that infliximab induces rapid, efficacious, and sustainable clinical response for the treatment of moderate-to-severe plaque psoriasis.
Study of Psoriasis with Infliximab Induction Therapy
The SPIRIT trial was a multicenter double-blinded, placebo-controlled phase 2 trial that randomized 249 patients to receive IV infusions of infliximab or placebo. Selected patients had plaque psoriasis for at least 6 months, Psoriasis Area Severity Index (PASI) ≥12 or more, and 10% or greater body surface area (BSA) affected. The primary endpoint was the proportion of patients who achieved PASI-75 at week 10. Patients were randomly assigned in a 1:2:2 ratio to infusions of infliximab 3 mg/kg, 5 mg/kg, or placebo and were treated at weeks 0, 2, and 6, and followed to week 30.
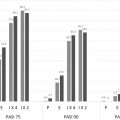
Responses were seen after the first infusion of infliximab. By week 2, 34% and 40% of patients in the infliximab 3 and 5 mg/kg groups had achieved PASI-50, compared with 4% in the placebo group. At week 10, 72% of the 3-mg/kg group and 88% of the 5-mg/kg group achieved PASI-75 compared with 6% of placebo. These results correlated with Physician’s Global Assessment (PGA) findings, with 72% and 90% of the infliximab 3- and 5-mg/kg groups, respectively, achieving PGAs of minimal (PGA = 1) or cleared (PGA = 0) psoriasis by week 10. Improvements in quality of life were also noted–the median change in Dermatology Life Quality Index (DLQI) from baseline to week 10 was −8 and −10 for the 3- and 5-mg/kg groups, respectively, compared with 10 in the placebo group.
To assess the safety of re-treatment, patients with PGA ≥ 3 were eligible for a single IV infusion of their assigned treatment at week 26. A month after re-treatment, 38%, 64%, and 18% of patients in the 3-mg/kg, 5-mg/kg, and placebo group had a PsA less than 3 (considered mild, minimal, or clear psoriasis).
Patients were followed up to 20 weeks to assess the duration of response. The maximum response was achieved at week 10 for both the 3- and the 5-mg/kg groups. The response began to decline at week 10 in the 3-mg/kg group and week 14 in the 5-mg/kg group.
In summary, SPIRIT demonstrated that infliximab induction therapy resulted in significant improvement in psoriasis for most patients in the study. After 3 doses of infliximab, the response rates in this study were similar to those seen with cyclosporine, and greater than those achieved by other biologics. The response seen at week 4, with about one-third and one-half of patients in the 3- and 5-mg/kg infliximab groups, was only seen after 12 or more weeks of therapy with other biologics. The improvement in quality of life, as measured by DLQI, was also greater than other biologics or hospital-based treatment ( Fig. 9.2 ).
European Infliximab for Psoriasis Efficacy and Safety Study I
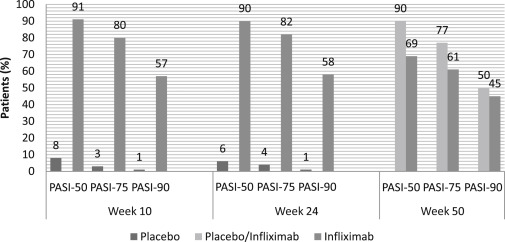
The EXPRESS I trial was a multicenter, double-blinded, phase 3 trial that evaluated the efficacy of infliximab induction and maintenance therapy for moderate-to-severe psoriasis. The primary endpoint was the percentage of patients achieving PASI-75 at week 10. The secondary endpoints were PASI-50 and -90 at week 10. Selected patients had moderate-to-severe plaque psoriasis, PASI ≥12, BSA ≥10%, and were candidates for phototherapy and systemic therapy.
In the EXPRESS I trial, 378 patients were randomized to infusions of infliximab 5 mg/kg or placebo. Infusions were given at weeks 0, 2, 6, and then every 8 weeks up to week 46. Patients in the placebo group received placebo infusions at weeks 0, 2, 6, 14, 22 and then crossed over in a double-blinded fashion to receive infliximab 5 mg/kg at weeks 24, 26, 30, and every 8 weeks through week 46.
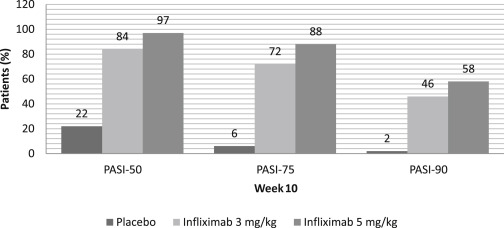
At week 10, 80% of patients achieved PASI-75 and 57% achieved PASI-90 in the infliximab group. Only 3% and 1% of the placebo group achieved PASI-75 and PASI-90, respectively. This response was maintained at week 24. By week 50, PASI-75 and PASI-90 had dropped to 61% and 45%, respectively. Similarly, 83% of patients attained a PGA of clear or minimal psoriasis by week 10, which dropped to 53% at week 50. This decline in clinical response was associated with low infliximab serum concentrations due to the development of anti-infliximab antibodies. Within the group of patients who achieved PASI-75 at week 10, only 39% who were antibody positive maintained this response through week 50 versus 81% and 96% of the antibody-negative and inconclusive patients, respectively. Being antibody positive did not preclude those patients from having a clinical response, although fewer antibody-positive patients achieved PASI-75 or a maintained clinical response.
Maintenance of clinical response was related to the ability to achieve stable serum concentrations of infliximab. Of the patients who maintained their PASI response through week 50, median preinfusion serum infliximab concentrations were 1.0 μg/mL or greater at weeks 30 and later. The patients who lost response by week 50 had median preinfusion serum infliximab concentrations 1.0 μg/mL or less. Among the patients who achieved PASI-75 at week 10, 89% of patients maintained this response at 6 months and 65% maintained this response at 1 year. No dose escalation or additional therapy was needed, apart from low-potency topical corticosteroids on the face or groin.
In summary, the EXPRESS trial demonstrated the efficacy, rapid onset, and maintenance of therapeutic benefit of infliximab as monotherapy in moderate-to-severe psoriasis. By week 6, about two-thirds of patients achieved PASI-75 and one-third achieved PASI-90, with proportions of these patients continuing to increase during the course of the study. By week 10, one-fourth of patients reported complete clearing. This study also demonstrated that maintenance treatment with infliximab 5 mg/kg every 8 weeks was effective in maintaining clinical response and well tolerated in most patients ( Fig. 9.3 ).
European Infliximab for Psoriasis Efficacy and Safety Study II
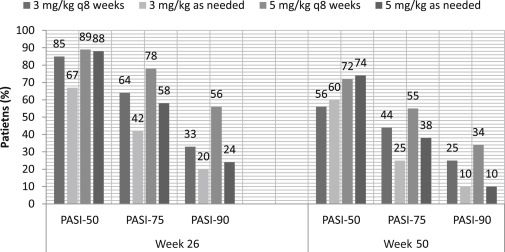
The EXPRESS II trial was a phase 3, randomized, double-blinded placebo-controlled trial that investigated the benefit of regular versus on-demand maintenance regimens of infliximab. The EXPRESS II trial was the first biologic study to directly compare maintenance regimens of continuous, every-8-week therapy versus intermittent, as-needed therapy.
The primary endpoint was the percentage of patients achieving PASI-75 at week 10. Other endpoints were improvement in the PGA and DLQI by week 10, and the average percentage improvement from baseline PASI between weeks 16 and 30 and weeks 16 and 50.
All patients had moderate-to-severe psoriasis, with a PASI ≥12 and 10% or greater BSA involvement. This study consisted of 835 patients who were assigned to infliximab 3 mg/kg, 5 mg/kg, or placebo at weeks 0, 2, and 6. At week 14, patients were randomized again to either continuous maintenance therapy or as-needed intermittent therapy at the same dose as the induction phase. In the maintenance group, patients received their assigned infliximab doses at 8-week intervals (weeks 14, 22, 30, 38). In the as-needed group, infliximab was given when the PASI improvement from baseline dropped to less than 75% at the weekly study visits. If PASI improvement was at least 75%, placebo was given. The placebo group was crossed over at week 16 to receive infliximab 5 mg/kg at weeks 16, 18, 22, and every 8 weeks until week 46.
At week 10, 70% and 75% of the 3-mg/kg and 5-mg/kg group had achieved PASI-75 versus 1.9% in the placebo group. PASI-90 was achieved by 37% and 45% of the 3-mg/kg and 5-mg/kg group versus 0.5% in the placebo group.
Efficacy for the maintenance phase was evaluated from week 14 through week 50. Among the week 10 PASI-75 responders, response was better maintained in the every-8-week continuous maintenance group as compared with the as-needed intermittent group. In the patients receiving 3 mg/kg, the median of the average percentage improvement in PASI was 80.6% for the every-8-week group versus 72.4% for the as-needed group. In the patients receiving 5 mg/kg, the median of the average percentage improvement in PASI was 89.6% versus 76.4% for the every-8-week group versus as-needed group. Among the week 10 PASI-75 nonresponders, 59.0% eventually achieved responder status at some point during the maintenance phase across all groups.
This study concluded that the patients in the 5-mg/kg every-8-week continuous therapy group maintained PASI responses better than the 5-mg/kg intermittent treatment group and 3-mg/kg groups. Of the patients assigned to 3 mg/kg, those who received infliximab every 8 weeks maintained PASI responses better than the intermittent treatment group as well. Although infliximab has been shown to provide excellent initial control, a maintenance regimen is important in sustaining adequate response. At the same time, long-term drug exposure should be limited by choosing the lowest effective dose or least frequent interval of drug administration ( Fig. 9.4 ).
Efficacy in psoriatic arthritis
Treatment options for PsA are wide ranging, with nonsteroidal anti-inflammatory drugs (NSAIDs) being used for mild cases and disease-modifying antirheumatic drugs (DMARDs) for more severe cases. However, many patients do not respond significantly, and thus, the anti-TNF agents are becoming increasingly popular.
The Infliximab Multinational PsA Controlled Trial (IMPACT) study was a phase 3, double-blinded, randomized, controlled trial that assessed the efficacy of infliximab for PsA in patients who had failed prior treatment with DMARDs or NSAIDs. The primary endpoint was American College of Rheumatology (ACR)-20 response at week 98. This study recruited 104 patients with active peripheral polyarticular joint involvement and was conducted in 3 stages. During stage 1, patients were assigned to placebo or infliximab 5 mg/kg at weeks 0, 2, 6, and 14. Stage 2 began at week 16, when the placebo group was crossed over to infliximab 5 mg/kg every 8 weeks through week 46. The original infliximab group continued to receive infliximab 5 mg/kg every 8 weeks until week 46. Stage 3 was an open-label extension from week 54 to week 98, during which patients received infliximab 5 mg/kg every 8 weeks.
At week 14, 58% of patients in the infliximab group and 11% in the placebo group achieved ACR20 response. By week 50, 74% of patients had achieved 20% or greater improvement in their ACR criteria. At week 98, 62% maintained this ACR20 response, with 45% and 35% achieving ACR50 and ACR70. At week 98, 82% or more of patients showed articular improvement, as evidenced by a moderate or good disease activity score (DAS) 28 response. DAS28 improved 48% and 43% from baseline at weeks 50 and 98, respectively. Physical function also improved, with Health Assessment Questionnaire scores approaching normal levels of functioning by week 50 (49% improvement from mean baseline of 1.1). Average annual radiographic progression was also significantly reduced in the infliximab group as compared with placebo. The modified van der Heijde-Sharp (vdH-S score) was used to score bone erosion, joint space narrowing, and total radiographic scores in 40 joints in both hands and 12 joints in both feet. At baseline, the mean estimated annual progression was 5.74 modified vdH-S points. At year 2 of infliximab treatment, the mean estimated progression was significantly reduced to 0.65 modified vdH-S points. One drawback of the modified vdH-S scoring system is that changes in just 1 joint of a patient may result in a higher score and overemphasize the disease course. This data together indicate that infliximab 5 mg/kg was able to maintain a significant, sustained clinical improvement in treatment-refractory PsA.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree