Fig. 4.1
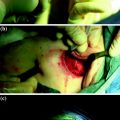
Cosmetic outcome 1 year after left video-assisted nipple sparing mastectomy for breast cancer in a 59-year-old-patient with medium/large, grade III ptosis (advanced ptosis: the nipple is below the inframammary fold and at the level of maximum breast projection). Immediate reconstruction was performed with definitive 360 cc anatomically shaped silicon prosthesis. Good symmetry in oblique a, b, frontal c view was achieved without the need for contralateral procedures; from the left lateral view d is visible the axillary scar
4.3 Endoscopic Breast Surgery for Breast Cancer
Oncological patients candidates to NSM with minimally invasive approach should share the same indication of open NSM. In fact, the techniques of new generation started by Sakamoto et al. [4], then revised by our team with development of brand new techniques [5], have been developed to reach the same surgical aim of conventional open NSM: whole breast-gland and nipple core dissection while skin and NAC are spared. From this point of view, oncological safety of this new MM should be at least equal to that of the open technique, with possible further benefits due to the dissection performed under direct magnified vision of the endoscope.
Since the major concern in NSM, independently from the technique, is the oncological safeness due to the risk of a local recurrence in the remaining ductal tissue beneath the nipple, many efforts have been made to know how to maintain the risk as low as possible. Pathological studies and prediction models to determine the likelihood of NAC occult involvement have been published with wide range of results. Tumor cells in the NAC have been found in 0–58 % of cases, even if most Authors report a 25–30 % (see Chap. 5). Furthermore, this finding has been related with many variables, such as tumor size, distance between the NAC and the neoplasia, multicentricity, tumor differentiation, positive axillary lymph nodes, central location, extensive intraductal component; however, the most consistent findings were T size and its distance from the NAC [6–8].
Those findings explain why NSM has emerged as an option for patients with small invasive and non-invasive cancers located far from the nipple-areola complex (NAC). Those indications are still in progress: more widespread use of NSM has broadened the spectrum of candidates which still differ among surgeons. However, high consensus is shared for several criteria.
1. Tumor size. Since small tumor size is also the basic condition for breast conserving surgery (BCS), NSM is offered in case of unfavorable ratio breast cancer/breast size, and when micromasty precludes BCS with good cosmetic results at all. Multifocal/multicentric infiltrating cancers with small peripheral nodules, large or multicentric non-infiltrating neoplasia are also oncological conditions suitable for NSM, since represent early diseases in which BCS is not feasible. Any tumor developed in a woman at high risk due to family history or proven deleterious mutation BRCA1-2 also represent a criterium which may lead to choose mastectomy rather than BCS. However, definitive consensus on T-size has not been already reached.
A simple rule has been demonstrated: the more the size, the more the percentage of NAC occult involvement. However, the clinical relevance of this finding and surgeon’s ability to obtain a radical nipple core has questioned the strictly application of such a rule. Presently, all Authors will agree in indication for in situ/T1 tumors; most for T < 3 cm. In recent series of open NSM the cutoff was 4 cm [7] and some Authors claim that size should no more be considered among selection criteria [9, 10].
The same heterogeneity in T size is mirrored in the early experience of MM (Table 4.1). Early reports from East of endoscopic subcutaneous mastectomy [11–17] usually include just intraepithelial/small infiltrating tumors, although Ito et al. considered eligible for such surgery T1–T3 patients [15]. In 2009, Sakamoto and colleagues reported on video-assisted NSM with the new oncological concept of careful dissection of the NAC performed in a heterogeneous patient population, including T2–T3 (5.6 cm) tumors (mean size 2.1 cm) [4]. Since such a widened inclusion criteria do not help in future comparison on oncological safety of endoscopic techniques as compared with open NSM, in our series eligibility for undergoing new techniques for MM was limited to either in situ disease or T < 3 cm in size, in order to match the most accepted criteria for open NSM [5].
Table 4.1
Early report of mini-invasive nipple spring mastectomy: a comparison of oncological indication and surgical approach
Years | Author | N. | Indications | Breast oncologic surgery | Access to the gland | Retro-areolar exam: % positive rate |
|---|---|---|---|---|---|---|
2002 | Kitamura et al. | 21 | BC | SM | Axilla 6 cm | No data |
2002 | Nakajima et al. | 17 | BC | SM, AD | Axilla 5–7 cm | |
2002 | Ho et al. | 9 | Early BC | SM, AD | Axilla 5–7 cm ± Areola | 0 % |
2006 | Yamashita and Shimizu | 2 | DCIS | SM | Lateral chest 5 cm | Ne |
2008 | Ito et al. | 33 | BC Tis, 3T1, 1T2 (2 postCHT) | SM, SNB ± AD | Axilla 5 cm + B skin | 24.3 % |
2008 | Yamaguchi et al. | 21 | BC | SM | Axilla 8 cm or IMF | No data |
2009 | Fan et al. | 43 | BC T < 3 cm (ok postCHT) | SM, AD | 3 B skin/ axilla 0,5 cm | No data |
2009 | Sakomoto et al. | 89 | BC T1–T3 | NSM, SNB, ± AD | Axilla 3–5 cm and Areola ± Nipple | 2.2 % |
2012 | Ferrari A, Sgarella A, Zonta S | 39 | BC, Tis, T < 3 cm RRS | NSM, SNB ± AD | 3–4 cm Axilla | 2.5 % |
2. Distance tumor-NAC. The preoperative distance tumor/NAC, measured by either clinical exam or breast imaging; mammogram, ultrasound, or magnetic resonance (this latter not obligatory performed) imaging, has always represented a selection criteria to safely spare the NAC. A simple rule does exist also for this parameter. It has been demonstrated that the greater the distance, the lower the risk of occult tumor in the NAC, although no evidence is available about the safety limit. Most Authors started their experience with 2 cm as the cut off distance [18] and this limit remains the most accepted worldwide, although from a recent review the ideal candidate for NSM should present with a small (<2.5 cm) tumor distant from the NAC 4 cm at least [1]. On the other hand, after many reports on the safety of NSM, many surgeons decided to widen women’s eligibility to the intervention by narrowing the distane tumor-NAC limit to 1 cm [8, 19] or less [9].
3. Retroareolar margin. To safely spare the NAC, the dissected retroareolar tissue, which represents the mastectomy margin, has to result free from tumor at pathologic exam [20]. This condition is considered mandatory by almost all Authors, even if Kneubil and coll. reported on a patient population with either false negative at frozen section (FS) or close margin treated without NAC surgical removal: in these conditions they report a loco-regional relapses rate “considerably low” [21].
The width of a safe retroareolar-free margin is on debate, as well as the issue of margins remains a hot topic in BCS; both in NSM and BCS margins are considered safe if ranging from 10 to 1 mm, although it is likely that the trend of “bigger is not better” suggested for BCS [22] will be applied in NSM too.
One more issue concern the need for intraoperative frozen section (FS) of the retroareolar margin in NSM. The benefit is that knowing positivity intraoperatively allows to remove the NAC in one-step operation, converting NSM into a SSM; in some Authors’ opinion this step is considered mandatory [1]. On the other hand, false negative results of FS have been reported [23] from all authors, requiring anyway a postoperative NAC excision which can be simply performed in ambulatory setting. Furthermore, the lack of standardization of intraoperative technique and pathologic sampling of retroareola tissues result in heterogeneous and partially unreliable results. Freezing artifact, loss of tissue during FS, and the potential for false negative results has led some surgeons to prefer waiting for formal paraffin-fixed analysis of the nipple core and retroareolar tissue [24]. In case of well-selected patient population the permanent pathologic result of retroareolar tissue will show a low (0–18 %) [7] percentage of positive margins (9 % of 121 therapeutic NSM at MSKKC) [8], so both frozen section and permanent pathology to examine retroareolar tissue remain good options. Furthermore, in follow-up studies local recurrences were not increased in NSM as compared with SSM and M [18], and usually located under skin-flap. Recurrence in the nipple is an unusual finding, which can be easily and successfully treated just with local NAC removal [25].
Among MM, few data can be already obtained, due to few numbers, lack of data and different techniques (Table 4.1). Early reports from East on SM never describe frozen section of tissue beneath the NAC and only Ito et al. give information on the status of retroareolar margin: 24.3 % of tumor involvement, which required surgical excision postoperatively [15]. Such a high rate would be considered unacceptable as compared to the the lower finding in most reported open NSM; it can be considered a consequence of omission of NAC dissection when SM is performed in oncological patients. The only two experiences of endoscopic NSM, reported by Sakamoto et al. and by our team, succeed in obtaining a low rate (2.2 % and 2.5 %, respectively) of positive retroareolar margins [4, 5].
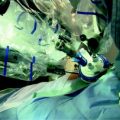
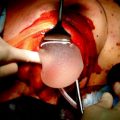
In our surgical technique retroareolar margin was marked at the apex of the nipple core immediately after dissection with a clip and a stich, and colored along the NAC projection on dissected gland tissue in order to facilitate an accurate pathologic exam of retroareolar tissue (Fig. 4.2). We choose postoperative exam by permanent sections; NAC preservation was considered safe if the underneath tissue was free from cancer for at least 5 mm in depth. If this condition was not satisfied, NAC was removed postoperatively.
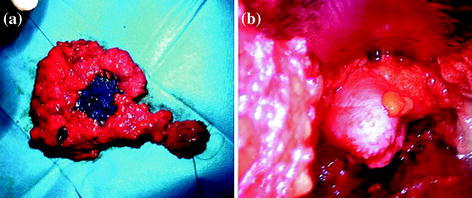

Fig. 4.2
a Endoscopic internal vision of everted nipple after hydrodissection. Magnificated details of the major galactophorous ducts resected on nipple tip may be well recognized. b Mastectomy specimen: a stich has substituted the clip on nipple core and the areola area has been colored, thus to facilitate the retroareolar discoid examination by the pathologist
4. Axillay lymph nodes, Multicentricity. The likelihood of NAC occult involvement has been much reviewed in the literature. Among related variables positive axillary lymph nodes and multicentric disease have been reported, although other studies have not confirmed these risk factors [6–8]. Among heterogeneous criteria used by Authors in patients’ selection for NSM, positive axillary nodes have been considered a contraindication for some Authors [1], but not for Others [7, 9]. Including N-positive patients and/or multicentric disease for NSM does not seem to increase the local relapse, so that the trend is likely toward extending criteria also for this patient population.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree