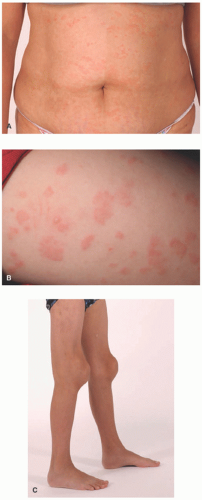
interleukin-1β (IL-1β). Cryopyrin is a critical component of the inflammasome, a multiprotein complex responsible for generation IL-1β in response to pathogens or “danger signals” from dead or dying host cells. NLRP3 mutations in patients with CAPS appear to activate the inflammasome in the absence of pathogenic insults.3 Nearly all patients with FCAS and MWS have inherited mutations in NLRP3, whereas NLRP3 mutations found in NOMID patients tend to occur de novo.10 Furthermore, about half of patients with NOMID do not have detectable germline NLRP3 mutations; however, many somatic mutations have been found in these patients.15
TABLE 13-1. Clinical features, histology, and genetics of selected autoinflammatory syndromes with unique histopathologic features | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
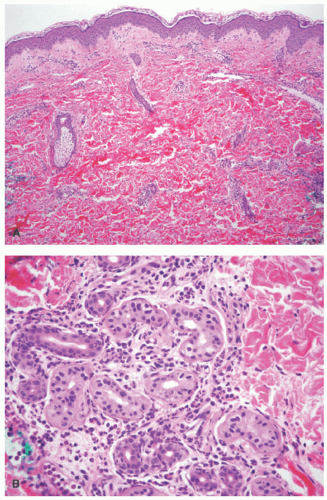
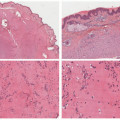
feature is characteristic perieccrine neutrophilic infiltration similar to neutrophilic eccrine hidradenitis.21 There is an absence of karyorrhexis or leukocytoclasis and vasculitis. There are no epidermal changes and no eosinophils.
inflammation and leukocytoclasis, but there is an absence of perieccrine involvement, vasculitis, or dermal edema. The histologic pattern found in CAPS characteristically involves perieccrine neutrophilic infiltration but lacks significant leukocytoclasis. Urticarial vasculitis has the presence of both leukocytoclasis and vasculitis. Sweet syndrome is characterized by an intense dermal infiltration of neutrophils with leukocytoclasis and significant dermal edema.
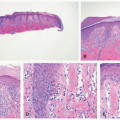
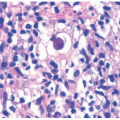
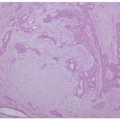
feature of DIRA is folliculocentric infiltrate of neutrophils with involvement of the follicular infundibula, as other disorders that present with subcorneal pustules rarely involve the hair follicle. Pustular psoriasis may show features common in more chronic plaque psoriasis such as an elongation of rete ridges, papillary epidermal thinning, and dilated, tortuous papillary vessels. In addition, the lesions of pustular psoriasis are characterized by parakeratosis, spongiosis, and the spongioform pustules of Kogoj. Deficiency of IL-36 receptor antagonist (DITRA) also presents with an erythematous pustular rash mimicking psoriasis but lacks internal organ manifestation.33,34,35 The histology of DITRA demonstrates spongiform pustules, acanthosis, and parakeratosis.34 Subcorneal pustular dermatosis (Sneddon-Wilkinson disease) shows perivascular neutrophilic infiltrate with occasional monocytes and eosinophils. Acute generalized exanthematous pustulosis is nonfollicular and often contains eosinophils.
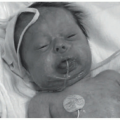
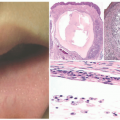
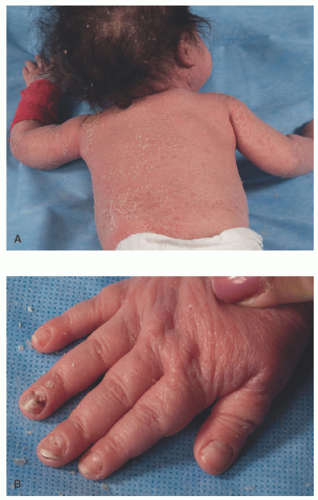 FIGURE 13-3. Deficiency of the IL-1 receptor antagonist. A, Erythroderma, pinpoint pustules, and ichthyosiform scale in a neonate. B, Nail changes including pitting and onychomadesis. |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree