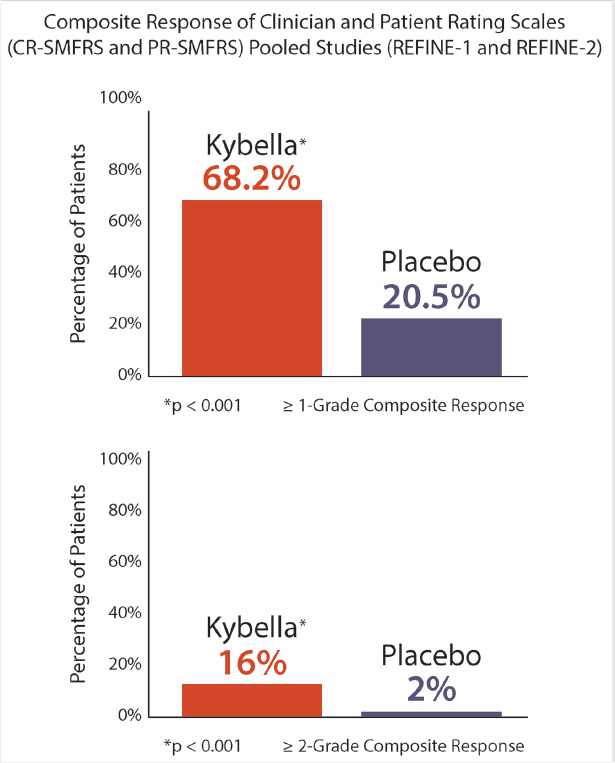
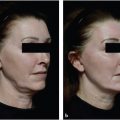
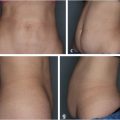
15 Future Considerations Summary Noninvasive and minimally invasive cosmetic medicine applications are undergoing rapid innovation and development on nearly a monthly basis, with new and exciting opportunities for future development. This chapter presents several new and emerging technologies that follow the typical core principles of noninvasive cosmetic medicine. Keywords: CoolSculpting, core principles, cosmetic medicine, imaging, innovation, Kybella, microneedling, nonsurgical skin tightening, poly lactic co-glycolic acid (PLGA), Silhouette InstaLift, skin laxity, soft tissue ptosis, Thermi, three-dimensional (3D) imaging Key Points • The core principles of noninvasive cosmetic medicine are to restore lost volume, remove excess, and lift and/or tighten where appropriate. • Several new technologies (Kybella® [Allergan, Inc.], Silhouette InstaLift® [Sinclair Pharma], and Dermal Rejuvenation Enhanced by Amniotic Membrane [DREAM, Advanced Dermal Sciences, LLC]), although in their infancy, show great promise and may have far-reaching effects on the field of cosmetic medicine. • Imaging modalities, such as Vectra (CIS), not only help with patient communication but also may help to also accurately direct therapy. • Cosmetic medicine will likely go the way of interventional cardiology, in that technological innovation will advance the field, whereas noninvasive or minimally invasive therapies, in a stackable approach, will lead to surgical or near-surgical results. Noninvasive and minimally invasive cosmetic medicine applications are undergoing rapid innovation and development on nearly a monthly basis, with new and exciting opportunities for future development on the horizon. Behind any emerging technology or modality, the core principles of noninvasive cosmetic medicine are typically followed, including the return of lost volume, the removal of excess, and lifting and/or tightening where indicated. These common themes or principles help define future innovations and allow for continued development and easy integration with existing effective platforms. Within this chapter, we will present several new and emerging technological considerations, which we feel adhere well to the previously mentioned principles and will integrate well and help advance the field of noninvasive cosmetic medicine to help the provider gain near-surgical results. We will present several areas of interest that we believe are likely areas of future investigation based on a review of current literature, both in print and pending publication. While the following represents speculation, it is based in sound science and will allow the reader to consider how one may go about evolving a clinical practice in the ever-changing world of cosmetic medicine. Facial volume loss is a core component seen in every facial aging patient seeking rejuvenation. Implants, fillers, and autologous fat injection have been the aesthetic surgeons answer to the problem of facial volume loss. We are seeing a new emphasis on biocompatibility in implantable technology to better compete with the concept of autologous fat grafting. There will likely be an evolution to fast, affordable, genetically engineered filler substances that will not require a donor site, given the increasingly available ability to sequence DNA/RNA through saliva/buccal swabs (see Video 1.1). The nonsurgical removal of unwanted fat is a common theme among cosmetic medicine patients. CoolSculpting® (ZELTIQ Aesthetics, Inc.) has become a mainstay modality in noninvasive fat removal (see Product Index (p. 180)). One issue that remains in both surgical and nonsurgical fat removal is how to treat the patient with skin laxity that is worsened by fat reduction. The ability to use nonsurgical skin tightening provides a new frontier to enhance body contouring and is the subject of an ongoing study. As technology continues to evolve in the world of nonsurgical skin tightening and resuspension, we will continue to see more near-surgical and possibly even betterthan-surgical results. Kybella® (Allergan, Inc.) is a newly FDA-approved noninvasive injection for the treatment of moderate to severe convexity or fullness associated with submental fat in adults (see Product Index (p. 170)).1 Formulated as a nonhuman, nonanimal version of deoxycholic acid (10 mg/mL), Kybella is a cytolytic agent that destroys cell membranes. Once injected into subcutaneous fat, it causes the lysis of adipocytes, no longer allowing them to store or accumulate fat. The administration of Kybella is considered fairly straightforward. In 15 to 20 minutes, the patient’s submental area is typically injected with 4 to 6 mL (2 to 3 vials) of agent divided into 0.2 mL aliquots separated by 1 cm, not to exceed 10 mL per session.1 In two randomized, double-blind controlled trials versus placebo (N = 514 vs. 508), in North America, Kybella proved to be efficacious within statistical significance (p < 0.001) with a 79% patient satisfaction rate1,2 ( Although it appears to be safe and efficacious, side effects have been noted. Because of its cytotoxic properties, Kybella’s ability to destroy cell membranes is not specific, so there exists the theoretical risk of unwanted tissue destruction (i.e., skin and muscle). Additionally, within the clinical trials, 4% of patients suffered temporary marginal mandibular injury, 2% suffered temporary dysphagia, and most commonly, 72% of patients suffered from various degrees of edema, bruising, pain, numbness, or redness. These findings led to the warning of not injecting within 1 to 1.5 cm of the marginal mandibular nerve and to avoid its use in patients with active or a history of dysphagia.1 Fig. 15.1 Composite responder rates of Kybella compared to placebo. (CR, clinician reported; PR, patient reported; SMFRS, submental fat rating scale.) (Reproduced with permission from http://hcp.mykybella.com/about-kybella/clinicalresults, 2015.) Kybella truly is a product in its infancy, whose potential is still being unlocked and discovered. It is our opinion that it will ultimately find its utility in small areas of excess reduction, particularly when used with energy-based modalities to perform nonsurgical facial and neck rejuvenation, which will be exciting areas of research to come. This is a product of great potential excitement, not as much in its current presentation but rather as an agent to combine with focused ultrasound or radiofrequency to enhance skin tightening, skin contouring, or enhancement in skin resurfacing. Given what we know about the inflammatory cascade and the ability of Kybella to stimulate inflammation and even targeted cell death, this could be a product that could create more permanent neurolysis and potentially replace neurotoxin use. If one allows for scientific consideration, inflammation is a necessary and key component of all things in cosmetic medicine—adipocyte lysis, thermal destruction with collagen and epithelial replacement, and longevity of surgical and nonsurgical lifting through the stimulation of collagen production in the form of what we consider scar formation. The ability to modulate these pathways is extremely exciting and will likely allow for the reduced need for traditional invasive applications. Skin laxity and soft tissue descent are common themes in the aging face that have historically been treated through the surgical facelift. However, because younger patients have started to seek out cosmetic surgery, the need for less invasive procedures with minimal downtime have become desirable. Multiple modalities have been developed to fill this void, most of which are energy-based, namely in the form of lasers, microfocused ultrasound, or radiofrequency. Regarding soft tissue ptosis, suture suspension systems, socalled thread lifting, have fallen in and out of favor over the years.3 Barbed suture was once thought to be the answer for noninvasive soft tissue lift4 ( Silhouette InstaLift (Sinclair Pharma), is a newly FDA approved facial soft tissue suspension system. Based closely on its European counterpart, Silhouette Soft® (Sinclair Pharma), InstaLift is a fully absorbable suspension suture with 8, 12, or 16 bidirectional cones per suture, all made from poly lactic coglycolic acid (PLGA), which is biocompatible and biodegradable5(
15.1 Introduction
15.2 Modalities
 Fig. 15.1). Of those studied, nearly 60% of patients required 6 treatments (at 1-month intervals) to obtain the highest percentage of improvement, with many patients noting results within 2 to 4 treatments.
Fig. 15.1). Of those studied, nearly 60% of patients required 6 treatments (at 1-month intervals) to obtain the highest percentage of improvement, with many patients noting results within 2 to 4 treatments.
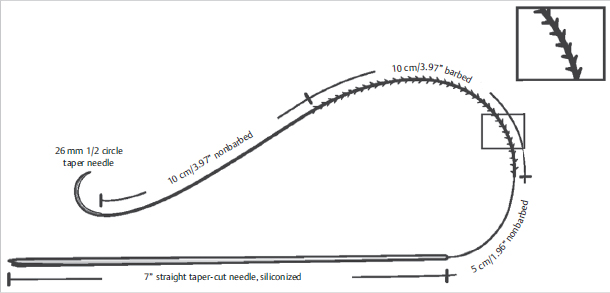
 Fig. 15.2). However, due to its unidirectional design, lack of absorption, and need for superficial placement to catch dermis, barbed suture was fraught with complications such as breakage, palpability, extrusion, and short-lasting results.
Fig. 15.2). However, due to its unidirectional design, lack of absorption, and need for superficial placement to catch dermis, barbed suture was fraught with complications such as breakage, palpability, extrusion, and short-lasting results.
 Fig. 15.3). The biological makeup of the material has been shown to be biostimulating when combined with injectable filler such as PLGA. This may lead to the possibility of adding volume to the lift area, thereby potentiating the result by placing injectable filler adjacent to the suspension-lifting bidirectional suture. Additionally, the resorbable cones help to provide an increased surface area to maximize suspension traction and create a strong fixation in the subcutaneous tissue, while encapsulation around the cones creates a solid support to the soft tissue, helping to mitigate the risk of migration and extrusion.6
Fig. 15.3). The biological makeup of the material has been shown to be biostimulating when combined with injectable filler such as PLGA. This may lead to the possibility of adding volume to the lift area, thereby potentiating the result by placing injectable filler adjacent to the suspension-lifting bidirectional suture. Additionally, the resorbable cones help to provide an increased surface area to maximize suspension traction and create a strong fixation in the subcutaneous tissue, while encapsulation around the cones creates a solid support to the soft tissue, helping to mitigate the risk of migration and extrusion.6
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine