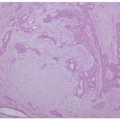
TABLE 7-1. Selected nonsyndromic ichthyoses | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||
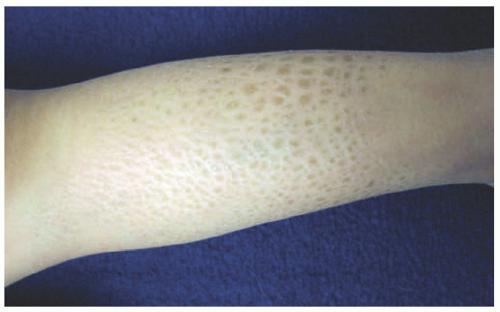 FIGURE 7-1. A child with ichthyosis vulgaris. The lower leg shows dry, polygonal, hyperpigmented scaling. |
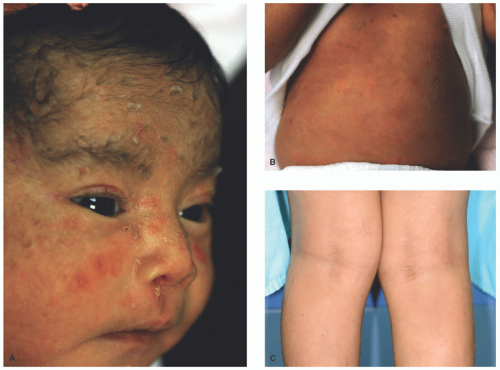
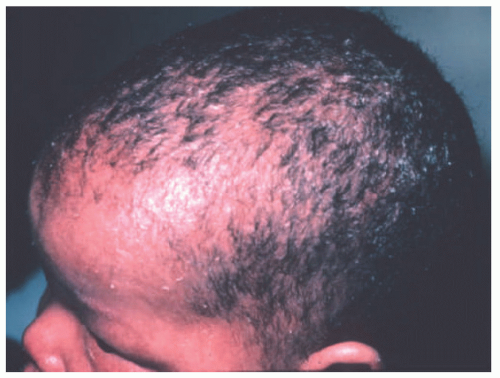
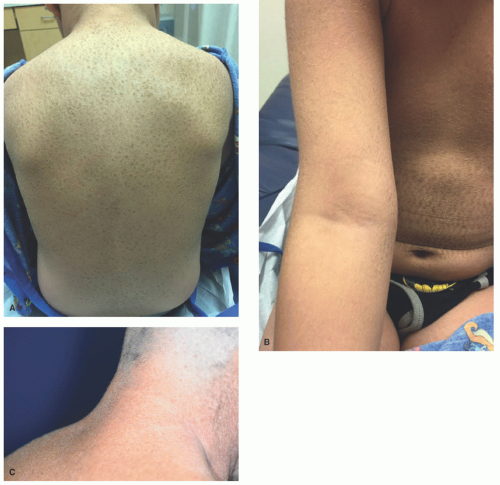 FIGURE 7-2. X-linked recessive ichthyosis with characteristic hyperpigmented scale on the back extending onto the neck (A and B) and conspicuous sparing of the antecubital fossa (C). |
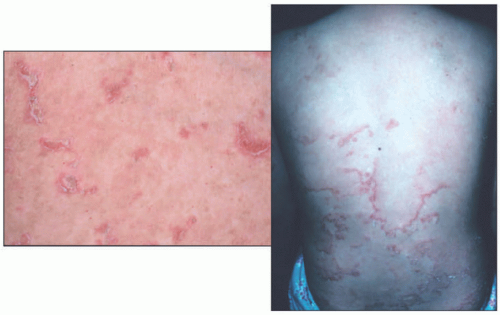
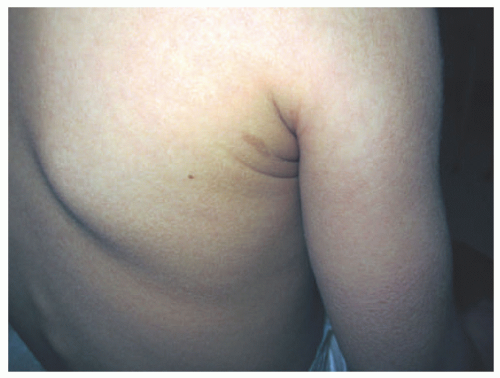 FIGURE 7-4. Autosomal recessive congenital ichthyosis with a congenital ichthyosiform erythroderma phenotype, showing fine, diffuse scale with mild underlying erythema. |
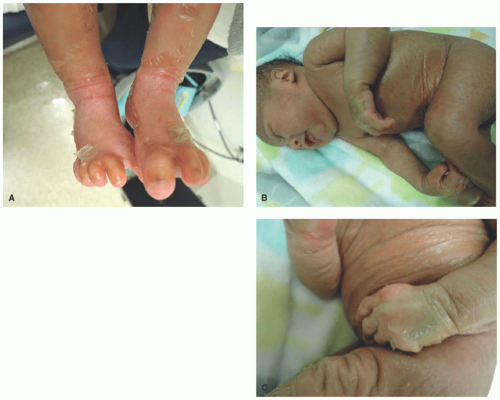 FIGURE 7-5. Collodion baby. This neonate is encased in a thick, yellowish, taut, glistening, parchment-like membrane that restricts its movements (A-C). |
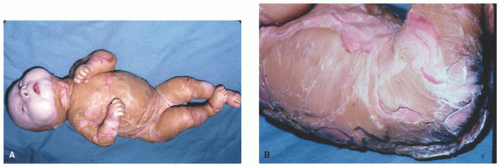 FIGURE 7-6. An infant with Harlequin ichthyosis with extremely thick, fissured scaled skin (A and B). Courtesy of Dr Kenneth Greer. Department of Dermatology. University of Virginia. |
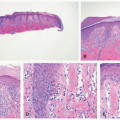
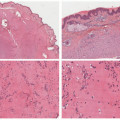
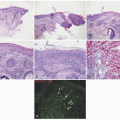
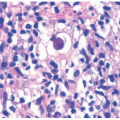
TABLE 7-2. Histopathologic findings for selected nonsyndromic ichthyoses | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||
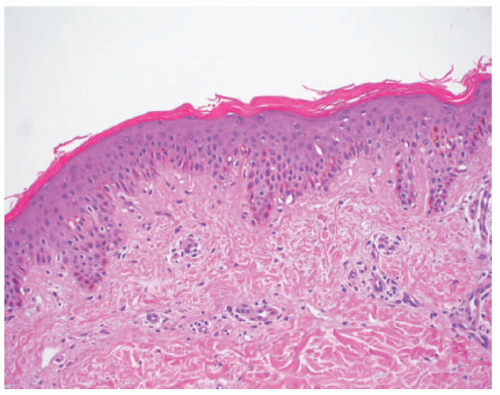
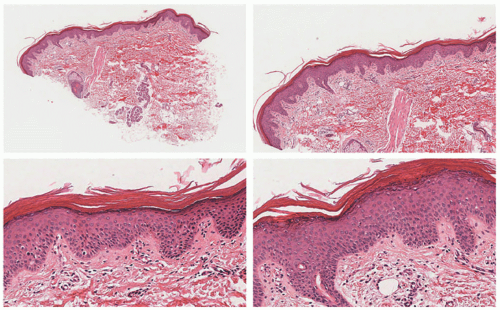
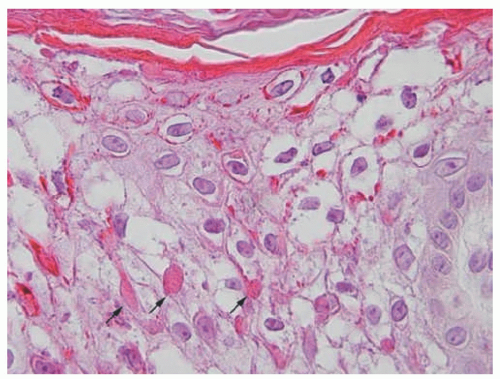
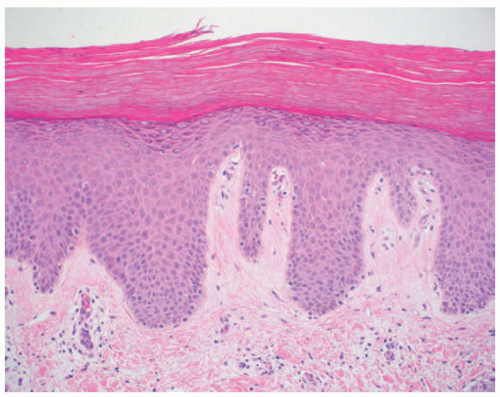
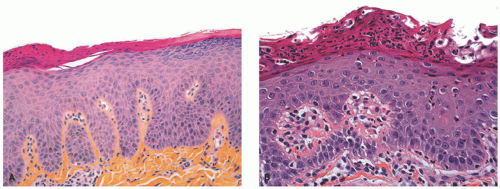
acanthosis. Focal parakeratosis, hypogranulosis, and exocytosis of neutrophils are variably present (Figure 7-14).47,48,49 In erythrodermic cases, parakeratosis can be confluent. A perivascular lymphohistiocytic infiltrate may be present in the superficial dermis.50 Periodic acid-Schiff staining demonstrates densely staining granules in upper epidermal layers.8 A microscopic evaluation of affected hair shafts reveals trichorrhexis invaginata, with a telescoping of the distal hair shaft into the proximal shaft.43 Other hair shaft findings, such as trichorrhexis nodosa, pili torti, and “matchstick” abnormality, can also be seen.
syndromic and nonsyndromic ichthyoses, eczematous dermatitis, and dermatophytosis.
progressive neurologic abnormalities and mild ichthyosis. Symptoms and progression are mitigated by a diet low in phytols. Skin biopsy specimens show hyperorthokeratosis and a diminished granular layer and can be differentiated from ichthyosis vulgaris by the presence of lipid accumulation on electron microscopy and by clinical history.
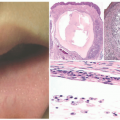
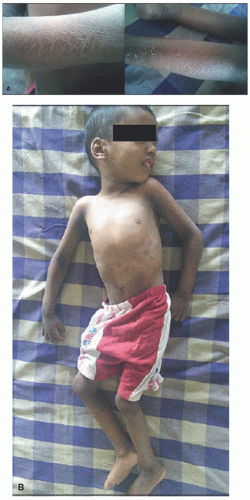 FIGURE 7-15. Marked ichthyosis in a patient with Sjögren-Larsson syndrome. A, Brown color diamond-shaped adherent scales on the upper limb and peeling of skin on the lower limb. B, Kyphoscoliosis of the trunk is present. Obtained with permission from Subramanian V, Hariharan P, Balaji J. Sjögren-Larsson syndrome: a rare neurocutaneous disorder. J Pediatr Neurosci. 2016;11(1):68-70, Figure 1 and 2. |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree