Introduction
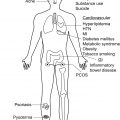
In many epidemiologic studies, hidradenitis suppurativa (HS) has been found to disproportionately affect women of childbearing age. Nearly three-quarters of new diagnoses are in women, with the highest incidence in female patients aged 30 to 39. Furthermore, the reported average annual incidence in the United States is 12.1 per 100,000 for women, which is more than twice that of men (5.1 per 100,000). Women may also have an earlier onset of disease coincident with the earlier onset of puberty in girls. In women, HS can be found in the inframammary folds, breasts, and vulva, in addition to the axilla, inguinal, and buttock regions, as well as other anatomic sites. A hormonal influence on HS in women is strongly suggested given changes observed in HS disease status peri-menstruation and during pregnancy. Female sex hormones present in high levels, such as estrogen and progesterone, have been thought to play a role in the sexual dichotomy of HS prevalence. Women face unique challenges in managing their HS related to menses, pregnancy, lactation, and menopause. Special consideration should be taken to provide comprehensive care to women with HS and improve their quality of life.
Menses
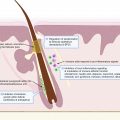
The range of peri-menstrual HS flares has been reported to be between 44% and 63%. The ovarian cycle enters the luteal phase after ovulation, where the remaining granulosa cells undergo luteinization and combine with theca-lutein cells to form the corpus luteum. The corpus luteum functions as an endocrine organ and primarily secretes progesterone to prepare the endometrium for possible embryo implantation. Typically, eight or nine days after ovulation, during the mid-luteal phase, serum levels of progesterone and estradiol peak. If human chorionic gonadotropin (hCG) is not produced as a result of pregnancy, the corpus luteum will undergo luteolysis, leading to a decline in progesterone production and resulting in the menstrual period. Estradiol also decreases during the luteal phase of the ovarian cycle, though to a lesser degree. Premenstrual HS flares are hypothesized to be caused by the change in steroid sex hormone levels immediately preceding the onset of menstruation ( Fig. 31.1 ), which leads to immunologic changes; however, the definitive relationship between progesterone, estrogen, and HS disease status has yet to be established.
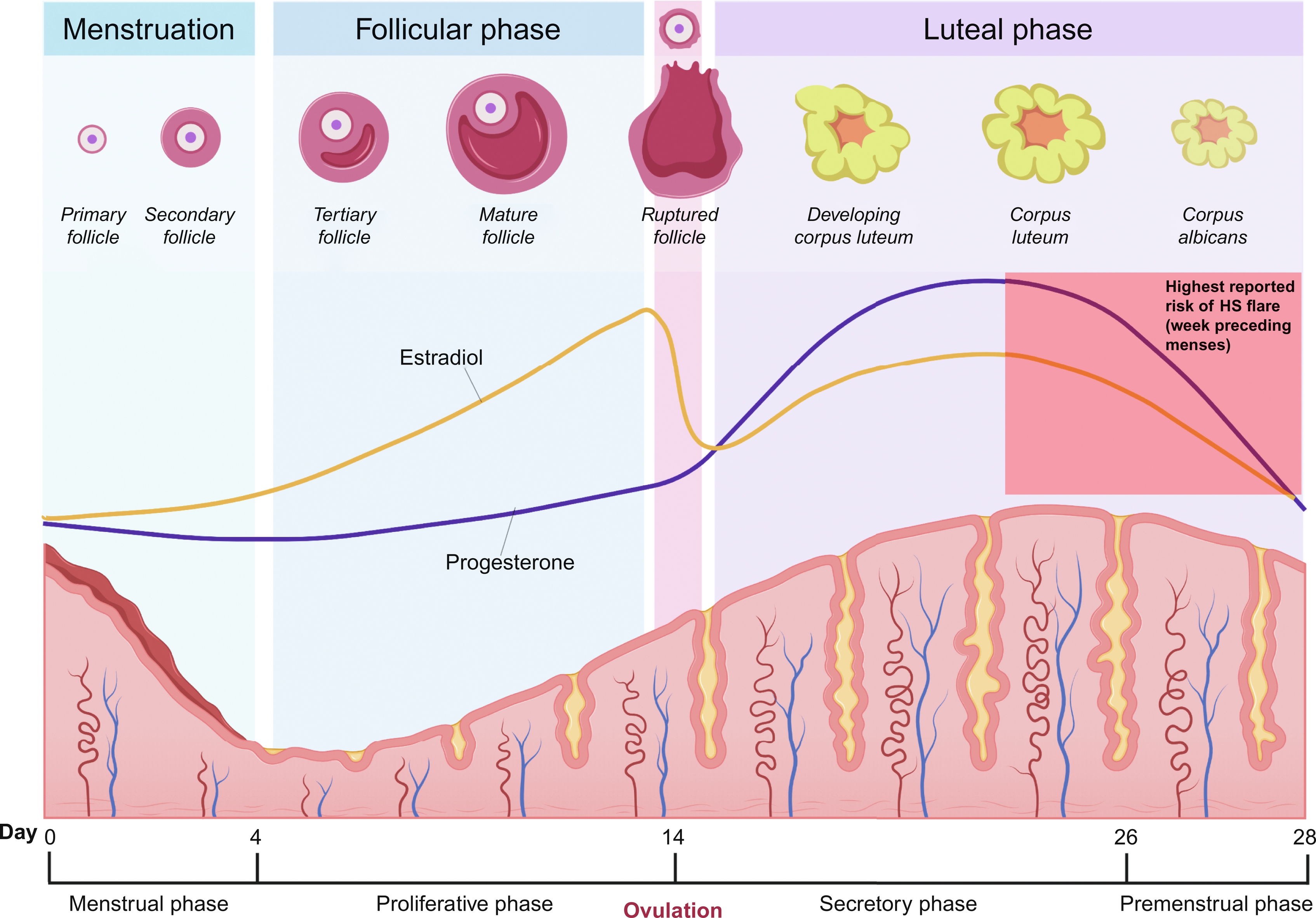
Hormonally Mediated Immune Alterations That Impact Hidradenitis Suppurativa
Immune dysregulation has been implicated in HS pathogenesis, with studies reporting elevation in several pro-inflammatory (and some anti-inflammatory) cytokines, including TNF-α, interleukin (IL)-1β, IL-10, IL-17, IL-12, IL-23, IL-22, and IL-20 in lesional HS skin compared to healthy control skin.
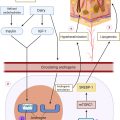
Estrogen and progesterone have been shown to differentially modulate immune tolerance through varying physiological mechanisms ( Fig. 31.2 ). Estrogen stimulates the production of interferon alpha (IFN-α) by plasmacytoid dendritic cells. In mice, this has been shown to occur via estrogen receptor alpha (ER-α)-mediated upregulation of the transcription of type 1 interferon (IFN) producing genes. Type 1 IFNs (including IFN-α, β, ɛ, τ, and δ) then further increase estrogen signaling within immune cells by inducing expression of more ER-α, resulting in a “feed-forward” loop. Estrogen can also enhance the release of inflammatory cytokines including IL-1 and TNF-α from human monocytes and macrophages and pro- and anti-inflammatory cytokine IL-6. In addition, in vivo mouse studies suggest that estrogen induces ER-α mediated differentiation of naïve CD4 + T cells into T helper (Th) 1 cells, which secrete pro-inflammatory IFN-γ.
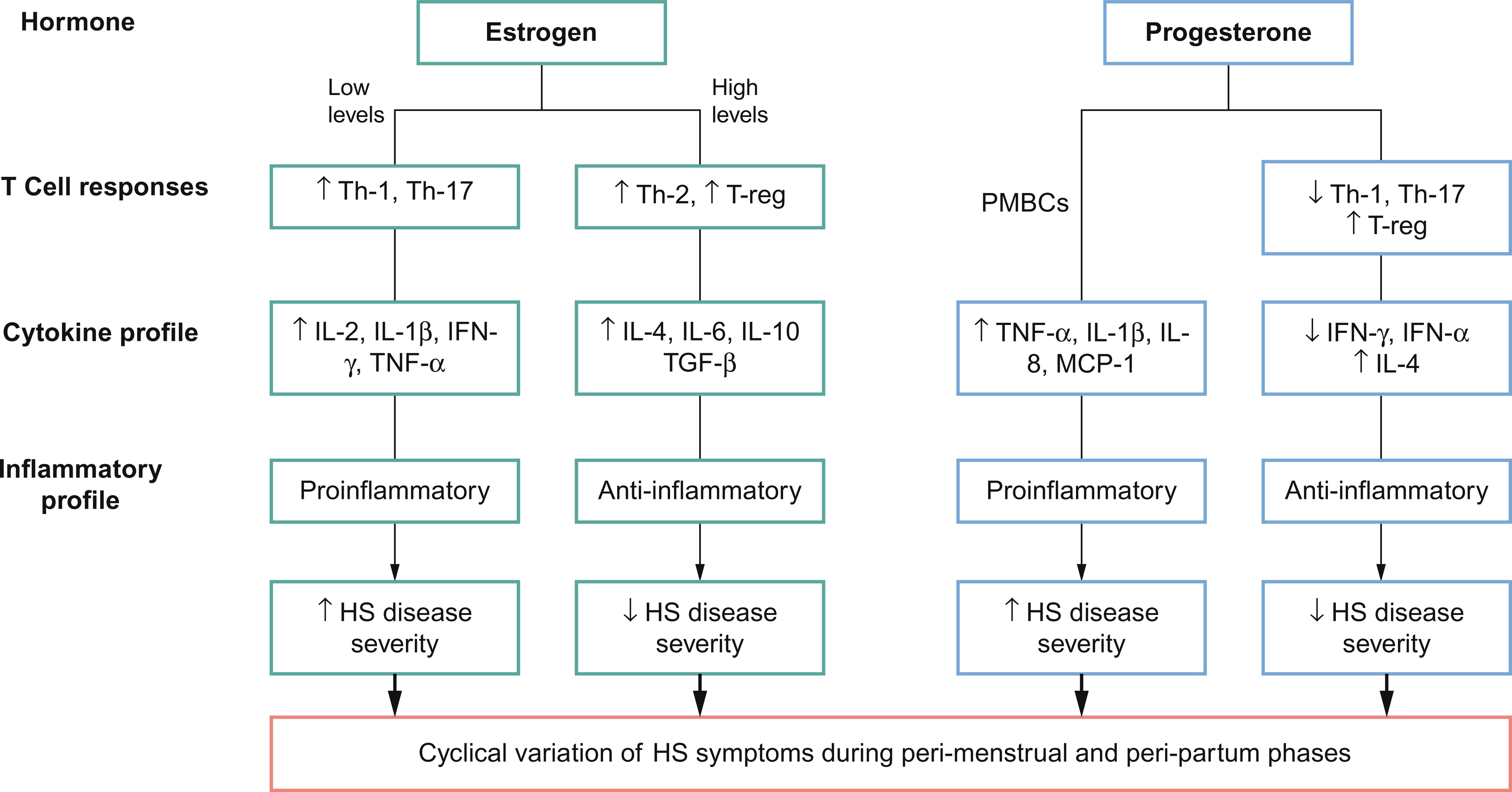
Though these results suggest that estrogen plays a predominantly pro-inflammatory role, discordant mouse studies have demonstrated inhibition of CD4 + T cell differentiation into Th1 and Th17 cells via ER-α signaling in T lymphocytes. Other in vivo mice studies have suggested that the effects of estrogen on cell-mediated immunity vary according to dose, with low doses of estrogen promoting enhanced pro-inflammatory Th1 responses and high doses leading to anti-inflammatory Th2 responses. Estrogens have also been shown to have a biphasic effect on TNF-α in human T cell clones, with enhancement at low concentrations and inhibition at high concentrations.
The anti-inflammatory effects of progesterone have been well-documented. Progesterone has been found to suppress type 1 IFN production as well as production of IFN-γ from Th1 cells in mouse studies. Studies have shown that medroxyprogesterone acetate at doses sufficient for contraceptive efficacy can suppress IFN-α production by plasmacytoid dendritic cells in mice in vivo and human plasmacytoid dendritic cells in vitro. Moreover, progesterone suppresses proliferation of CD4 + T cells and Th17 cells, which are involved with chronic inflammation seen in HS. Evidence from mouse models also supports the role of progesterone in facilitating regulatory T cells (Treg) differentiation involved in preventing autoimmunity and mitigating the damaging inflammation from naïve CD4 + T cells via progesterone receptor signaling.
Few studies suggest that progesterone may also have pro-inflammatory effects. Yuan et al. (2008) studied the effects of progesterone on the spontaneous production of TNF-α, IL-1β, IL-8, and macrophage chemotactic protein by unstimulated peripheral blood mononuclear cells (PMBCs) from patients with chronic hepatitis C. They discovered that the levels of each cytokine increased significantly in PBMCs treated with progesterone.
Premenstrual HS exacerbation has been suggested to result from a reduction in anti-inflammatory mediators due to lower progesterone levels prior to menses. However, the timing of HS symptom onset needs further elucidation; it is unclear if HS symptoms are triggered by high levels of progesterone and estrogen in the mid-luteal phase, or by the declining levels of progesterone and estrogen right before menses. One study found that, compared to patients with premenstrual HS flares, women without premenstrual flare had lower progesterone levels and higher testosterone and androstenedione levels. Additional research is needed to fully elucidate the role of steroid sex hormone in HS immunology.
Medical Therapy
Oral Contraceptive Pills
Evidence is mixed regarding the use of oral contraceptive pills (OCPs) in the management of HS flares. Early cases have suggested a temporal relationship between HS onset and initiation of OCPs, with complete resolution occurring with either discontinuation of OCPs or switching to a formulation with a higher estrogen:progestogen ratio. Other studies have shown no differences in HS symptoms between OCP and non-OCP groups, or have shown substantial disease improvement with either ethinyloestradiol 50 μg/cyproterone acetate (CPA) 50 mg EKC or ethinyloestradiol 50 μg/norgestrel 500 μg. The literature also contains anecdotal reports of OCPs with high estrogen to progesterone ratio and low androgenic progestogens leading to HS disease amelioration. Current evidence does not allow for concrete recommendations on which specific OCP to use for the management of HS. However, if OCPs are being considered, choosing one with a less androgenic progestogen, such as norgestimate, desogestrel, gestodene, or drospirenone, may be beneficial. Though further investigation is needed, some studies suggest that progesterone-only pills, progesterone implants, or depot progesterone can cause HS exacerbation.
Spironolactone
Spironolactone, a potassium-sparing diuretic with anti-androgenic properties, has demonstrated varying efficacy in HS management. Studies have shown HS clinical improvement with spironolactone at 3 months follow-up. One study found no differences in improvement between patients who received less than 75 EKC mg of spironolactone daily and those who received over 100 mg daily EKC. The 2019 North American HS management guidelines recommend the use of hormonal therapies, such as spironolactone and OCPs, in female patients with mild-to-moderate HS or as adjunctive agents in severe disease, and suggest that those with perimenstrual flares may be most likely to benefit.
Wound Care During Menstruation
Mechanical friction has been suggested as a pathogenic mechanism in the development of HS, supported by cases of HS lesions arising in areas of amputated limbs with prostheses-related friction and the development of new HS lesions at sites exposed to excess friction. Women may potentially experience discomfort and exacerbation of vulvar or perineal lesions during menstruation due to mechanical friction imposed by sanitary napkins. Therefore, the use of tampons in lieu of menstrual pads can help reduce friction and moisture in the area. For women who prefer menstrual pads, choosing non-fragranced pads made from gentle materials such as cotton or bamboo may be beneficial. For painful or heavily draining wounds in the groin region, women may benefit from the use of a hydrofiber dressing with silver, calcium alginates, or foams. Appropriately fitting biker shorts can be worn to help hold dressings in place.
Fertility
Limited evidence from prospective studies suggests that approximately 12% to 18% of women in the U.S. are not pregnant within 12 months of attempting conception. There is a paucity of data in the literature regarding the impact of HS on female fertility. However, it is well known that HS is associated with several somatic and psychiatric comorbidities associated with fertility issues.
Somatic Diseases and Infertility
The pathophysiology of infertility in women often involves anatomical dysfunctions, including history of tubal infections, endometriosis, and various uterine pathologies such as adhesions, polyps, fibroids, or asymptomatic tumors. There is a higher prevalence of polycystic ovarian syndrome (PCOS) among HS patients than the general population (9.0% vs. 2.9%, respectively), which could contribute to increased infertility. Lifestyle and modifiable behavioral factors can also negatively impact fertility in female HS patients. The overall incidence rate of HS in tobacco smokers is nearly double the incidence rate among nonsmokers (0.20% vs. 0.11%, respectively); chronic smoking can cause rapid depletion of ovarian follicles, conception delay, and increased risk of spontaneous abortion. Obesity is also significantly associated with HS (pooled OR of 3.45 found in the systematic review); obese women have a threefold higher risk of infertility compared to non-obese women. This risk is potentially due to derangements in gonadotrophin secretion as a result of peripheral aromatization of androgens to estrogens and hyperandrogenemia from insulin resistance and hyperinsulinemia.
Psychologic Stress and Infertility
HS patients may suffer from stress related to their illness, financial hardship from disease burden, and psychiatric comorbidities. Persistent psychological stress can alter oocyte maturation and lead to infertility. Female HS patients struggling with infertility should be screened for comorbid conditions such as PCOS and psychiatric disorders if that has not already been done. Lifestyle modifications should be encouraged to achieve and maintain a healthy weight and join smoking cessation programs if applicable. Large population-based studies exploring infertility rates in HS patients compared to the general population are needed.
Pregnancy
The effects of pregnancy on HS disease course are mixed. Two European survey studies found that pregnancy is associated with HS symptom improvement in 20% and 30.2% of patients, worsening in 8% and 16.7%, with the majority reporting no change (72% and 53.1%). However, a recent retrospective chart review study of patients at an academic referral center in the U.S. found that the majority (62%) of HS patients reported HS disease worsening during pregnancy. There remains a lack of consensus in the literature regarding the impact of pregnancy on HS; thus, patients should be counseled to monitor their HS disease during pregnancy and contact their healthcare providers if symptoms are worsening.
Pregnancy-Related Protective Factors for Hidradenitis Suppurativa
Progesterone and estrogen levels rise throughout pregnancy, which has been implicated in the amelioration of HS symptoms reported by some pregnant women. Estrogens produced by the placenta are primarily derived from fetal dehydroepiandrosterone (DHEA) sulfate, which is then converted to androstenedione and testosterone through enzymatic pathways in steroid-producing tissues. The placenta then aromatizes these androgens to estrone and estradiol. 17α-hydroxysteroid dehydrogenase type II catalyzes the conversion of estradiol to estrone. A third type of estrogen, estriol, is formed as a metabolite from DHEA sulfate. Throughout pregnancy, serum estrone and estradiol concentrations increase to approximately 50-fold their maximal pre-pregnancy values, while estriol increases 1000-fold.
This excess estrogen affects inflammatory pathways in various ways. Human in vitro and mouse in vivo studies show that estrogens can affect T helper cells in a biphasic way, with low doses of estrogen enhancing Th1 responses, and high levels augmenting Th2 responses (see Fig. 31.2 ). Therefore, as estrogens rise during pregnancy, there is a shift in the Th cytokine profile from pro-inflammatory (IL-2, IFN-γ, TNF-α) to anti-inflammatory (IL-4, IL-6, IL-10, TGF-β). During pregnancy, estrogen also activates regulatory T cells (Treg), promoting their proliferation via the PI3K/AKT signaling pathway and augmenting their immune-suppressive capabilities. Improvement of other chronic inflammatory diseases during pregnancy, such as rheumatoid arthritis, has been correlated with elevated and sustained levels of Treg activity. Thus, the high levels of estrogen during pregnancy may confer beneficial effects on patients’ HS disease; however, the precise mechanisms for estrogen-induced immunomodulation during pregnancy and the impact on HS disease remain unclear.
Progesterone-mediated anti-inflammatory effects may also contribute to the amelioration of HS symptoms during pregnancy. Around the tenth week of pregnancy, the placenta takes over progesterone production and the corpus luteum involutes. The placental enzymes cleave maternal cholesterol sidechains to yield pregnenolone, which is later isomerized to progesterone. By the third trimester, 250 to 350 mg of progesterone are produced daily, with the majority entering the maternal circulation. As mentioned, progesterone promotes an anti-inflammatory cytokine milieu. Progesterone also induces the release of IL-4, an important cytokine for the suppression of inflammation (mediates Th2 cell function and regulates Treg activity), and together, both act to inhibit Th1 responses during pregnancy.
In addition to the immune-modulating activities of estrogen and progesterone during pregnancy, there are also immunological changes that are not directly hormonally influenced, which take place within the pregnant female body that may mitigate HS disease symptoms. For example, uterine natural killer cells (NK cells), monocytes, and Tregs within the decidua (the membrane lining the uterus during pregnancy) are key producers of IL-10, which inhibits excess inflammation. IL-10 production is increased during the first and second trimester, falls by the third trimester, and then increases again postpartum. Furthermore, IL-4 can be produced by immune cells of the placenta, maternal decidua, amniochorionic membranes, cytotrophoblasts, and maternal and fetal endothelial cells.
Cytokine receptor antagonists, such as IL1Ra, can also act to inhibit pro-inflammatory mechanisms by blocking receptor binding. A study by Østensen et al. found that levels of IL1Ra and soluble TNF receptor (TNFR) were elevated in healthy pregnant women and pregnant women with rheumatoid arthritis, when compared to non-pregnant controls, with a significant decrease in the post-partum phase. Thus, increased levels of IL-10, IL-4, IL1Ra, and TNFR seen in pregnancy could contribute to the amelioration of HS disease experienced by some patients during pregnancy.
Pregnancy-Related Harmful Factors for Hidradenitis Suppurativa
Pregnancy begins with an inflammatory reaction triggered by implantation of the embryo within the uterine wall. During this reaction, inflammatory cytokines, such as IL-1, IL-6, and leukemic inhibitory factors are upregulated. Various leukocytes migrate to the decidua and directly interact with the trophoblasts of the developing placenta, promoting their survival. This initial pro-inflammatory state could be a contributing factor in those patients with HS disease worsening with the onset of pregnancy.
Weight gain during pregnancy may increase friction and maceration at intertriginous sites, which may contribute to HS flares. Additionally, adipocytes may independently promote the secretion of pro-inflammatory cytokines, such as TNF-α. One cross-sectional study of 186 patients did not demonstrate a correlation between body mass index and severity of HS symptoms during pregnancy; however, the impact of increased weight gain during pregnancy on the severity of HS symptoms was not evaluated.
Medical Therapy During Pregnancy and Lactation
When deciding on a therapeutic agent for a pregnant HS patient, important considerations include: (1) expected efficacy of the intervention for HS and (2) safety profile of the intervention for both the mother and the fetus. The U.S. Food and Drug Administration (FDA) pregnancy categories have now been replaced by individual medication risk summaries ; however, these former pregnancy categories will still be included in this chapter for historical reference ( Table 31.1 ). A list of commonly used HS medications and their associated pregnancy and lactation safety information is found in Table 31.2 .
| Category | Risk |
|---|---|
| A | Well-controlled studies in pregnant women have failed to demonstrate fetal risk |
| B | Animal reproduction studies have failed to demonstrate fetal risk and no well-controlled studies exist for pregnant women; animal reproduction studies have demonstrated adverse effects, but well-controlled studies in pregnant women show no adverse fetal effects |
| C | Animal reproduction studies have shown adverse fetal effects; or no animal reproduction studies exist and no well-controlled studies in humans exist |
| D | Positive evidence of fetal risk; benefits may outweigh risks |
| X | Positive evidence of fetal risk; risks clearly outweigh possible benefits |
a FDA pregnancy categories have been replaced by medication specific risk summaries (effective June 30, 2015); however, they are included here for historical reference.
| Medication | Former FDA Pregnancy Category a | Safety Comments for Pregnancy | Safety Comments for Breastfeeding |
|---|---|---|---|
| Topical Antibiotics | |||
| Clindamycin | B | Minimal systemic absorption; no evidence of teratogenicity with systemic exposure during 2nd and 3rd trimester ; remote cases of pseudomembranous colitis with topical use | Not known if excreted in breast milk with topical application; considered compatible |
| Erythromycin | B | Low levels of systemic absorption; no evidence of teratogenicity or any other adverse effects in animal studies | Not known if excreted in human milk after topical application; considered compatible |
| Metronidazole | B | Negligible systemic absorption; no evidence of embryotoxicity or teratogenicity | May be present in low levels in breast milk; considered compatible |
| Systemic Antibiotics | |||
| Clindamycin | B | No evidence of congenital abnormalities when given in 2nd and 3rd trimester; lack of adequate studies during 1st trimester | Can pass into breast milk; has the potential to cause adverse effects on infant’s gastrointestinal flora ; considered compatible |
| Dapsone | C | No evidence of fetal abnormalities with administration during any trimester ; has been shown to cause neonatal hyperbilirubinemia and hemolysis; G6PD screening recommended due to risk of hemolytic anemia | Excreted in breast milk in substantial amounts; possible hemolysis in neonate ; caution advised with breastfeeding |
| Ertapenem | B | No adequate studies in pregnant women; no evidence of teratogenicity in animal studies; slight decrease in fetal weight and effects on vertebral ossification in mice | Present in human milk; limited data regarding effects on breastfed infant; considered compatible |
| Metronidazole | B | Crosses placental barrier and enters fetal circulation rapidly; no evidence of fetotoxicity in mice ; no well-controlled studies in pregnant women; evidence in observational studies suggest low risk | Secreted in breast milk in concentrations similar to plasma levels; discontinue 12–24 h before breastfeeding |
| Moxifloxacin | C | No human studies; animal studies show no teratogenicity but there may be other possible harmful effects to the fetus, such as decreased weight and delayed skeletal development ; safer alternatives are generally preferred | Unknown if excreted in human breast milk, but likely ; caution advised with breastfeeding |
| Rifampin | C | No adequate well-controlled studies in pregnant women; reported to cross the placental barrier and appear in cord blood; congenital malformations reported in animal studies ; postnatal hemorrhages in mothers and infants following exposure during last few weeks of pregnancy ; use only if benefits outweigh risks | Excreted in breast milk but no evidence of adverse effects in infants; considered compatible |
| Immunosuppressants | |||
| Corticosteroids | C | Animal studies have shown cleft palate, increased fetal lethality, and decreased body weights; observational studies in women report intrauterine growth retardation and fetal hypoadrenalism with high doses ; recommended to limit prolonged use to 7.5 mg/day (avoid doses at or above 20 mg daily) | Minimal excretion in breast milk; wait 4 h prior to breastfeeding |
| Cyclosporine | C | Embryo- and fetotoxic in animals at 2–5 times human dose; no well-controlled studies in pregnant women ; increased risk of premature birth and low birth weight shown in observational studies | Present in breast milk; not recommended during breastfeeding due to concern that drug may interfere with cellular metabolism in infant |
| Biologics | |||
| Adalimumab | B | Studies do not reliably establish an association with birth defects; increased active transfer across placenta in 3rd trimester and may affect the immune response of infant; in animal studies, no fetal harm or malformations was observed ; consider discontinuing at end of 2nd trimester | Data suggest systemic exposure to adalimumab in the breastfed infant is low because it is a large molecule and degraded in the gastrointestinal tract; no reports of adverse effects on the infant ; likely compatible |
| Anakinra | B | No evidence of fetal harm in animal studies; insufficient data in pregnant women to determine the risk of birth defects or maternal adverse events ; animal studies suggest low risk but may be best avoided until additional safety data is available | Limited data precludes risk assessment; case reports on use during lactation have not shown adverse effects on breastfed infants |
| Infliximab | B | Unknown if causes fetal harm; no evidence of maternal toxicity, embryotoxicity, or teratogenicity in mice; crosses the human placenta in late gestation and has been detected in infants up to 6 months after delivery ; congenital anomalies reported in observational studies though causation not established ; consider discontinuing at the end of the 2nd trimester | Minimal transfer into breastmilk; case reports of exposure in nursing infants show no adverse effects ; likely compatible |
| Ustekinumab | B | Insufficient data in pregnant women to inform risk assessment; animal studies show no adverse effects; additional safety data required | Present in milk in animal studies; exposure to infant expected to be low |
| Other Immunomodulators | |||
| Apremilast | C | Animal studies reveal possible increased risk of fetal loss; data on pregnant women are extremely limited | Detected in the milk of lactating mice; no data on presence in human milk or effects on the breastfed infant ; caution advised with breastfeeding |
| Colchicine | C | No adequate well-controlled studies in pregnant women; crosses the human placenta; animal studies demonstrate teratogenicity, embryofetal toxicity, and altered postnatal development ; human observational studies suggest risk to the fetus is low | Excreted in human milk; may affect gastrointestinal cell renewal and permeability in infants ; caution advised with breastfeeding |
| Other Medications | |||
| Acetaminophen | B | Analgesic of choice in pregnancy ; no known teratogenicity; possible increased risk of childhood attention-deficit/hyperactivity disorder and autism spectrum disorder after in-utero exposure | Low excretion in breast milk; one case report of rash in newborn following exposure; compatible |
| Ibuprofen | B/D b | Avoid during late pregnancy to prevent premature closing of the ductus arteriosus | Low levels in breast milk; compatible |
| Metformin | B | Not teratogenic in animal studies; crosses human placenta | Excreted into breast milk; potential risk of hypoglycemia in the infant; benefits and risks should be weighed ; considered compatible |
| Zinc gluconate | Not assigned | No adverse effects on maternal or neonatal outcomes in review studies with the use of up to 50 mg daily; excess zinc consumption may lead to copper deficiency | Does not appear to be transferred into breast milk ; considered compatible |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree