div class=”ChapterContextInformation”>
2. Isolated Human and Animal Stratum Corneum as a Partial Model for the 15 Steps of Percutaneous Absorption: Emphasizing Decontamination Part II
Keywords
Stratum corneumChemical bindingRubbing15 steps related to the process of percutaneous absorptiona
Number | Factors determining percutaneous absorption |
|---|---|
1 | Release from vehicleb Varies with solubility in vehicle, concentration, pH, etc. |
2 | Kinetics of skin penetrationb Influenced by anatomical site, degree of occlusion, intrinsic skin condition, animal age, concentration of dosing solution, surface area dosed, frequency of dosing, post absorption, etc. |
3 | Excretion kineticsb |
4 | Tissue disposition |
5 | Substantivity to skinb |
6 | Wash effectsb Wash resistance Wash enhancement |
7 | Rub effectsb Rub resistance Rub enhancement |
8 | Transfer––skin, clothing + inanimate surfaceb |
9 | Exfoliationb |
10 | Volatilityb |
11 | Binding––all layersb |
12 | Anatomic pathways |
13 | Lateral spreadb |
14 | Vascular perfusionb |
15 | Cutaneous metabolismb |
The abovementioned observations provide novel insights for improving decontamination techniques. Despite many emergency treatment protocols, numerous topically applied chemicals are not easily removed by water washing. Skin decontamination is the primary required intervention for chemical, biological, and radiological exposures and involves immediate removal of the contaminant via the most effective manner. Contaminant removal techniques can consist of physical removal including the use of friction, use of a liquid vehicle to solvate or emulsify the contaminant, contaminant transfer to another medium through absorption/adsorption, and chemical alteration of the contaminant. Decontaminants should effectively and rapidly remove contaminants without causing enhancement, wash-in effect, or skin damage while easily removing itself without undesirable residue. It should additionally be readily available, affordable, and easily disposed of [8]. In the second part of this review, we continue to explore the many steps involved in percutaneous penetration, the laboratory techniques providing the background for this knowledge, and the insights advancing our knowledge of chemical exposure risk assessment, exposure prevention and barrier methods, and postexposure decontamination. We address knowledge gaps to guide future research to guide our ability to minimize the harmful effects of hazardous dermal chemical exposure.
Tissue Disposition and Binding: All Layers
The skin is an organized, heterogeneous, and multilayered organ. The SC, epidermis, dermis, appendages, and vasculature constitute the outer living protective system. Percutaneous absorption of topically applied agents is the sum of the penetration and permeation of a chemical through the SC, epidermis, and part of the dermis [10, 22, 32]. The effect of the skin layers on percutaneous absorption was evaluated by quantifying the partitioning of compounds in water and isopropyl myristate (IPM) between skin layers. The influence of vehicle drug concentration, equilibration time, hydrophilicity, lipophilicity, and pH on the partitioning behavior of compounds was examined [13].
Chemical Binding and Dispersion to Different Skin Layers
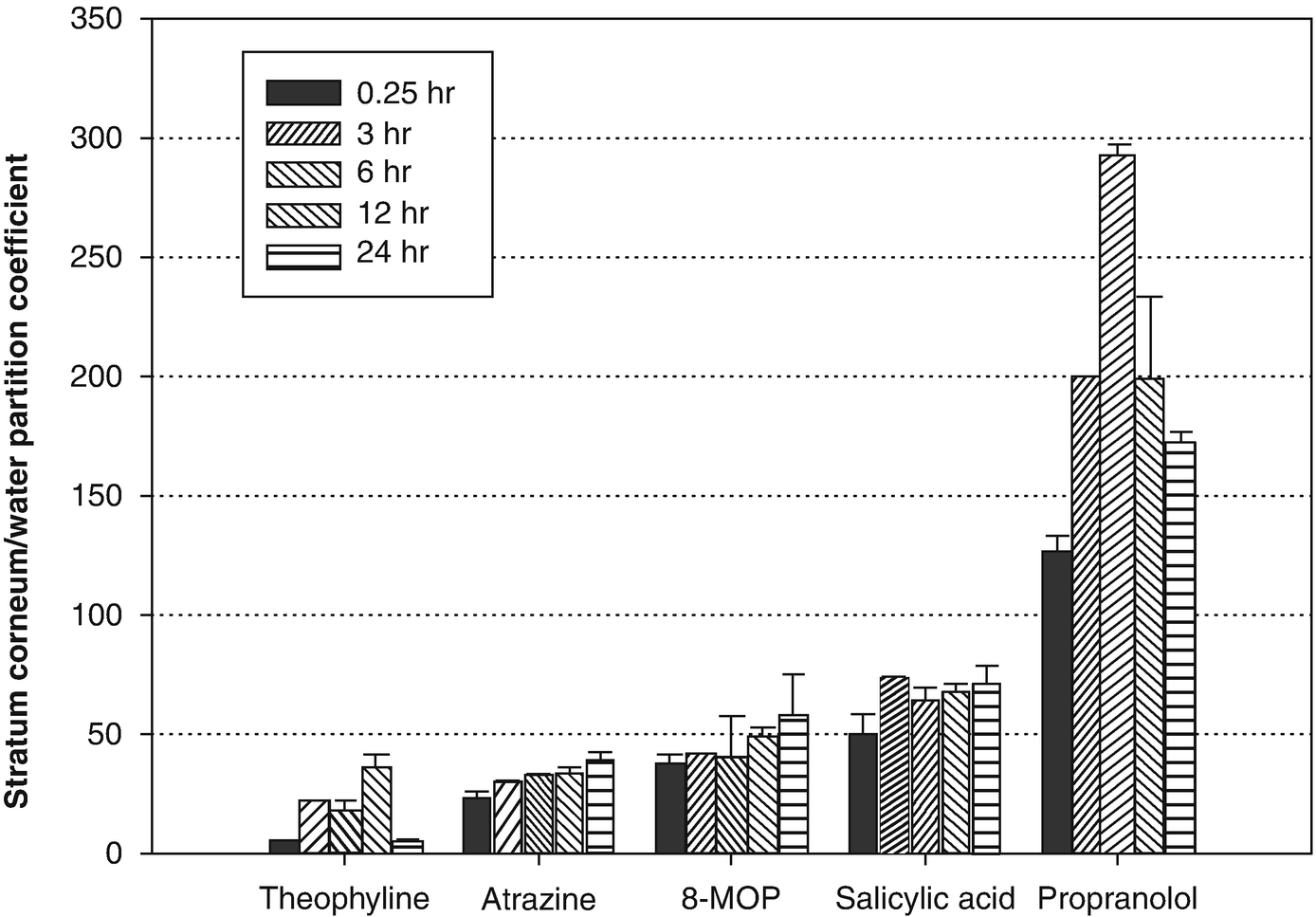
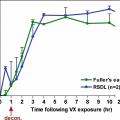
The graph is modified with permission from Gogoleva et al. [13]. Stratum corneum (sheet)/water partition coefficient (mean ± SD, n = 3) of model compounds as a function of equilibrium time. In the stratum corneum, the lipophilic chemicals propranolol, atrazine, and salicylic acid reached equilibrium in 3–6 hours. The hydrophilic compound theophylline required 12 hours to achieve equilibrium. Solutions of chemicals used were at saturation solubility concentrations. The lipophilic chemicals, propranolol, atrazine, and salicylic acid, reached equilibrium in 3–6 hours. The most hydrophilic compound, theophylline, required 12 hours to achieve equilibrium
The data presented here corroborates that in previous literature reports. The calculated partition coefficients for the compounds as a function of equilibration time, chemical concentration, and solvent nature may be valuable in predicting the in vivo and in vitro transport of drugs and environmental agents through human skin.
Raykar et al. [26] suggested that a lipid and protein domain exist in the SC. The uptake of highly lipophilic compounds (log P values near 3.0) may be governed by the lipid domain of SC, and hydrophilic solutes are taken up in the protein domain. Because theophylline partitions equally into the skin layers, the retention could be secondary to its binding to the protein domain. The data presented here verify earlier observations [5, 7, 17].
Wash Effects and Skin Decontamination
The Powdered Stratum Corneum Model to Rapidly Evaluate Wash Resistance and Decontamination Efficiency
Most material safety data sheets (MSDS) recommend soap and water to remove chemicals from the skin surface. Because of the varying physical-chemical properties, each chemical has different SC binding sites with diverse affinities. Therefore, understanding a chemical’s wash resistance can help to identify optimal decontaminants.
The powdered SC model provides a tool to rapidly evaluate the chemical’s potential skin binding and partitioning behavior and to determine washing solution efficacy. A radiolabeled contaminant chemical was mixed with powdered SC and incubated in a 37 °C water bath for 0.5–2 hours. The chemical and SC mixture was washed multiple times with the selected solution and centrifuged to separate the washing solution from the SC pellet, and then the radioactivity of each sample was measured. Using this protocol, the decontaminant effects of multiple decontaminants could be compared [16].
Decontamination Efficiency Related to Chemical Dose-Exposure Time
Water decontaminates environmental hazardous chemicals (2,4-d) from human skina
Dose (μCi) | Radioactivity recovery as percent dose of [14C]-2,4-D (%) applied | ||||||
|---|---|---|---|---|---|---|---|
Supernatant | Powdered stratum corneum | ||||||
Before washing | After first washing | After second washing | After third washing | After fourth washing | After last washing | ||
0.22 | 87.4 ± 0.5 | 12.6 ± 0.5 | 4.8 ± 0.8 | 4.5 ± 0.8 | 2.2 ± 0.2 | 0.9 ± 0.1 | 0.9 ± 0.1 |
0.44 | 84.6 ± 1.9 | 15.4 ± 1.9 | 6.8 ± 1.6 | 4.4 ± 0.5 | 2.6 ± 0.4 | 1.1 ± 0.2 | 1.1 ± 0.1 |
0.88 | 82.9 ± 1.9 | 17.1 ± 1.9 | 8.3 ± 2.0 | 4.9 ± 0.7 | 2.3 ± 0.5 | 1.1 ± 0.2 | 1.1 ± 0.1 |
1.76 | 86.1 ± 1.2 | 13.9 ± 1.2 | 5.5 ± 0.7 | 4.2 ± 0.7 | 2.5 ± 0.6 | 1.2 ± 0.2 | 1.1 ± 0.2 |
2.20 | 78.9 ± 1.8 | 20.1 ± 1.8 | 10.9 ± 2.9 | 4.6 ± 1.2 | 2.4 ± 0.3 | 1.4 ± 0.3 | 1.2 ± 0.3 |
3.52 | 81.7 ± 4.1 | 18.3 ± 4.1 | 8.7 ± 4.0 | 5.0 ± 0.6 | 2.0 ± 0.2 | 1.6 ± 0.5 | 1.5 ± 0.5 |
Comparison of Decontamination Efficiency Post Short-Time Exposure
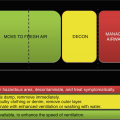
Potential environmental exposures may happen during manufacturing, distribution, application, deliberate exposure, or by residual presence. The concentrations of chemical agents and the contact duration are primary determinants of the degree of skin destruction and systemic poisoning. To prevent and diminish chemical absorption, immediate washing with appropriate decontaminating agents is essential to reduce damage and percutaneous penetration.
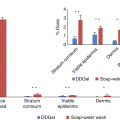
Glyphosate is a broad-spectrum, postemergence, translocated herbicide. Human exposure can occur through production or general use. The extent of glyphosate binding, systemic absorption, and removal post exposure was studied.
Glyphosate partition from roundup vehicle to powdered stratum corneuma
Radioactivity recovery as percent dose of [14C]-glyphosate applied | |||
|---|---|---|---|
Undiluted | Diluted 1:20 | Diluted 1:32 | |
30 minutes exposure | |||
Vehicle | 96 ± 0.9 | 102 ± 1.2 | 96 ± 1.6 |
Water washing | 2.4 ± 0.2 | 1.2 ± 0.07 | 1.1 ± 0.2 |
Stratum corneum | 0.04 ± 0.04 | 0.03 ± 0.03 | 0.02 ± 0.01 |
4 hour exposure | |||
Vehicle | 96 ± 2.4 | 104 ± 1.3 | 91 ± 2.9 |
Water washing | 3.3 ± 0.8 | 1.2 ± 0.2 | 5.0 ± 2.2 |
Stratum corneum | 0.01 ± 0.01 | 0.02 ± 0.03 | 0.05 ± 0.04 |
8 hour exposure | |||
Vehicle | 96 ± 0.4 | 98 ± 6.6 | 95 ± 1.4 |
Water washing | 3.0 ± 0.2 | 2.2 ± 0.5 | 2.3 ± 1.2 |
Stratum corneum | 0.02 ± 0.0 | 0.01 ± 0.01 | 0.02 ± 0.01 |
Washing with water or water with soap is traditionally used to reduce potential damage and percutaneous penetration of chemicals topically applied, but may not be effective for decontamination of lipophilic compounds. For instance, the removal of alachlor with water is less effective than soap with water. In contrast, both soap-and-water wash and emergency water shower are relatively ineffective at removing the potent contact sensitizer methylene bisphenyl isocyanate from the skin [45]. Residual chemical on the skin post standard washing procedures can have toxic consequences [19]. Additionally, the “wash-in” effect can potentially enhance percutaneous penetration and systemic absorption of chemicals [21].
Human skin decontamination of glyphosate in vitroa
Radioactivity recovery as percent dose of [14C]-glyphosate applied | |||
|---|---|---|---|
1 minutes exposure | 3 minutes exposure | 30 minutes exposure | |
Water washing | |||
Wash solution | 93.52 ± 16.42 | 94.60 ± 26.39 | 85.09 ± 14.06 |
Stratum corneum | 0.10 ± 0.10 | 0.14 ± 0.10 | 1.78 ± 0.84 |
Skin residue | 2.21 ± 1.99 | 0.70 ± 0.44 | 7.50 ± 3.82 |
Systemic absorption | 0.04 ± 0.05 | 0.05 ± 0.10 | 2.26 ± 2.19 |
Hypertonic saline | |||
Wash solution | 101.23 ± 14.20 | 98.69 ± 21.16 | 85.04 ± 14.62 |
Stratum corneum | 0.12 ± 0.07 | 0.14 ± 0.09 | 1.54 ± 1.06 |
Skin residue | 1.02 ± 1.27 | 1.45 ± 1.69 | 5.06 ± 3.29 |
Systemic absorption | 0.04 ± 0.04 | 0.54 ± 0.84 | 3.17 ± 6.66 |
Isotonic saline | |||
Wash solution | 98.72 ± 18.34 | 95.56 ± 28.08 | 78.95 ± 9.38 |
Stratum corneum | 0.08 ± 0.05 | 0.14 ± 0.11 | 2.23 ± 2.10 |
Skin residue | 0.74 ± 0.90 | 3.53 ± 5.15 | 9.84 ± 12.58 |
Systemic absorption | 0.07 ± 0.10 | 0.32 ± 0.39 | 5.42 ± 7.07 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree