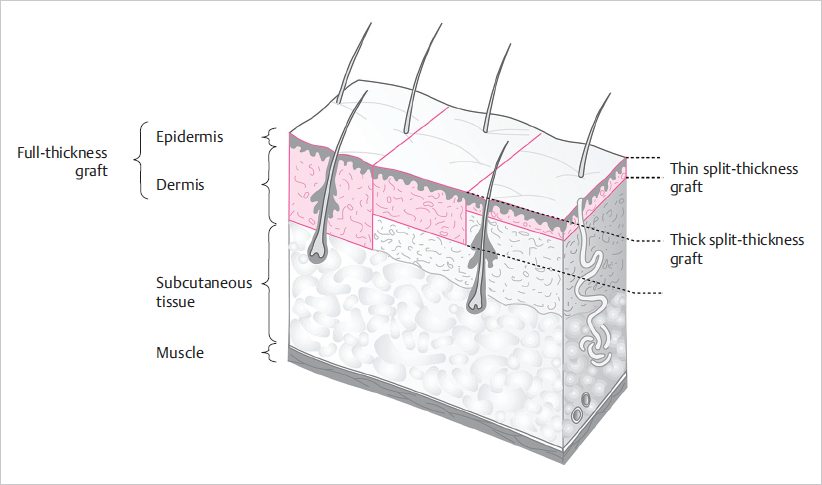
13 Grafts: Skin, Fascia, Nerve, Tendon, Cartilage, and Bone Abstract A graft is a segment of tissue of any kind that is transferred from one area to the other without maintaining its vascular supply, being therefore completely dependent on the wound bed for survival. Any tissue can potentially be transferred as a graft provided it is placed on the right environment. As with virtually any procedure in reconstructive surgery, the critical issue with grafting is vascularization, which in the case of grafts is governed by a balance between the thickness of the graft, its tolerance to ischemia, and the characteristics of the recipient bed, so that the thicker the graft or the poorer the bed, the lesser the chances of “take” and incorporation. In addition, because they lack their own blood supply, grafts are very vulnerable to contamination/infection. This chapter describes the basic principles of grafting of skin, fascia, nerve, tendon, cartilage, and bone together with the basic surgical technique of harvest for each of them. Keywords: graft, wound bed, inosculation, nerve injury, tendon repair The use of skin grafts can be traced as far back as 3,000 years ago in ancient India. Grafts are one of the most commonly performed procedures in plastic surgery today, allowing like-with-like reconstruction of a wide range of defects. Any tissue, from skin to bone, can be grafted, including dermis, fat, fascia, cartilage, and nerve. A graft is essentially a segment of tissue that is transferred from one area (donor site) to another (recipient site) without a blood supply of its own, thus depending on diffusion from the wound bed to survive until it becomes revascularized. Because of their lack of blood supply, the critical issue with grafts is related to the quality of the wound bed in terms of its vascularization and degree of contamination. Skin grafting represents one of the simplest and most straightforward methods in the reconstructive ladder, only above secondary intention healing and primary closure. Compared with healing by secondary intention, skin grafting helps to accelerate healing, decrease fluid loss, reduce scar contracture, and improve overall appearance of the healed wound. According to their thickness, skin grafts can be classified as full-thickness skin grafts (FTSGs) or split-thickness skin grafts (STSGs). FTSGs are composed of epidermis and the entire thickness of dermis, including adnexal structures, such as hair follicles and sweat glands, whereas STSGs include the epidermis and only a portion of the dermis ( The process of graft take is divided into three overlapping phases: plasmatic imbibition, inosculation, and revascularization. 1. Plasmatic imbibition (0–2 days): Following placement, the graft adheres to the wound bed by means of fibrin bonds and is kept alive by diffusion of nutrients from the bed. During this phase the graft appears edematous and may increase its volume by 40% because of fluid absorption. Fig. 13.1 Composition of skin grafts depending on the amount of dermis taken with the graft. (Reproduced from Weerda, Reconstructive Facial Plastic Surgery, ©2001, Thieme Publishers, Stuttgart.) 2. Inosculation (2–4 days): At this stage capillary buds in the recipient bed start to proliferate and connect with open vessels on the undersurface of the graft, establishing a fine vascular network that gives the graft a pink coloration. These vessels, however, are immature, and their flow is low. In addition, proliferation of fibroblasts and collagen deposition replace the fibrin network, keeping the skin graft adhered to the bed. 3. Revascularization (5–7 days): Approximately 5 days after grafting, the vascular connections become mature and fully functional, restoring blood flow through the graft. Angiogenesis, which is the development of new blood vessels from preexisting ones, further enhances graft vascularization. Note Graft adherence to the wound bed is most critical during the first 72 hours after grafting. Successful take of a skin graft is dependent, partly, on its thickness. The dermis is less vascularized in its deeper surface, and the number of cut capillary ends exposed in a thick graft is smaller than in a thin graft, which is why thinner grafts exhibit faster and more reliable “take” than thick or full-thickness grafts. Lymphatic circulation resumes within the first week, concomitantly with the revascularization process. Reinnervation starts approximately 1 month after grafting and may take several months to years to be completed, occurring faster in STSGs but to a higher extent in FTSGs. Pain is recovered first, followed by touch and finally temperature, with all three assuming the characteristics of the recipient site. Cutaneous appendages, due to their localization within the dermis, have a higher chance of regenerating in thicker grafts. Once reinnervated, sweat glands behave like those at the recipient site, whereas sebaceous glands retain the original characteristics of the donor skin. Likewise, hair, which has the potential to grow in FTSGs only, maintains donor-site characteristics ( All skin grafts undergo two processes of contracture: primary and secondary. Primary contracture occurs immediately after harvest and is mediated by contraction of elastic fibers. Because elastin is predominantly found in the dermis, the thicker the graft, the greater its primary contraction, which for FTSGs can be up to 40% of the original surface versus 10–20% for STSGs. Secondary contracture occurs as a consequence of scarring and is mediated by the contracting effect of myofibroblasts at the recipient site. The dermis in the graft exerts an in hibitory effect over these myofibroblasts; therefore, the thinner the graft, the greater its secondary contracture. Whereas (secondary) contracture may be beneficial in some situations to keep the skin graft as small as possible, in others it may lead to severe functional impairment or disfigurement, such as in the face or near joints. Table 13.1 Summary of characteristics of skin grafts
13.1 Introduction
13.2 Skin Grafts
13.2.1 Basic Science
 Fig. 13.1). Depending on the amount of dermis included STSGs can be further subdivided into thin (0.15–0.3 mm), intermediate (0.3–0.45 mm), and thick (0.45–0.75 mm). Inclusion of only a portion of the dermis in STSGs allows for spontaneous healing of the donor site through reepithelialization from residual dermal appendages and wound edges. Conversely, FTSG donor sites need to be closed with sutures because the removal of the full dermis leaves no potential for spontaneous reepithelialization.
Fig. 13.1). Depending on the amount of dermis included STSGs can be further subdivided into thin (0.15–0.3 mm), intermediate (0.3–0.45 mm), and thick (0.45–0.75 mm). Inclusion of only a portion of the dermis in STSGs allows for spontaneous healing of the donor site through reepithelialization from residual dermal appendages and wound edges. Conversely, FTSG donor sites need to be closed with sutures because the removal of the full dermis leaves no potential for spontaneous reepithelialization.
Mechanism of Graft Take
 Table 13.1).
Table 13.1).
Graft Contracture: Primary versus Secondary
| Split thickness | Full thickness |
Composition | Epidermis and partial dermis • Thin: 0.15–0.3 mm • Intermediate: 0.3–0.45 mm • Thick: 0.45–0.75 mm | Dermis and full epidermis |
Take | Easier and more predictable | Less robust and more dependent on wound bed conditions |
Availability | High | Low |
Donor site morbidity | Low—heals by reepithelialization, slower in thick grafts | Variable—leaves scar in donor site that needs to be concealed |
Contracture | Primary < secondary | Primary > secondary |
Reinnervation | Faster | Slower but better |
Regeneration of skin appendages | Absent in thin grafts; variable in thick grafts | More likely |
Hair growth | Absent | Possible, assuming donor-site characteristics |
Graft growth | Very little | Graft grows with child development |
Pigmentation | Unpredictable | More predictable |
Cosmetic appearance | Poor; not to be used in cosmetically or functionally sensitive areas Meshed grafts usually worst In some situation grafted muscle flaps can yield good aesthetic outcome (e.g., scalp reconstruction in bald patients) | Very good; possibility of replacing like with like |
13.2.2 Indications and Contraindications for Skin Grafting
In the broad sense, the main indication for skin grafting is any clean wound sufficiently vascularized to support the graft it is intended to be covered with. Conversely, the main contraindications would be infection, the presence of a poorly vascularized bed, and exposed structures, such as vessels, nerves, and bone. Likewise, skin grafts should be avoided in irradiated wounds. Full-thickness grafts are preferable for small defects in cosmetic or functional areas, where color match and absence of secondary contracture are a priority. Additionally, FTSGs are especially indicated for small wounds in the pediatric population because they retain the ability to grow with the developing child, thus reducing potential scar contracture. Finally, it is important to note that, in many cases, “graftable” defects are in fact much better reconstructed with flaps than with grafts.
13.2.3 Donor Sites for Skin Grafts
STSGs can be taken virtually from any area of the body, including the scalp and scrotum, a fact of special interest in burned patients with limited availability of healthy skin. By definition, donor sites of STSGs heal spontaneously in a period ranging from 7 to 21 days, depending on the thickness of the graft and the patient’s health status. Importantly, once healed, donor sites may be reused or, alternatively, they may be expanded prior to harvest in order to increase their available surface. Conversely, donor sites of FTSGs are limited by the fact that their harvest inevitably results in a linear scar. Ideally, these grafts should be obtained from inconspicuous areas that best match the characteristics of the skin at the recipient site. Facial defects may be reconstructed with skin grafts from the preauricular, postauricular, nasolabial fold, glabellar, and supraclavicular areas. Redundant skin from the upper eyelid provides an excellent match for eyelid reconstruction. Wounds in the hand and fingers are usually resurfaced with FTSGs from the hypothenar eminence, volar wrist crease, and volar aspect of the forearm. Skin from the abdomen and thigh may be used as FTSGs as well.
13.2.4 Wound Bed Preparation
The quality of the wound bed is critical to ensure a successful graft take, with FTSGS generally needing better vascularized beds to survive due to their thickness. All necrotic tissue must be removed, and there should be no signs of infection. Serial debridements and/or negative pressure wound therapy is sometimes needed to prepare the wound to receive a skin graft. As discussed in Chapter 4, negative pressure wound therapy reduces edema, improves perfusion, and decreases bacterial colonization and wound exudate, all of which favor formation of well-vascularized “graftable” granulation tissue, which should be flat and beefy red. Considering that one of the most common causes of failure is hematoma, good hemostasis cannot be overemphasized. Although the graft itself does have some hemostatic properties, coagulation of bleeding points is mandatory. Also, before placing the graft, wound margins must be sharply excised to allow good adherence of the graft to the edges.
Apart from ensuring the best possible local environment, the patient’s systemic conditions should also be addressed and optimized prior to skin grafting. Poorly controlled diabetes, malnutrition, vasculitis, chronic steroidal therapy, smoking, anticoagulant treatment, vascular insufficiency, history of radiotherapy, and ongoing chemotherapy can all impair the graft’s survival. Finally, whenever possible, donor site characteristics as to color, texture, and thickness should be considered and should ideally match those of the recipient site to obtain the best possible cosmetic outcome.
Note
The deleterious effects of chronic steroids on wound healing can reversed by the administration of vitamin A 25,000 IU/d orally for 3–5 days or 200,000 IU topically three times a day.
13.2.5 Harvest Technique
Split-Thickness Skin Grafts
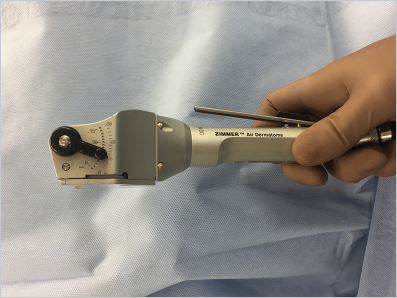
STSGs are harvested using dermatomes. Over the history of plastic surgery, a number of dermatomes have been introduced, a summary of which has been exhaustively elaborated by Ameer et al. Dermatomes can be broadly classified into freehand knives, drum dermatomes, and powered dermatomes (electric or air driven), with most units today using powered ones, due to their ease of use and consistency of results. The Zimmer air dermatome is one such example, which is powered by compressed water–pumped dry nitrogen (99.7% pure) operated at 100 psi ( Fig. 13.2). This instrument is less technique dependent than freehand knives, and the harvested grafts are usually more uniform, with predefined width and thickness. Still, some surgeons prefer to use freehand knives, such as the Humby or Watson dermatome, which, when used correctly, can yield uniform grafts as well.
Fig. 13.2). This instrument is less technique dependent than freehand knives, and the harvested grafts are usually more uniform, with predefined width and thickness. Still, some surgeons prefer to use freehand knives, such as the Humby or Watson dermatome, which, when used correctly, can yield uniform grafts as well.
Surgical Technique
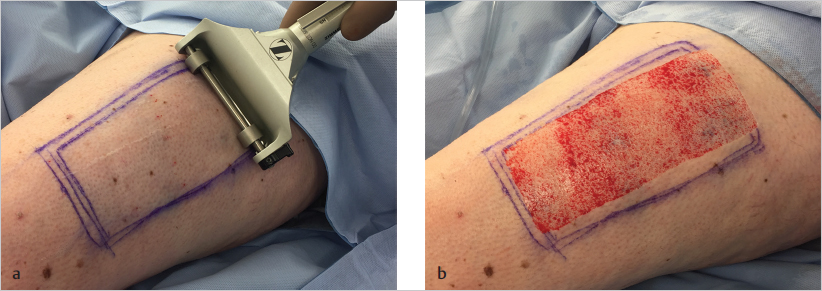
The donor site is shaved as necessary and properly prepared and draped. To reduce bleeding and postoperative pain, infiltration with an adrenalinecontaining local anesthetic can be performed. The dermatome is placed on the skin at a 30to 45-degree angle and advanced over the skin, applying uniform pressure until the end of the graft is reached and the dermatome is gently curved upward to cut the graft. Freehand knives involve a back-and-forth motion at the same time that the instrument is advanced on the skin until the desired graft size is obtained. For either procedure, the assistant’s role is crucial in maintaining an even and taut surface during harvesting to prevent step-offs and tears ( Fig. 13.3).
Fig. 13.3).
Once obtained, the graft is carefully handled with nontoothed forceps and applied to the wound with its dermal surface facing down. The shiny appearance of the dermis helps to distinguish it from the epidermis, which is duller. Also, the borders of the graft will usually roll toward the dermal side ( Fig. 13.4). Finally the graft is fixed with sutures, staples, or glue as needed, and a dressing is applied on top.
Fig. 13.4). Finally the graft is fixed with sutures, staples, or glue as needed, and a dressing is applied on top.
Meshed Skin Grafts
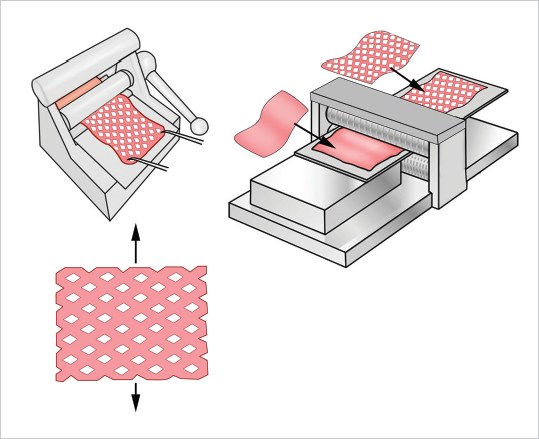
STSGs can be meshed to expand their surface, which is very useful in situations when the availability of healthy skin is limited in relation to the surface needing coverage. In addition, meshing allows harvesting a graft smaller than the wound, thereby minimizing morbidity at the recipient site. The mesher is a device that creates pie cuts on the graft as it is passed through ( Fig. 13.5), allowing expansion of 1.5:1, 2:1, 3:1 or larger. A ratio of 1.5:1 is usually used for small defects, whereas larger defects may require 3:1. Grafts may also be meshed manually using a no. 11 scalpel blade. Besides increasing the graft’s surface, meshing als o makes it more “flexible” and therefore more easily adaptable to irregular and concave surfaces. Furthermore, fluid drainage through the slits prevents hematoma or seroma formation beneath the graft. On the contrary, disadvantages of meshed grafts include increased fragility and twisting—identification of the dermal side may be more difficult in these cases. Also, gap epithelialization takes longer in highly meshed grafts, and secondary contracture is directly proportional to the ratio of meshing. The cosmetic appearance of meshed grafts is also inferior compared to that of nonmeshed grafts.
Fig. 13.5), allowing expansion of 1.5:1, 2:1, 3:1 or larger. A ratio of 1.5:1 is usually used for small defects, whereas larger defects may require 3:1. Grafts may also be meshed manually using a no. 11 scalpel blade. Besides increasing the graft’s surface, meshing als o makes it more “flexible” and therefore more easily adaptable to irregular and concave surfaces. Furthermore, fluid drainage through the slits prevents hematoma or seroma formation beneath the graft. On the contrary, disadvantages of meshed grafts include increased fragility and twisting—identification of the dermal side may be more difficult in these cases. Also, gap epithelialization takes longer in highly meshed grafts, and secondary contracture is directly proportional to the ratio of meshing. The cosmetic appearance of meshed grafts is also inferior compared to that of nonmeshed grafts.
Full-Thickness Skin Grafts
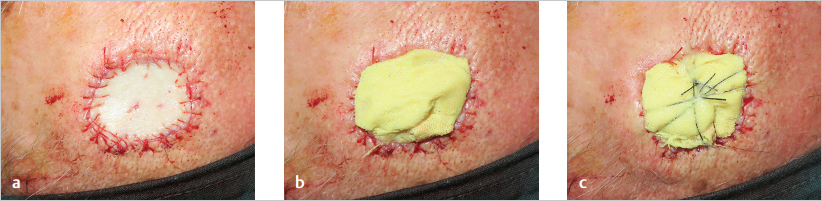
Harvesting of an FTSG is performed with a scalpel in the shape of an ellipse to allow linear closure of the donor site. In general, these grafts are harvested in a plane just deep to the dermis, trying to include as little fat as possible because this may compromise graft take. Remnants of fat should be trimmed off the graft with scissors. FTSGs are never meshed, though few incisions can be made to prevent hematoma formation. Finally, because harvesting of FTSG leaves behind a full-thickness wound incapable of reepithelializing, the donor site must be closed using standard suturing techniques ( Fig. 13.6).
Fig. 13.6).
Fig. 13.3 Harvest of split-thickness skin graft with the Zimmer air dermatome. (a) The dermatome is held against the skin at a 45-degree angle and run evenly and at constant pressure against the skin surface. (b) Donor site after harvesting.
Fig. 13.4 Split-thickness skin graft. Note the shiny appearance of the dermis facing up and edges of the graft rolling toward the dermal side.
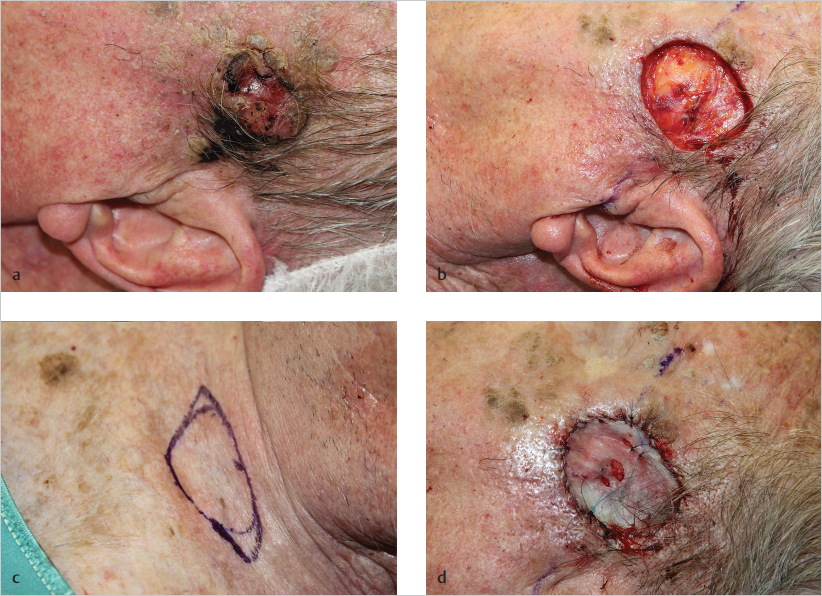
Fig. 13.6 Full-thickness skin grafting. (a) A 68-year-old patient presented with a squamous cell carcinoma on the left temple. (b) After excision. (c) Marking for harvest of a left supraclavicular full-thickness skin graft. (d) The wound was covered with the full-thickness skin graft.
13.2.6 Skin Graft Dressing
Grafts can be fixed in place using sutures, staples, or glue. Dressing then starts with a layer of nonadhesive gauze, such as Xeroform (Covidien) or Jelonet gauze (Smith & Nephew). A small amount of antibiotic ointment may be included in this layer. The nonadherent layer prevents the graft from adhering to the overlying sponge or gauze, which would cause the graft to be partially pulled away from the wound at the time of dressing change. The wound is then covered with a bulky dressing made of gauze or sponge (scrub or prep sponges are usually quite handy for this purpose), which is fixed to the surrounding skin with staples or sutured as a tie-over bolster dressing ( Fig. 13.7). Ensuring adequate compression of the graft against the wound is critical to prevent shear and formation of hematoma as well as to maximize contact to allow inosculation and revascularization. Considering the natural evolution of graft take, the dressing should not be removed for at least 5–7 days, provided that no signs of infection, hematoma, or other complications are seen. Negative pressure wound therapy can be used as an effective dressing, especially for medium to large defects, those over mobile areas, or when bed vascularization is not optimal, such as in patients with diabetes or peripheral vascular disease. Apart from effectively immobilizing the graft, negative pressure wound therapy increases the quantity and quality of take of STSGs when compared to tie-over dressings.
Fig. 13.7). Ensuring adequate compression of the graft against the wound is critical to prevent shear and formation of hematoma as well as to maximize contact to allow inosculation and revascularization. Considering the natural evolution of graft take, the dressing should not be removed for at least 5–7 days, provided that no signs of infection, hematoma, or other complications are seen. Negative pressure wound therapy can be used as an effective dressing, especially for medium to large defects, those over mobile areas, or when bed vascularization is not optimal, such as in patients with diabetes or peripheral vascular disease. Apart from effectively immobilizing the graft, negative pressure wound therapy increases the quantity and quality of take of STSGs when compared to tie-over dressings.
13.2.7 Donor-Site Management
By definition, donor sites of STSGs heal by reepithelialization from the wound edges and remaining adnexal structures in a period ranging from 7 to 21 days, depending on graft thickness and the size of the donor area. Importantly, it is the epidermis and not the dermis that regenerates. The management of the donor area varies greatly among units, which shows that STSG donor sites will heal regardless of the dressing used, as long as they are kept clean and moist. These are simple superficial wounds, similar to an abrasion, that in most instances do not need much more than basic wound care. That said, patient comfort, frequency of change, and other logistical factors are important when choosing the best dressing. A large number of dressings are available for donor site management. Typically a layer of nonadherent Xeroform gauze is applied in direct contact with the wound, covered with gauze and a bandage and left to dry and adhere to the healing skin until it detaches once reepithelialization is complete. Artificial semipermeable transparent dressings (e.g., Opsite [Smith & Nephew], Biobrane [Smith & Nephew], or Tegaderm [3M]) promote exudate accumulation, which requires drainage with a syringe or through perforations made prior to application. By being transparent, these dressings allow continuous inspection of the wound without the need to remove them. Several other dressings have been studied, including biological (e.g., cadaveric skin, sterile irradiated allograft, pig skin, amniotic membrane, cultured keratinocyte grafts), alginates, or silver-base dressings. Finally, spare graft provides an excellent dressing and should be applied if present. Donor sites of FTSGs are managed as any primarily closed wound.