Since the first implant-based breast surgeries were performed nearly 70 years ago, breast augmentation it has changed drastically. As understanding of breast augmentation has advanced, so too have the technologies and techniques used to improve results and minimize the risk of complications in breast implant surgery. This article reviews some of the novel techniques and technologies used today in breast augmentation surgery. How these tools and techniques will withstand the test of time remains to be seen, but they no doubt will add to the fascinating and ever-evolving history of breast augmentation.
Key points
- •
With the transition back to an era of smooth breast implants, subfascial breast augmentation may represent a middle ground in breast augmentation surgery, addressing the concerns of both the subglandular (eg, implant visibility and capsular contracture) and subpectoral (eg, animation deformity and double-bubble deformity) techniques.
- •
Antimicrobial implant pocket irrigation solutions containing povidone-iodine provide superior bactericidal activity against organisms commonly implicated in periprosthetic breast implant infection and capsular contracture.
- •
Radiofrequency identification (RFID) technology allows for the identification of in situ breast implants without the need for implant information cards.
- •
Poly-4-hydroxybutyrate (P4HB) scaffold is a monofilament, long-term resorbable implant that is useful for soft tissue support in breast surgery.
Background
It may be difficult to believe, but breasts have been augmented for more than 120 years. Long before the discovery of penicillin (1928) or the development of insulin (1922), people have been using alloplastic materials in an effort to enhance breast shape and size. Since the first reports of paraffin wax injections to the breast dating back to 1899, a veritable assortment of materials has been injected into the breast parenchyma, with varying degrees of success (or, more accurately, failure). The modern era of implant-based breast augmentation has a similarly fascinating history, dating back to the 1950s with the insertion of solid polyurethane, polytetrafluoroethylene (Teflon), and expanded polyvinyl alcohol formaldehyde (Ivalon) sponge devices for aesthetic breast enhancement. , Since the development of the first silicone gel (1963) and saline (1965) breast prostheses, there have been several generations of breast implants that have led to the current breast implants in use today. ,
Even after all these years of trial and error combined with innovation and scientific development, breast augmentation remains one of the most hotly debated and widely discussed areas in aesthetic surgery. Although this is in large part due to the fact that breast augmentation remains the most commonly performed aesthetic surgical procedure, it also speaks to the desire of plastic surgeons to improve results and minimize the risk of complications for patients.
This article reviews some of the novel techniques and technologies that are used today in breast augmentation surgery. How these tools and techniques will withstand the test of time remains to be seen, but they no doubt will add to the fascinating and ever-evolving history of aesthetic breast augmentation.
Subfascial breast augmentation
The implant pocket remains one of the most debated topics in aesthetic breast augmentation. When alloplastic breast augmentation first was performed, implants typically were placed in the subglandular plane. Due to concerns surrounding implant palpability, visibility, and rippling in the early years of subglandular breast augmentation, however, subpectoral and dual-plane techniques were developed to provide a more natural appearance to the upper pole of the augmented breast. , In 2002, Tebbetts classically described the upper pole pinch thickness test, advocating for implant placement in a subpectoral or dual plane for patients with a soft tissue pinch thickness of less than 2 cm. The concept of soft tissue coverage, combined with results from early meta-analyses and systematic reviews showing an increased risk of capsular contracture with smooth implants placed in the subglandular plane, led to a shift in practice whereby a majority of plastic surgeons were placing breast implants at least partially under the pectoralis major muscle.
Subpectoral and dual-plane breast augmentation, however, are not without their own inherent issues. One problem commonly seen after subpectoral and dual-plane augmentation mammaplasty is animation deformity, an issue found bothersome in a majority of patients. Furthermore, the pectoralis major muscle serves as one of the primary stabilizers of the shoulder joint, contributing to shoulder adduction, internal rotation, and flexion. Placement of a breast implant in the subpectoral plane has been shown to result in a significant decrease in pectoralis major muscle volume on magnetic resonance imaging up to 1 year postoperatively, , which can lead to alterations in shoulder stability and limit shoulder adduction strength. , ,
Recent developments in understanding of complications and risks associated specifically with textured implants, including breast implant–associated (BIA)-anaplastic large cell lymphoma (ALCL), have complicated the implant pocket discussion further. , With plastic surgeons forced to reconsider the role of textured implants in breast augmentation, the pervasive and dogmatic belief that subglandular augmentation is associated with a higher risk of capsular contracture when using smooth implants has returned to the forefront. Despite advancements in breast implant technology that have broadened the applications for subglandular implant placement, alongside the fact that many of the primary studies these meta-analyses and systematic reviews were based on had been performed prior to the introduction of the improved surgical techniques that now are widely accepted for reducing capsular contracture risk (eg, antibiotic irrigation, minimal-touch implant insertion, prospective hemostasis, and nipple shields), and a recent study demonstrating that smooth surface implants placed in the subglandular plane were not at a significantly increased risk of capsular contracture compared with textured surface implants, some surgeons are not yet prepared to place smooth surface implants in a subglandular plane. Fortunately, the solution to this issue may be subfascial breast augmentation.
First described in 2000 by Graf and colleagues, subfascial breast augmentation involves placement of a breast implant under the superficial layer of the pectoral fascia but above the pectoralis major muscle itself ( Fig. 1 ). Early proponents of this technique recognized that implant placement in the subfascial plane addressed the issues of animation deformity and inferolateral implant displacement associated with subpectoral implant placement while simultaneously providing additional soft tissue coverage to address the issues of implant visibility and palpability in the superior pole associated with subglandular implant placement. Some investigators also have suggested that subfascial implant placement results in improved long-term breast aesthetics by preserving attachments between the breast parenchyma and the chest wall, in addition to the pectoral fascia itself acting as an internal support for the implant. A recent study by Junior and colleagues, however, that randomized 20 patients to undergo either subfascial or subglandular breast augmentation, found no significant difference on clinical assessment of breast aesthetics or MRI assessment of capsule thickness or implant projection between the 2 groups.
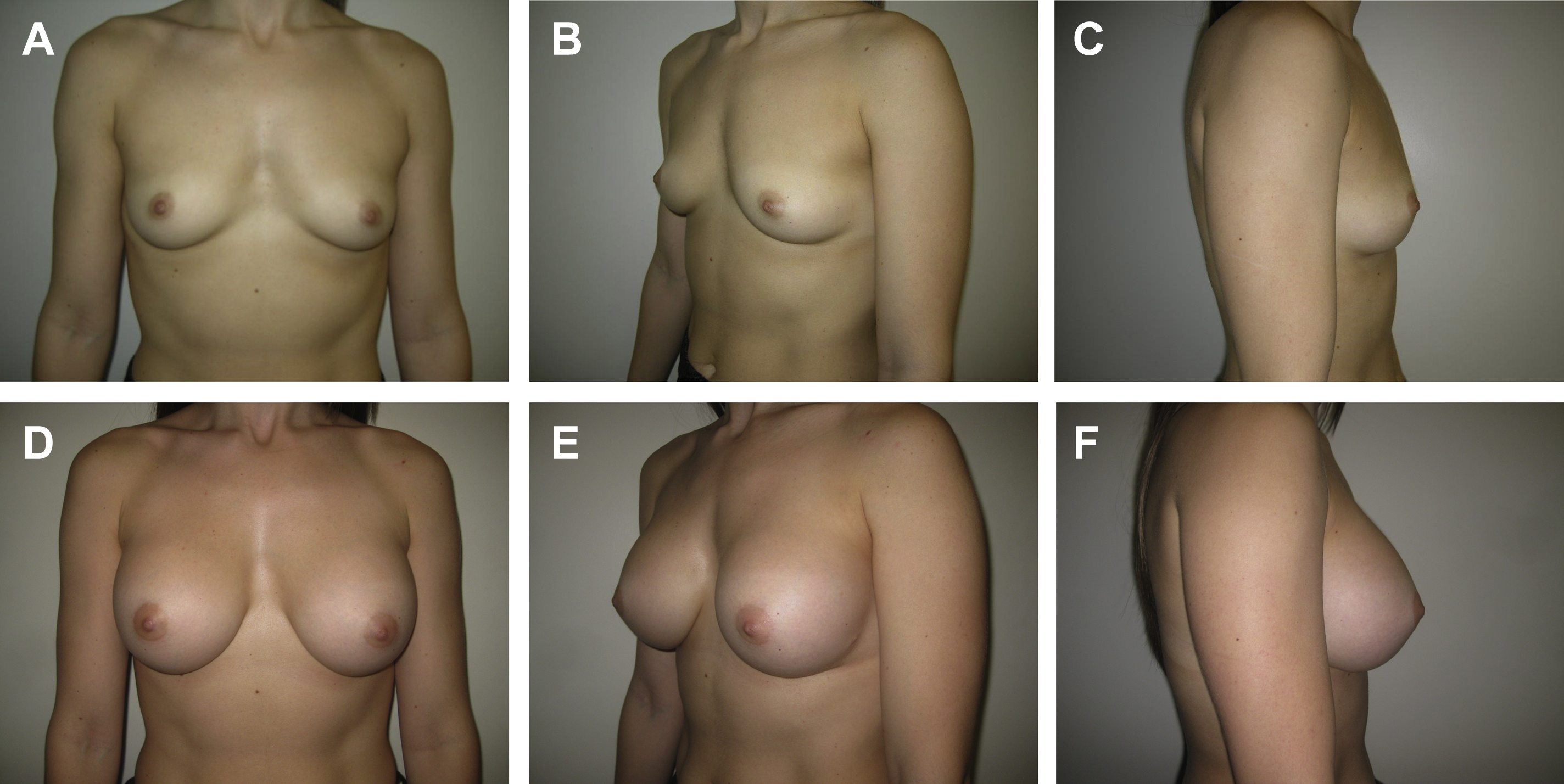
Some investigators also have suggested that subfascial implant placement is associated with a lower incidence of capsular contracture than subglandular augmentation. , , , , Although a systematic review of implant pocket selection by Shen and colleagues suggested that subfascial augmentation may be associated with a lower risk of capsular contracture, hematoma, and seroma than subglandular augmentation, there was insufficient statistical strength to support these findings. Given the current evidence regarding the role of bacterial contamination, subclinical infection, and biofilm as potentiators of capsular contracture, , the concept of placing the implant under the pectoral fascia to provide an additional barrier layer between the implant and the breast parenchyma does conceptually make sense.
Although further study of subfascial breast augmentation is required to better determine the risk and benefit profile in comparison with subglandular and subpectoral implant placement, this technique may represent a suitable middle ground for implant pocket selection with the transition back to an era of smooth breast implants.
Antimicrobial implant pocket irrigation in breast surgery
Consensus among plastic surgeons is a difficult thing to achieve, and this is no different when it comes to the discussion of antimicrobial implant pocket irrigation solutions in breast augmentation. Starting in the early 1980s, plastic surgeons began to recognize the association between bacterial contamination and the development of capsular contracture. In 1986, Burkhardt and colleagues published the first randomized prospective study demonstrating that the use of periprosthetic antimicrobial solutions decreased the incidence of capsular contracture by approximately 50%. Even in these early days, however, an inherent lack of consensus existed, as Burkhardt and colleagues pooled the results of a cephalosporin irrigation, cephalosporin with Betadine irrigation (Avrio Health, Stamford, Connecticut), and Betadine irrigation versus controls to reach their conclusions.
In 2000, almost immediately after Adams and colleagues published the results of their in vitro study of the effectiveness of 10% Betadine combined with gentamycin and cefazolin against common bacteria implicated in capsular contracture, the Food and Drug Administration (FDA) banned the use of povidone-iodine in breast implant surgery due to a reported increase in the risk of implant shell failure. , In this new post-Betadine era, Adams and colleagues first reported on use of the triple-antibiotic solution, a combination of cefazolin with bacitracin and gentamycin, as an alternative to povidone-iodine containing irrigation solutions. Since that time, although several systematic reviews and meta-analyses have reported on the benefits of antimicrobial implant pocket irrigation in the reduction of capsular contracture rates, these analyses frequently comment on the need for more high-powered, randomized studies to support this conclusion. ,
So, where does the use of antimicrobial implant pocket irrigation solutions in breast augmentation do we stand in 2020? After the FDA reversed its position in 2017 for one of the implant manufacturing companies, thereby allowing for the use of povidone-iodine solutions in breast implant surgery, , there has been an increased effort to study, optimize, and standardize antimicrobial implant pocket irrigation. The lack of standardization in breast pocket irrigation was made abundantly clear by a 2019 survey of 407 plastic surgeons that reported more than 35 unique antimicrobial implant pocket irrigation mixtures in use by plastic surgeons across the United States, including 4.3% of plastic surgeons reporting that they do not use any antimicrobial implant pocket irrigation solution in breast augmentation. Considering the current understanding of the role that bacterial contamination and biofilms plays in capsular contracture, , and the association that has been made between gram-negative Ralstonia species and BIA-ALCL, this wide variability in practice regarding antimicrobial implant pocket irrigation solutions is particularly concerning.
So, which antimicrobial implant pocket irrigation mixtures is the best? In 2019, Culbertson and colleagues published an in vitro analysis of several antimicrobial implant pocket irrigation solutions against organisms commonly implicated in periprosthetic breast implant infections and capsular contracture, including Staphylococcus epidermidis , Escherichia coli , Pseudomonas aeruginosa , Ralstonia pickettii , and Mycobacterium fortuitum . The investigators tested dilutions of Betadine-containing and non–Betadine-containing antimicrobial solutions, including 0.025% hypochlorous acid, chlorhexidine-containing solutions, and silver-based irrigation solutions, at varying exposure time points. The results of this study demonstrated that Betadine was effectively bactericidal against all tested pathogens, even at dilutions as low as 6.25%. Betadine–triple-antibiotic solutions were similarly effective. Non-Betadine–triple antibiotic solutions were not reliably bactericidal against all pathogens tested, in particular Mycobacterium fortuitum , even with up to 18 hours of exposure. Chlorhexidine gluconate in isopropyl alcohol and undiluted hypochlorous acid also were found to be bactericidal against all organisms at all time points, although long-term safety data surrounding use of these products around breast implants are limited. Although this study did not specifically investigate antimicrobial resistant organisms or activity against planktonic bacteria within a biofilm, and in vivo correlation is lacking, the investigators present a thoughtful and scientifically based rationale for the use of specific antimicrobial implant pocket irrigation solutions. Any plastic surgeon performing breast augmentation should consider this a must-read publication.
A consensus may not have been reached among plastic surgeons regarding antimicrobial implant pocket irrigation solutions, but it is time to stop using tradition, preference, and conjecture to guide decisions. Instead, plastic surgeons should turn toward the available science and evidence, and that which are yet to come, to propel them forward into the next era of breast augmentation surgery.
Technology in breast implant identification
With recent concerns surrounding textured breast implants, there has been increased discussion surrounding the topic of breast implant identification. Traditionally, patients undergoing breast augmentation have been given a card containing information about their breast implants (eg, shape, size, projection, and texture) as well as the implant specific serial number. With more patients than ever before returning to discuss concerns regarding their breast implants, however, it has been learned over the course of time many of these implant cards are being forgotten, misplaced, or lost. This is problematic particularly when patients are not returning to see their primary surgeon, because without operative records or implant-specific information, efforts to educate patients on their implants and plan for revision surgery are made much more difficult. These issues are being complicated further by the fact that, as has been learned, obtaining individual implant-specific information from the implant manufacturers is difficult if not impossible.
Fortunately, novel technology in breast implants may provide a solution to this problem. Radiofrequency identification (RFID) technology utilizes electromagnetic fields to communicate information stored in an RFID transponder to a nearby RFID reader that is on set to the same radiofrequency signal. When the RFID reader is placed near the RFID transponder, the reader interrogates the transponder to emit its signal, typically an electronic serial number, a process known as passive tag RFID. Although this technology has been used in veterinary medicine for years, only recently has it crossed over into implantable medical devices. Motiva Implants (Establishment Labs Holding, New York, New York) now are being marketed and sold with an RFID transponder embedded within the silicone gel breast implant itself, thus allowing for rapid identification of any implant based on the electronic serial number encoded within the RFID tag. Although this technology would eliminate the need for patients to retain implant cards, it would require implant manufacturers to maintain an accessible central database into which all implant serial numbers and details were entered. One potential limitation of this technology surrounds the issue of radiographic artifact arising from the RFID transponder on magnetic resonance imaging, particularly in regard to breast cancer screening and diagnosis. Preliminary studies seem to suggest that this artifact is minimal, however, and can be overcome easily with the use of a second imaging modality (ie, ultrasound). ,
Nevertheless, the concept of using of RFID or other similar technology for the identification and tracking of breast implants presents an interesting frontier in breast implant surgery.
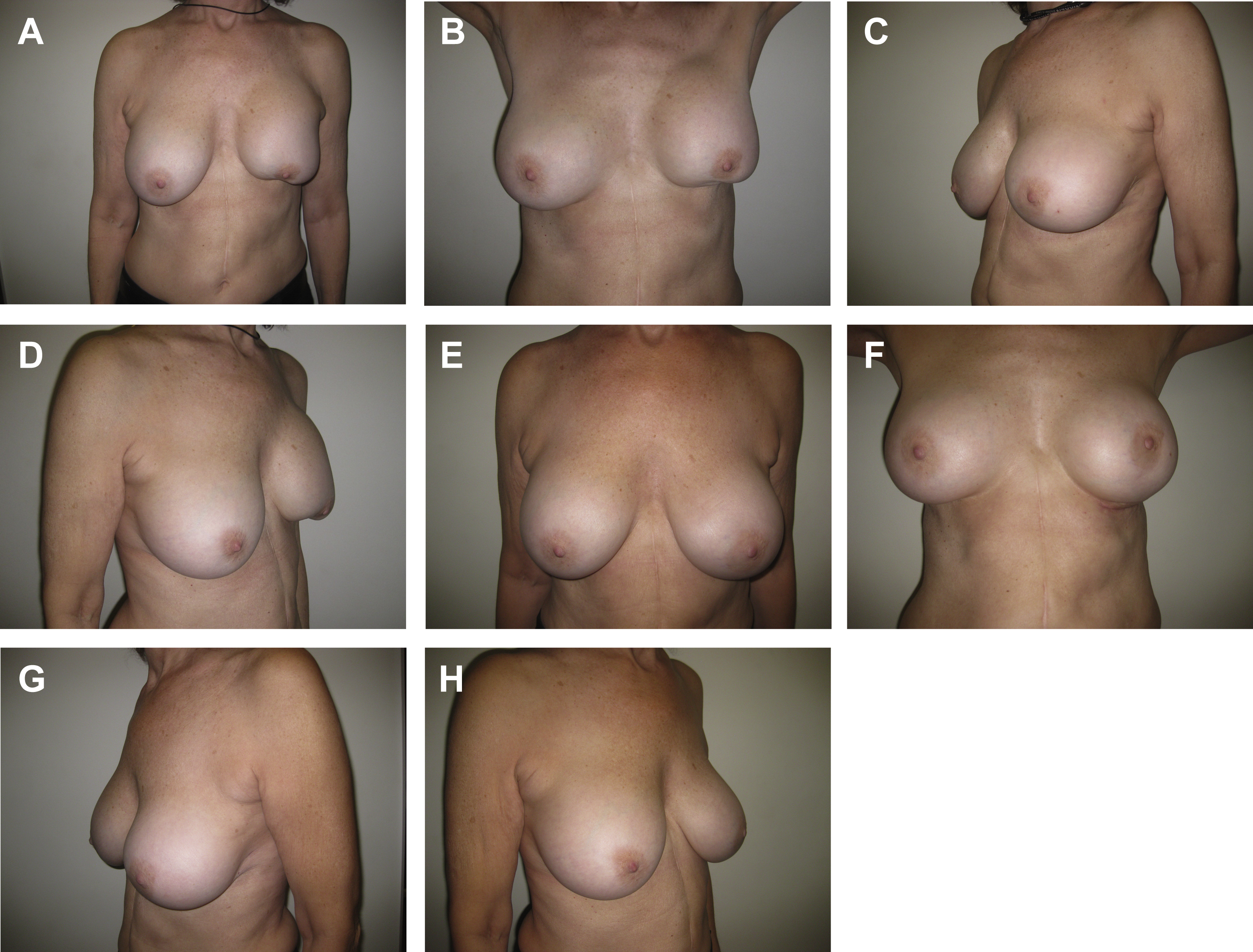
Soft tissue support in breast augmentation
A variety of biologic and synthetic materials are used for soft tissue support during breast surgery. Soft tissue support may be required to shape breast tissue or control breast implant position ( Fig. 2 ). Over the past decade, experience with several different types of acellular dermal matrices (ADMs) has been described extensively. Recently, early experience with poly-4-hydroxybutyrate (P4HB) scaffolds (Galatea Surgical, Lexington, Massachusetts) in breast surgery has been promising. GalaFLEX is a macroporous, monofilament, long-term resorbable implant. It is a scaffold that allows vascularized tissue ingrowth and becomes completely replaced with type I and type III collagen by 18 months to 24 months postoperatively. Adams and Moses described the use of P4HB for soft tissue reinforcement in the inferior pole during central mound mastopexy in 11 patients. Some breast settlement occurred between 1 month and 3 months postoperatively but there was no statistically significant change between 3 months and 12 months postoperatively from the sternal notch to the most inferior point on the breast. A multicenter observational study of 62 patients who underwent either mastopexy or reduction mammaplasty reported 89.7% of patients had successful ptosis correction and maintenance at 1 year postoperative. Nair and Mills reported positive experience in 5 patients treated with placement of GalaFLEX in direct contact with the breast implant for treatment of capsular contracture and to provide soft tissue support. Levy and colleagues recently compared 192 cases using ADMs and 112 cases using P4HB mesh in immediate expander-based breast reconstruction. Their findings suggest P4HB mesh is a safe and less costly alternative to ADMs in expander-based breast reconstruction, with trends toward decreased rates of infection, seroma, and need for device removal with P4HB. Although early reports for P4HB scaffolds in breast surgery have been positive, more studies are necessary to evaluate outcomes in breast augmentation and breast augmentation-mastopexy as well as revision surgery.