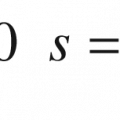div class=”ChapterContextInformation”>
1. Isolated Human and Animal Stratum Corneum As a Partial Model for the 15 Steps of Percutaneous Absorption: Emphasizing Decontamination, Part I
Keywords
Stratum corneumNatural moisturizing factorChemical warfare agentsRougier methodIntroduction: 15 Steps of Percutaneous Absorption
The stratum corneum (SC) of humans and animals holds key insights to the development of an efficient protective barrier against contamination and to devising effective decontamination interventions. SC constitutes the main barrier to the absorption of molecules while participating in the homeostasis of the organism particularly by limiting outward movement of water.
Current understanding no longer considers the SC of the skin barrier as dead “brick and mortar”, but rather a dynamic, living defense system. For instance, the acid mantle, a fine film with a slightly acidic pH on the surface of the skin, plays an integral role in making the skin less permeable to water and polar compounds and is formed in higher amounts with increased activity of enzyme phospholipase A2 [20, 57]. Additionally, acute perturbation of SC by organic solvents, detergents, or tape stripping resulting in lipid removal initiates a sequence of biological responses including epidermal lipid synthesis acceleration to rapidly restore the skin lipid content and barrier function [4].
15 Steps related to the process of percutaneous absorptiona
Number | Factors determining percutaneous absorption |
|---|---|
1 | Release from vehicleb Varies with solubility in vehicle, concentration, pH, etc. |
2 | Kinetics of skin penetrationb Influenced by anatomical site, degree of occlusion, intrinsic skin condition, animal age, concentration of dosing solution, surface area dosed, frequency of dosing, post absorption, etc. |
3 | Excretion kineticsb |
4 | Tissue disposition |
5 | Substantivity to skinb |
6 | Wash effectsb Wash resistance Wash enhancement |
7 | Rub effectsb Rub resistance Rub enhancement |
8 | Transfer––skin, clothing + inanimate surfaceb |
9 | Exfoliationb |
10 | Volatilityb |
11 | Binding––all layersb |
12 | Anatomic pathways |
13 | Lateral spreadb |
14 | Vascular perfusionb |
15 | Cutaneous metabolismb |
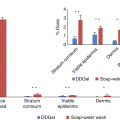
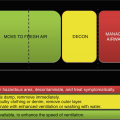
The abovementioned observations offer us new insights for improving decontamination techniques. The recommended emergency treatment of many chemical sprays and splashes to the skin consist of rinsing or washing with water and/or soap. However, many topically applied chemicals are not easily removed by water washing. Skin decontamination is the primary required intervention for chemical, biological, and radiological exposures and involves immediate removal of the contaminant via the most effective manner. Such removal techniques include physical removal of the contaminant, solvating or emulsifying the contaminant in a liquid vehicle, transferring the contaminant to another medium through absorption/adsorption of the chemical, chemical alteration of the contaminant, and the use of friction to dislodge the contaminant.
Ideally, a decontaminant effectively and rapidly removes the contaminant of interest, easily removes itself without undesirable residue, does not cause enhancement or wash-in effect, and does not damage the skin. Additionally, it is readily available, affordable, and easily disposed of [7]. In the first part of this review, we explore the many steps involved in percutaneous penetration, the laboratory techniques that provided the background for this knowledge, and the insights advancing our knowledge of chemical exposure risk assessment, exposure prevention and barrier methods, and postexposure decontamination.
Stratum Corneum
Stratum Corneum and Barrier Function
Stratum corneum structure and function
Name | Location—composition | Function |
|---|---|---|
Corneocyte | Cornified envelope: outer surface of the corneocyte Keratin filaments; γ-glutamyl isopeptide bonds | Mechanical barrier (impact and shear resistance) Resiliency of stratum corneum |
Cytosol: filaggrin proteolytic product; glycerol | Hydration | |
Cytosol: cis-urocanic acid (histidase activity) | Electromagnetic radiation barrier | |
Cytosol: cytokine activation; Proteolytic activation of pro IL-1α/β | Initiation of inflammation | |
Extracellular matrix | Lamellar bilayers: ceramides, cholesterol, nonessential fatty acids, proper ratio | Permeability |
Corneodesmosomes Intercelluar DSG1/DSC1 homodimers | Cohesion (integrity)/desquamation | |
Lamellar bilayers: antimicrobial peptides, FFA, sph | Antimicrobial barrier (innate immunity)a | |
Extracellular lacunae: hydrophilic products of corneodesmosomes | Toxic chemical/antigen exclusion | |
Lamellar bilayers: cholesterol, FFA, secreted vit. E, redox gradient | Antioxidant | |
Lamellar bilayers: barrier lipids | Psychosensory interface | |
Natural moisturizing factors | Within SC | Water holding capacity of SC |
pH and calcium gradients | Within SC and all through epidermis | Provides differentiation signals and lamellar granule secretion signals |
Specialized enzymes (lipases, glycosidase, proteases) | Within lamellar granules and all through epidermis | Processing and maturation of SC lipids, desquamation |
Melanin granules and “dust” | Produced by melanocytes of basal layer, melanin “dust” in SC | UV protection of skin |
The structure of SC is synonymous with that of “brick and mortar.” The bricks are corneocytes, protein complexes of tiny keratin threads in an organized matrix, and keratin is capable of holding large amounts of water between the fibers. SC contains approximately 12–16 layers of corneocytes, each with a mean thickness of 1 micrometer that varies with age, anatomical location, and UV radiation exposure [27].
Corneocytes are surrounded by crystalline lamellar lipid regions. Lamellar bodies are formed in the keratinocytes of the stratum spinosum and stratum granulosum layers, and as keratinocytes mature and ascend, enzymes degrade the lamellar body envelopes, releasing free fatty acids and ceramides that fuse to form a continuous lamellar lipid bilayer. This lipid bilayer is analogous to a “mortar” and is essential in maintaining the barrier properties of the skin [28, 36].
Each corneocyte is surrounded by an insoluble protein envelope primarily composed of two proteins, loricrin and involucrin, that have extensive structural links with one another. Cell envelopes are either “rigid” or “fragile” depending on the envelope’s interaction with the lamellar bilayer [28, 36].
Attached to the cell envelope is a layer of ceramide lipids that repel and trap water. This assembly prevents the absorption of water into the lower layers of the epidermis.
The corneocytes are held together by corneodesmosomes that function as “rivets” and are the primary structures that must be degraded for desquamation [28, 36].
Natural moisturizing factor (NMF), a collection of water-soluble compounds specific to the SC, represents approximately 20–30% of the dry weight of the corneocyte. NMF components absorb water molecules from the atmosphere to maintain SC hydration, but their water solubility allows them to easily leach from the cells upon water contact. Repeated contact with water can dehydrate the skin; however, the lipid layer surrounding the corneocyte helps to form a seal to inhibit NMF loss [37].
The stratum basale consists of a single layer of columnar epidermal stem cells attached to the basal lamina via hemidesmosomes. The stratum spinosum just above is rich with progressively enlarging lamellar bodies with ongoing keratin synthesis and lipogenesis. The stratum granulosum is next with mature lamellar bodies capable of differentiating into corneocytes. Here, the intracellular organelles undergo self-destruction, and the packaged lipid in the lamellar granules (LG) is released to the intercellular space. SC forms the outermost seal with 18–21 cell layers of dead corneocytes and lipids that is 20–40 micrometers thick in humans.
Filaggrin, a key protein in the hydration and formation of the SC barrier , acts as a source of hygroscopic amino acids and their derivatives including NMF [28, 36, 37].
Stratum Corneum Hydration
Normal skin has a consistent water content of 5–15%, independent of variations in environmental humidity. The superficial SC plays a dual barrier role of minimizing transepidermal water loss (TEWL) and preventing the entrance of external molecules. Hydration fluctuations alter SC permeability, and percutaneous absorption may be enhanced with increasing SC water content. Water is an endogenous skin constituent with minimal irritants or toxic exposure effects that are quickly reversible. Skin hydration can be increased easily with an occlusive vehicle or more elegantly with vehicles containing specific NMFs or polymer patch delivery systems [61].
High SC water content is essential in sustaining its flexibility, and its water holding capacity correlates directly with its protein and lipid domains and water-soluble substances [18].
Lipid content and water holding capacity of powdered human stratum corneuma
Stratum corneum source | Lipid content (% dry wt) | Water uptake (μg/mg dry powdered SC) | |||
|---|---|---|---|---|---|
Normal SC | Delipidized SC | ||||
Lipid | Protein | Total | |||
1 | 2.38 | 495.85 | 26.44 | 452.40 | 478.84 |
2 | 2.21 | 452.49 | 39.26 | 364.96 | 404.22 |
3 | 2.39 | 585.62 | 23.09 | 498.40 | 521.49 |
4 | 2.69 | 554.27 | 40.05 | 492.31 | 532.36 |
5 | 2.08 | 490.04 | 49.86 | 363.30 | 413.16 |
6 | 2.01 | 381.61 | 14.82 | 324.18 | 339.00 |
Mean | 2.29 | 493.31 | 32.26 | 415.92 | 448.18 |
SD | 0.25 | 72.66 | 12.97 | 74.50 | 75.47 |
Powdered SC not in an intact sheet exhibits lower water retention capacity. Powdering ruptures the corneocyte walls, allowing water to extract the hydrophilic NMFs, a process normally requiring a solvent. The results obtained suggest the protein domain of the powdered SC plays an important role in water absorption. Furthermore, depletion of the powdered SC lipid content did not significantly affect water retention (p > 0.05, [17]).
Additionally, if powdered SC is pretreated with water and ethanol or the dosing vehicle contains an ethanol concentration of 40% (v/v) or higher, the water retention capacity of the powdered SC can be reduced significantly (p < 0.05) when compared to untreated powdered SC or when vehicles with lower concentrations of ethanol are used (<40%, [17]). The data suggest that NMF is removed by the ethanol solution during processing.
Regional Variation in Stratum Corneum
Percutaneous absorption in both humans and animals varies depending on the anatomical location to which the chemical is applied. This implies a regional deviation of the barrier structure. Because the effect of any chemical applied to the skin is limited by the amount absorbed, estimation of the human health hazard effects from the environmental contaminant exposure illustrates the need to understand this variation. For example, after skin exposure to pesticide residue, estimation of prognostic factors and treatment doses requires the pesticide mass absorbed, and accuracy of this measure is crucial.
We review regional variation in skin absorption and its application to assess human risk. Risk assessment is achieved by determining skin absorption and multiplying it by the skin area involved. Accuracy improves by factoring in clothing and anatomical regions [50].
Feldmann and Maibach [11] explored the potential for regional variation in percutaneous absorption of hydrocortisone and/or parathion in men in vivo. The anatomical site absorbing the highest proportion of steroid was the scrotum, providing insight to the causation of scrotal cancer in chimney sweeps. Absorption was lowest for the sole of the foot and higher for the head and face.
Penetration indices for five anatomical sites assessed using hydrocortisone skin penetration data and pesticides (malathion and parathion) absorption resultsa
Site | Penetration index based on | |
|---|---|---|
Hydrocortisone data | Pesticide data | |
Genitals | 40 | 12 |
Arms | 1 | 1 |
Legs | 0.5 | 1 |
Trunk | 2.5 | 3 |
Head | 5 | 4 |
To examine how anatomical site affects chemical penetration and the amount remaining in the SC, Rougier et al. [40] applied benzoic acid (BA) to humans and performed tape stripping after 30 minutes. The total BA penetration varied by anatomical site with the forehead being three times more permeable than the back. Using the BA levels in the SC post 30-minute application and then its penetration over 4 days, additional correlations to anatomical sites were made (r = 0.97, p = 0.001). The forearm is commonly believed to be the best location to test for immediate contact irritation; however, Wertz et al. [49] showed the areas of greatest to least irritant response were the neck, the perioral region, the forehead, and the volar.
The face exhibits its own anatomical variation, and Marrakchi and Maibach [25] used noninvasive skin bioengineering technology to establish a map showing the six biophysical parameters : skin blood flow, transepidermal water loss (TEWL), SC hydration (capacitance), temperature, pH, and sebum content of the skin surface. Testing multiple locations on both young and old human volunteers, both age groups had the greatest skin blood flow on the nose, the chin was the most alkaline area, the neck had the highest capacitance value, and the perioral and nasolabial areas exhibited the highest TEWL. The highest skin temperature was on the neck in the young and the nasolabial area in the old. These values provide a framework for disease and intervention-related considerations.
Regional Stratum Corneum Variation: Application to Human Risk Assessment
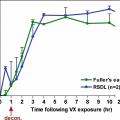
Chemical warfare agents (CWAs) are easily produced and of significant threat to military forces and the public. Most well-known CWAs are organophosphorus compounds, a number of commonly used pesticides, including parathion. Investigating the in vitro percutaneous absorption of parathion through dry and sweated uniformed and naked human skin, the percentage absorbed through naked skin (1.78 ± 0.41) was greater than that through dry (0.29 ± 0.17; p = 0.000) and sweated uniformed skin (0.65 ± 0.16; p = 0.000). A difference was also noted between sweated and dry uniformed skin (p = 0.007). Applying the uniformed skin absorption to the skin permeability of the CWA VX for the head, neck, arms, and hands shows a 50% mortality within the first hour of exposure for a sweated soldier wearing a uniform. By 8 hours post-trunk exposure in either a dry or sweated uniform, mortality is predicted. 96 hours after, any uniformed region is exposed individually would be lethal [56].
Permethrin bioavailability and body burden for uniformed soldiera
Human part | Body surface area (cm2) | Permethrin doseb (μg/cm2) | Percutaneous absorptionc | Body region indexd | Uniforme | Total (μg) |
|---|---|---|---|---|---|---|
Head | 1180 | 4 | 0.087 | X 4 | 1 | 1642.56 |
Neck | 420 | 4 | 0.087 | X 4 | 1 | 584.64 |
Trunk | 5690 | 125 | 0.087 | X 3 | 0.29 | 53834.51 |
Arms | 2280 | 125 | 0.087 | X 1 | 0.29 | 7190.55 |
Hands | 840 | 4 | 0.087 | X 1 | 1 | 292.32 |
Genitals | 180 | 125 | 0.087 | X 12 | 0.29 | 6812.10 |
Legs | 5050 | 125 | 0.087 | X 1 | 0.29 | 15926.44 |
Feet | 1120 | 4 | 0.087 | X 1 | 1 | 389.76 |
Total (μg) | 86672.88 | |||||
Total (mg) | 86.67 | |||||
Total body burdenf | 1.24 mg/kg |
In conclusion, review of current data shows important trends; however, regional variation of skin absorption has not been fully explored. Absorption disparities in all areas of animal skin have not been completely uncovered, but more importantly, many areas of human skin remain unstudied. The absorption differences of fingers, nails, eyelids, perirectal regions, and upper versus lower extremities need to be deciphered. While current data is sufficient to estimate human risk of percutaneous penetration by exposure region, evidence of absorption from clothing must also be explored for real-life applications. With more comprehensive absorption maps for a broader range of chemical moieties, refining our knowledge of skin penetration within dermatopharmacology and dermatotoxicology would be possible.
Rougier Method: A Quick Stripping Method to Predict Systemic Absorption Following Short-Time Dermal Exposure
Stratum corneum (SC) is the main barrier against dehydration and xenobiotics. A relatively noninvasive technique, adhesive tape stripping, can partially or entirely remove this micrometer-thin skin layer for a more thorough examination. Initially employed by Pinkus [31], this methodology has become common practice to remove SC layers, to reveal susceptible skin for irritant or allergen application, to predictably disrupt the water barrier for efficacy evaluation of barrier restoring compounds, or to obtain cells for mycological culture or to investigate SC quality [2]. Additionally, bioengineering methods including TEWL measurement can be employed post tape stripping to measure changes in water barrier properties [48].
Tape stripping may also assess the uptake of topically administered compounds into the SC. Rougier et al. [40–43] used tape stripping 30 minutes post chemical application to quantify the amount present in the SC. The reservoir capacity of the SC was determined at different anatomical regions and correlated with systemic excretions to predict the total molecular penetration over 4 days in both humans and rats in vivo.
Rougier Method to Determine Regional Variation of Chemicals in Human Stratum Corneum Post Exposure
The rate of molecule absorption depends on the application conditions, partially explaining the wide absorption variations reported for the same substances. The factors significantly affecting the absorption rate of a compound include the animal species, vehicle, dose applied, and length of contact. Contradicting explanations for regional permeability differences are often stated in literature reviews.
Percutaneous absorption of benzoic acid and anatomical sitea
Anatomical site | Urinary extraction 0–24 hour (1) | Total penetration in 4 days (2) | Amounts in stratum corneum 30 minutes after application | Predicted penetrations in 4 days |
|---|---|---|---|---|
(nmol/cm2) | (nmol/cm2) | (nmol/cm2) | (nmol/cm2) | |
Back | 6.41 ± 0.99 | 8.55 ± 1.32 | 6.19 ± 1.27 | 10.80 ± 2.32 |
Arm | 7.06 ± 0.77 | 9.41 ± 1.02 | 5.92 ± 0.62 | 10.31 ± 1.14 |
Chest | 8.78 ± 0.98 | 11.70 ± 1.31 | 8.07 ± 1.19 | 14.25 ± 2.18 |
Thigh | 9.38 ± 1.07 | 12.50 ± 1.43 | 6.80 ± 0.72 | 11.92 ± 1.31 |
Abdomen | 11.44 ± 1.44 | 15.26 ± 1.93 | 8.20 ± 1.24 | 14.48 ± 2.18 |
Forehead | 20.74 ± 2.71 | 27.65 ± 3.61 | 11.91 ± 2.28 | 21.24 ± 4.16 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree