Modern breast implant design emphasizes the host response at the surface. Implant surfaces are characterized by their roughness, surface area, and potential for bacterial attachment. The future of implant design may lie in the ability of bioengineers to transform both the structure and chemical properties of the device surface and therefore affect potential long-term outcomes.
Key points
- •
Breast implant surface technology has evolved over the last 50 years.
- •
New biomaterials are designed to create a specific host response.
- •
Shell surface design varies by manufacturer.
- •
Multiple classification systems define surfaces according to roughness, surface area, and attachment of bacteria.
- •
Surface engineers aim to alter shell surfaces topographically and chemically to modify the host–implant interface.
Why implant surfaces have evolved
The engineers of new biomaterials, including those used in aesthetic and reconstructive surgery, strive to develop implantable materials that are capable of producing a specific host tissue response. Advances in surface science instrumentation over the last 30 years have allowed bioengineers to evaluate the molecular events occurring at the biomaterial host interface. The early innovations in breast implant surface technology were focused on reducing device failure and gel bleed. One of the leading drivers in the development of novel textured surface breast implants by many manufacturers was a response to capsular contracture rates, reported to be as high as 40% to 50% with early generation smooth breast implants ( Fig. 1 ). Unfortunately, capsular contracture remains one of the most common reasons for reoperation today and a large number of potential etiologies have been proposed over the last 50 years. , We now also understand that the surface features of an implant play a more central role in the host response. Surface characteristics affect the local microenvironment and should be included along with discussions concerning pocket location, atraumatic surgical techniques, incision location, and the use of local and systemic anti-infective agents as we look to develop successful long-term outcomes in breast implant surgery.
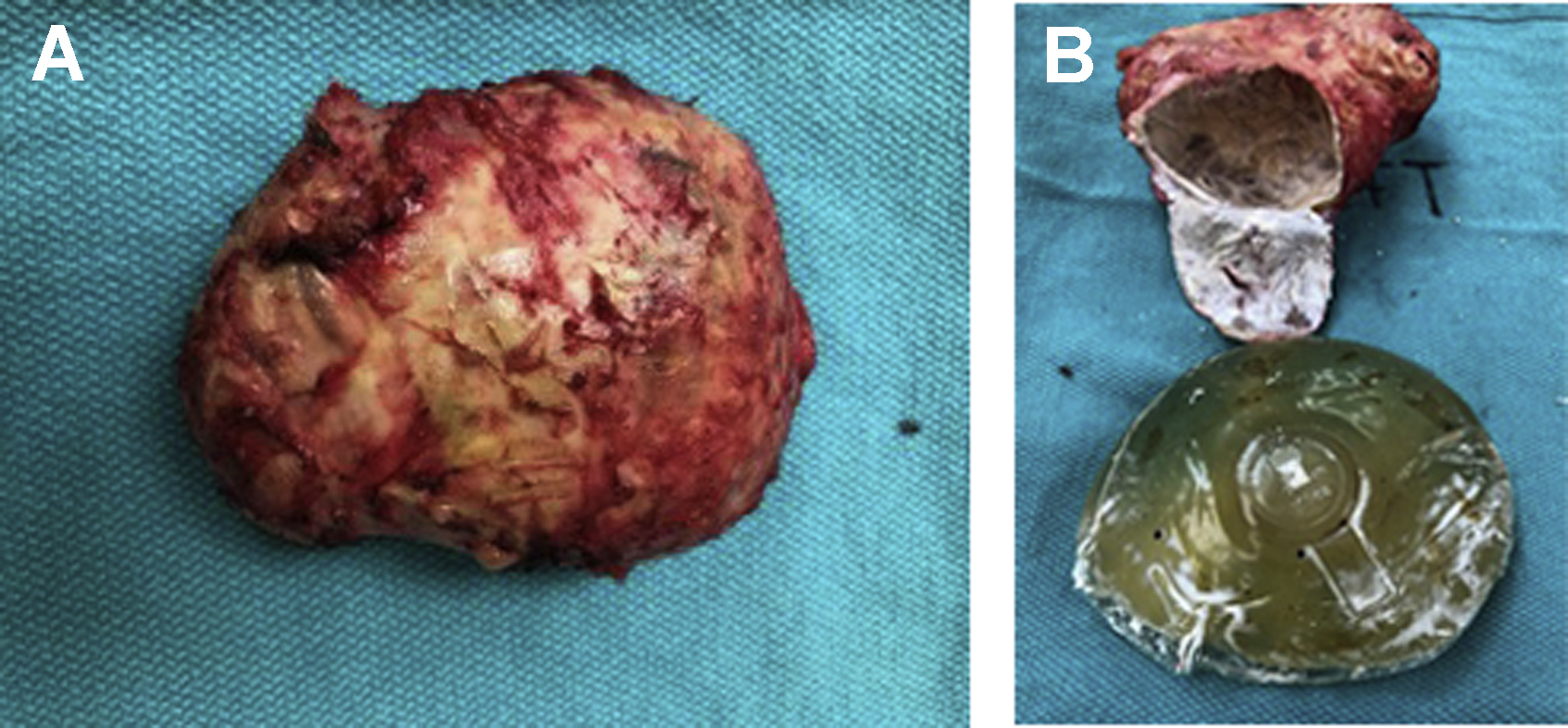
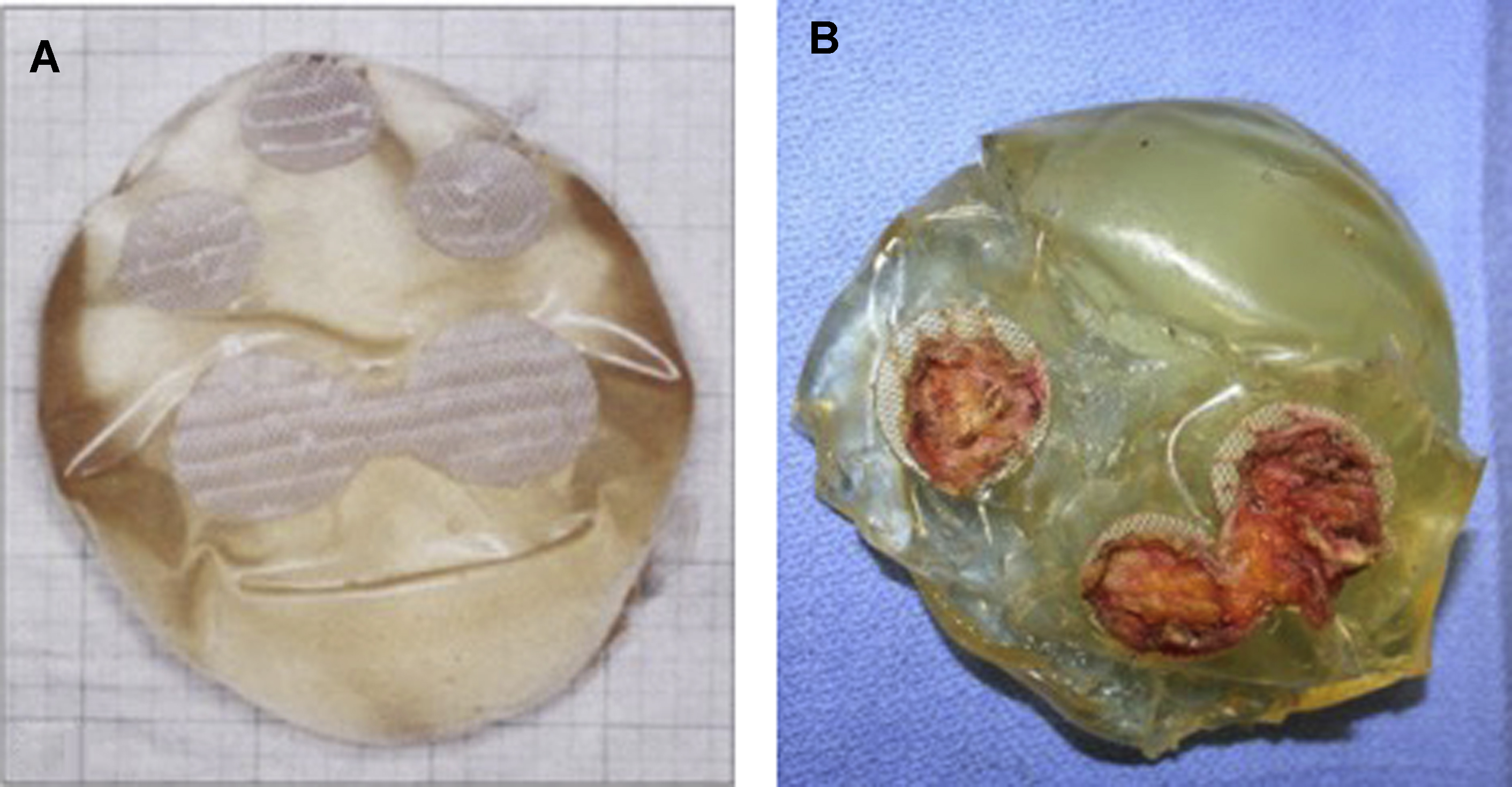
The concept of texturing breast implants and tissue expanders arose in the late 1980s out of a search to decrease the high incidence of reoperations specifically for capsular contracture. Initially, layers of polyether, polyester, or polyurethane foam materials were applied to the backside of implants so that tissue ingrowth might maintain the implant’s position. Dow Corning introduced their second-generation device in 1968 (Cronin Silastic 0) with Dacron fixation patches on the posterior surface. The patches varied in position and were arranged to encourage tissue ingrowth and position stability ( Fig. 2 ). The concept that texture might also prevent capsular contracture was founded on the lower rate of capsular contracture seen in patients with polyurethane foam devices. , The belief at the time was that the haphazard shell surface decreased the incidence of capsular contracture. In addition to potentially lowering the capsular contracture rate, surgeons at the time also wanted a tissue expander that could facilitate the creation of a well-defined inframammary fold and maintain its position on the chest wall during expansion. Manufacturers revisited the patch concept, but with a different approach. Although the manufacturers of polyurethane-coated breast implants never returned to the United States after the 1991 US Food and Drug Administration (FDA) moratorium on silicone breast implants, they have remained popular throughout the rest of the world.
Implant manufacturing and unique shell characteristics
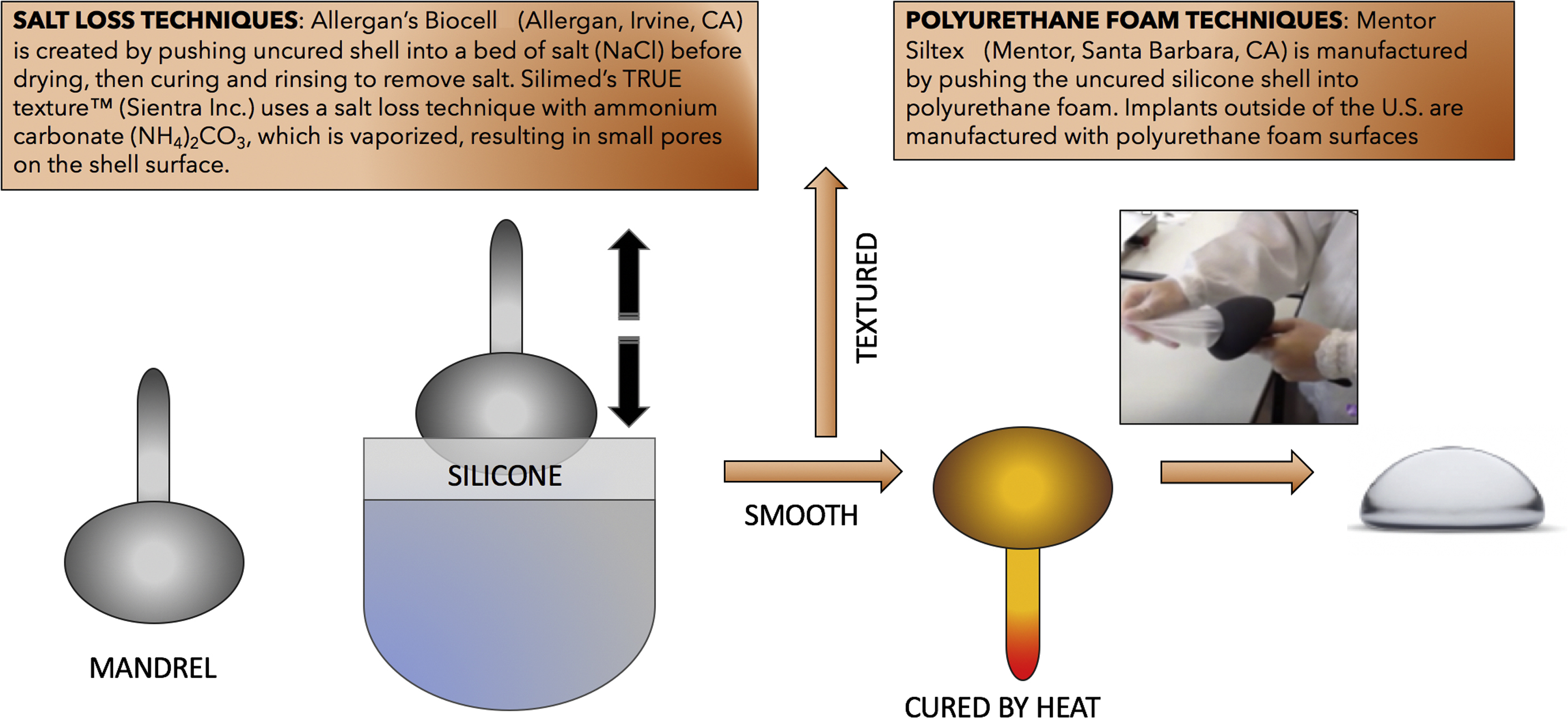
The manufacture of all implants begins with a specific mandrel being repeatedly dipped in or coated with an elastomeric compound (polyurethane, silicone, or latex) dispersed in a solvent, generally xylene. Between each coating, the mandrel is gently heated to allow the solvent to evaporate and let the elastomeric shell set or cure. It is during the process of curing that the silicone polymers are transformed into an elastic through a covalent crosslinking reaction. Because there are several layers to an implant shell, each manufacturer has developed their own unique method to create texture on the surface of the outermost layer. The concept of texturizing breast implants began with the use of polyurethane foam to coat implants. These implants were found to diminish the risk of contracture for a longer duration than any other textured or smooth device at the time. There were clear problems with the early polyurethane-coated implants because the foam separated from the shell and made explantation extremely difficult. To avoid the potential problems associated with the foam-covered implants and still take advantage of the lower incidence of capsular contracture associated with the use of these implants, inventors sought novel mechanisms to create an open cell texture on the surface of elastomeric shells ( Fig. 3 ). The first patent for the texturization of a breast implant surface was filed in 1989 by William Dubrul and Patrick Maxwell , (Allergan Biocell, Allergan Medical Corporation, Irvine, CA). Other manufacturers independently developed alternate methods to produce textured surfaces on their shells. Biocell is created by dipping the mandrel into uncured silicone and, before the surface dries, it is pressed into fine granular salt and cured in laminar flow. Brushing away the salt particles creates the open pores on the surface of the shell. Other textured surfaces were developed by alternative methods that did not require the application of additives to the surface. The surface of the Mentor Siltex (Mentor Corporation, Santa Barbara, CA) implant is created by imprinting or pressing the uncured silicone into a polyurethane foam to create a stamped surface with a comparable configuration. The Sientra True’s texture is created using ammonium carbonate grains layered over their surface. The particles are then vaporized into a gas leaving small open pores on the surface. More recently, Establishment Labs (Alejuela, Costa Rica) created an inversion process whereby the surface characteristics are on the mandrel and the cured shell is flipped inside out. Other patents have followed over the years, some with novel ideas such as bombarding the outer surface of an implant with solid carbon dioxide (dry ice) to create dimples (Mentor Worldwide, LLC 2006), but many innovative surface technologies have yet to make it to the US market.
Defining surface characteristics: topography and cell surface interactions
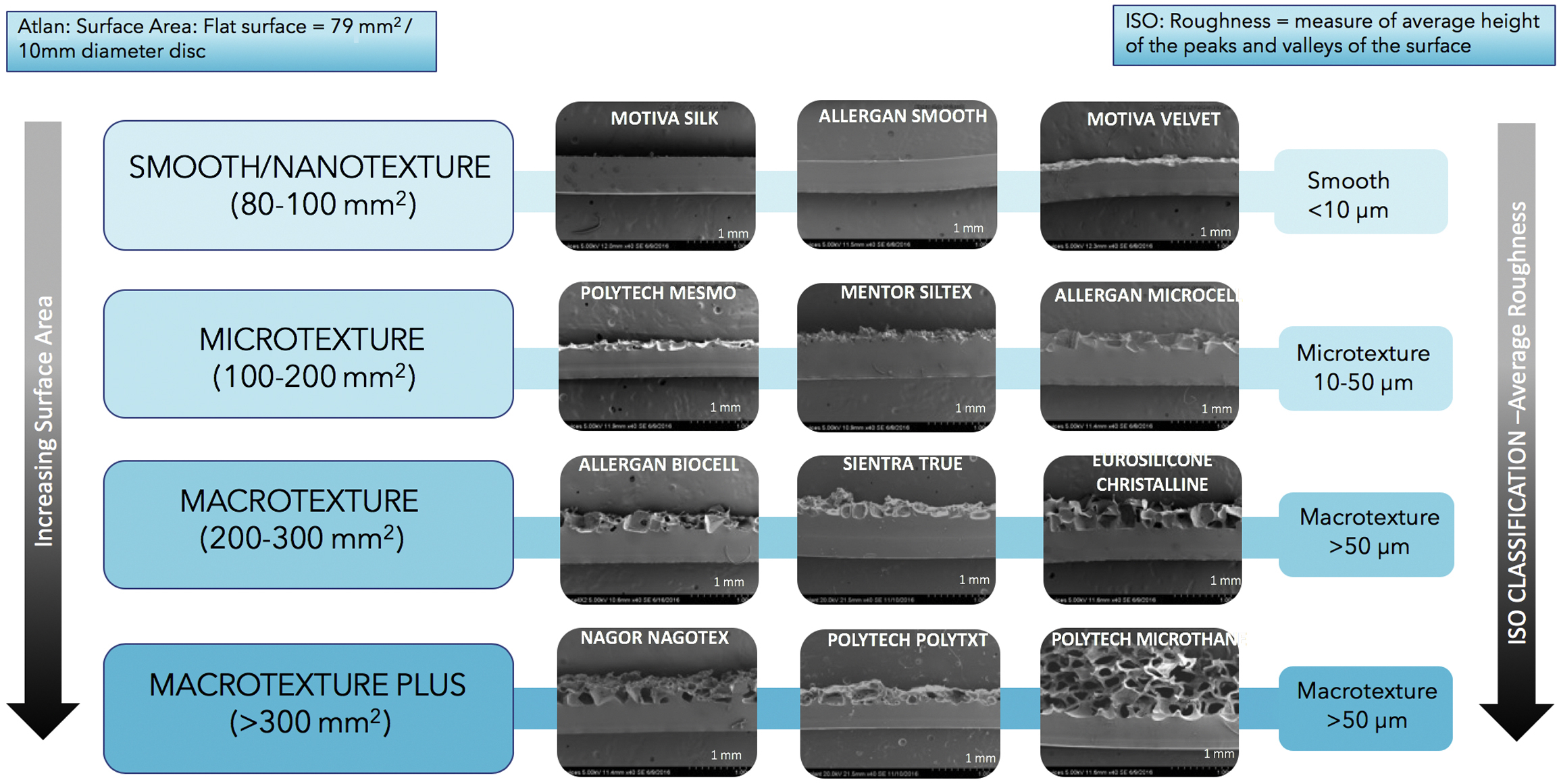
Most biological reactions in biology and medicine occur at surfaces and interfaces. The field of surface science provides us with an understanding of how the specific surface properties of a material may control the cellular response. The ultimate goal in implant surface design would be an entirely biocompatible device, with biocompatibility defined as “The ability of a material to perform with an appropriate host response in a specific application”. Early studies of surface textures were focused on the gross morphology and structure of the implant. Manufacturers, researchers, and independent organizations, such as the International Organization of Standardization (ISO), have developed surface classification systems. The ISO classification of surface textures has been the global standard used by most regulatory bodies such as the FDA and European Medicines Agency to evaluate, approve, and categorize breast implants. Under ISO 14607:2018, surface textures are classified into 3 categories based on their average surface roughness: smooth, less than 10 μm; microtexture, 10 to 50 μm; and macrotexture, greater than 50 μm. Roughness defines the contour irregularities on a surface and is measured by the variations in height of the peaks and troughs relative to a reference plane. Changes in the surface roughness influence host cell response. Other more recent studies have looked to include additional, more specific surface characteristics in any classification system, such as implant surface area as well as roughness. Manufacturing techniques vary not only in their surface roughness, but also with regard to their specific shell surface area. As the surface area increases, host cells attach more readily. More detailed surface characteristics can be analyzed using scanning electron microscopy. When classified by surface area, excluding exact pore size and number, surfaces can be grouped based on similarities in texture appearance and pore depth. One such classification of textures creates 4 groups: smooth or nanotexture, microtexture, macrotexture, and macro-plus texture. ( Fig. 4 ) Using newer microcomputed tomography scans to examine implant surfaces with increased precision, Deva and colleagues propose a third classification system of implant surfaces. His group divides shell surfaces into 4 grades, namely, high, intermediate, low, and minimal. They further link the implant surface characteristics, roughness and surface area, to the potential for bacterial growth ( Fig. 5 ). Their analysis of the varying implant surfaces demonstrates a significant correlation between increasing surface area and bacterial attachment and growth ( Fig. 6 ).