Fungal and Algal Cutaneous Infections
J. Olufemi Ogunbiyi
Mark R. Wick
The cutaneous mycoses are fungal diseases of the hair, nails, and skin. They may be further separated into superficial dermatophytoses and deep mycoses.
SUPERFICIAL DERMATOPHYTOSES
Definition and Epidemiology
Superficial dermatophytoses are fungal infections of the skin, hair, and nails. They have a worldwide occurrence, but are more prevalent in tropical climates. The prevalence of the causative fungi differs in various geographical locations, but modern travel patterns have altered the distribution of these infections. People of virtually all ages and both sexes can be affected, but some clinical variants—such as tinea capitis—are more common in children.
Etiology
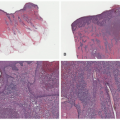
Dermatophytes belong to the genera Microsporum, Trichophyton, and Epidermophyton. These fungi can also be classified by the source of infection: anthropophilic—acquired from humans; zoophilic—acquired from animals; and geophilic—acquired from the soil. The organisms may be transmitted by direct contact or through a fomite intermediary. They invade the epidermis by adhering to surface keratinocytes and penetrating between the cells. A host response is variable and depends on several potential modifying factors. They include the presence of sebum, immunosuppression, and the specific source of the infection. As examples, nonanthrophilic organisms elicit a robust inflammatory reaction, and the prevalence of tinea capitis is lower in adolescents than in younger children because sebum—produced in greater quantity during puberty—has fungistatic properties. Particular clinical presentations of the dermatophytoses depend on the specific fungal organism, the immune status of the host, previous treatment, if any, and the site of skin involvement (Table 20-1) (Figure 20-1).
TABLE 20-1. Diagnostic terms for forms of tinea | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Tinea Infections
TINEA CAPITIS
Clinical Presentation
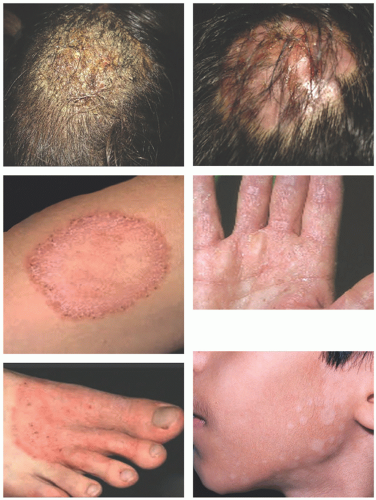
Tinea capitis is seen predominantly in the first decade of life. Responsible organisms include Microsporum audouinii, Trichophyton schoenleinii, and Tricholosporum violaceum. Involvement of the hair shafts may include the cortex, ectothrix, or endothrix. Clinical features include widespread scaling on the scalp with a seborrheic dermatitis-like pattern, as well as patchy alopecia. An inflammatory form of the disease, known as kerion, results from infection by zoophillic organisms such as Trichophyton verrucosum or Trichophyton mentagrophytes. Favus is a particularly severe form of kerion, usually caused by T. schoenleinii. Dermoscopic findings in tinea capitis include comma-shaped hairs with black dots.
Histologic Findings
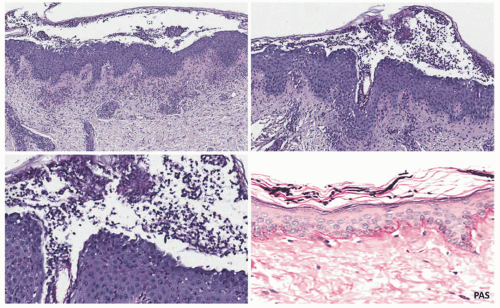
The causative organisms invade the stratum corneum and then involve hair follicles, extending into the shafts to the level of the mid-follicle. Endothrix infections are defined as those showing intrapiliary hyphae or arthrospores. Endo-ectothrix infections are those that exhibit fungal elements within and around altered hair shafts. In the superficial dermis, the inflammatory response varies from scant perivascular lymphocytic infiltration to dense and diffuse effacement of the corium by acute and chronic inflammatory cells in kerion (Figure 20-2). Neutrophils are commonly seen in the stratum corneum. Rupture of hair follicles may provoke a granulomatous response.
TINEA CORPORIS
Clinical Presentation
Tinea corporis presents as a pruritic annular rash with an active border that is studded with papules and vesicles. The lesional center shows postinflammatory dyspigmentation, scaling, and scarring. In children with fair skin, the lesions may be erythematous.
Histologic Findings
The histopathologic images that are seen most commonly in tinea corporis are those of subacute or chronic spongiotic dermatitis or erythema perstans. The expanding edges of the lesions show acanthosis and parakeratosis, with inflammatory cell exocytosis, including neutrophils, in the parakeratotic crust. Fungal hyphae are demonstrable in the stratum corneum with periodic acid-Schiff (PAS) stains. Occasionally, idiosyncratic inflammatory reactions can resemble those of erythema multiforme, granuloma faciale, and nodular granulomatous parafolliculitis, and subepidermal bullae may also be seen (Figure 20-3).
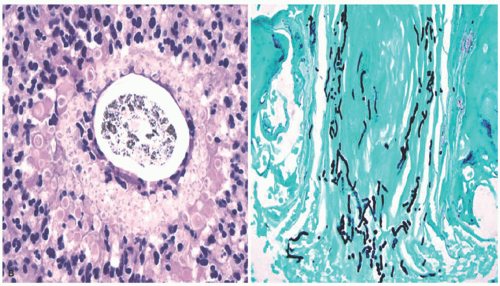 FIGURE 20-2. (continued) B, Acute and chronic inflammation surrounds infected hair follicles in kerion (left panel). A GMS stain demonstrates intrafollicular fungal organisms. |
TINEA PEDIS
Clinical Presentation
Tinea pedis, or “athlete’s foot,” is usually caused by anthropophilic fungi such as Trichophyton rubrum, Trichophyton mentagrophytes, and Epidermophyton floccosum. The most common form of this condition features interdigital lesions that are characterized by peeling, maceration, and fissuring. The fourth interdigital cleft is often affected, and lesions may also involve the plantar surfaces of the toes. Chronic hyperkeratotic tinea pedis is typified by diffuse scaling or hyperkeratosis of both feet. Typically, the dorsal surfaces are unaffected, but, in severe cases, lesions may involve the entire foot. Inflammatory and vesicular tinea pedis produces painful, pruritic vesicles or bullae, most often on the instep or the sole. Complications of tinea pedis include cellulitis, lymphangitis, and lymphadenopathy. A hypersensitivity reaction to the dermatophytosis (dermatophytids) may subsequently develop on the palms of one or both hands and the sides of the fingers. Papules, vesicles, bullae, or pustules may develop, often symmetrically. The resulting eruption may mimic dyshidrotic eczema (pompholyx).
Histologic Findings
Tinea pedis is commonly complicated by a secondary bacterial infection, with neutrophilic infiltrates and intracorneal pustules. In ordinary cases, the histopathologic images are those of acute vesicular dermatitis or chronic psoriasiform dermatitis. PAS stains will show the causative organisms in the stratum corneum.
TINEA NIGRA
This is a form of superficial dermatophyte infection caused by Hortaea werneckii.
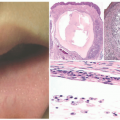
Clinical Presentation
Patients present with asymptomatic brown macules or patches on the palms, soles, neck, and trunk. The lesions have a darker pigment at the advancing borders, unlike acral nevi.
Histologic Findings
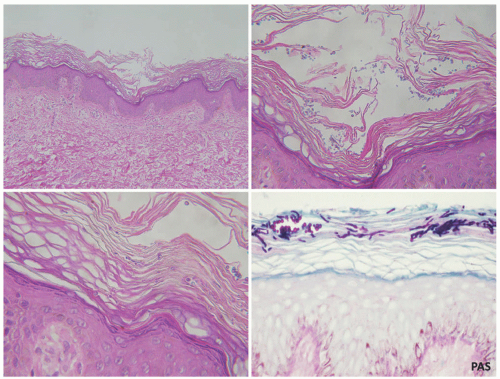
This form of infection has minimal to no inflammation. The epidermis and dermis of the acral skin infected with tinea nigra are largely unremarkable at low-power magnification. In the superficial aspects of the stratum corneum, numerous short, segmented hyphae and spores are seen. These organisms have a characteristic brown-yellow color on routine hematoxylin and eosin sections (Figure 20-4).
The hyphae can be proven with the use of special stains (Gomori methenamine-silver [GMS] and PAS). Melanin pigment is confined to the stratum corneum and can be demonstrated with the use of Fontana-Masson stain.
The hyphae can be proven with the use of special stains (Gomori methenamine-silver [GMS] and PAS). Melanin pigment is confined to the stratum corneum and can be demonstrated with the use of Fontana-Masson stain.
TINEA (PITYRIASIS) VERSICOLOR
Definition and Epidemiology
Pityriasis versicolor (PV) is a superficial fungal infection of the skin caused by Malassezia organisms. It is characterized by hypo- or hyperpigmented scaly macules. PV is seen internationally, but usually in tropical climates. It occurs most often in adolescents, but also has been seen in a few preteen children. Some reports have suggested an increased prevalence in females.
Etiology
Malassezia is a part of the normal skin flora. It is a dimorphic lipophilic fungus that can exist as both yeasts and mycelia. The latter is the pathogenic form. Environmental factors and host susceptibility have been implicated in the development of skin lesions. The genus Malassezia has been subdivided into seven species—Malassezia globosa, M. sympodialis, M. furfur, M. slooffiae, M. pachydermatis, M. restricta, and M. obtusa. It would appear that the predominant species for PV is geographically determined.
Clinical Presentation
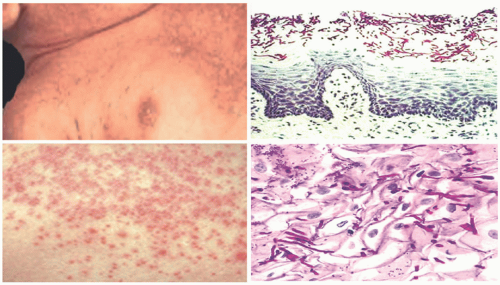
Characteristic lesions of PV are hypopigmented or hyperpigmented scaly macules that are initially perifollicular but then coalesce to form larger macules. Most lesions are asymptomatic, but they may be pruritic. Stretching the lesions on examination makes scaling obvious. Pigmentary changes are most apparent in dark-skinned persons. The Woods lamp examination is useful in delineating the borders of the lesions. Seborrheic areas of the face, chest, and upper back are commonly affected, although lesions may also extend to the extremities or be mainly located on them (“inverse” distribution). Extensive skin involvement may suggest an underlying immunosuppressive state. Mild erythema or inflammation may be seen around a few hypopigmented lesions.
Residual hypopigmentation persists after treatment, especially in covered areas; those in sun-exposed skin fields recolor faster. Diagnosis is usually based on clinical findings, together with the results of potassium hydroxide (KOH)-digested microscopic studies of skin scrapings.
Histologic Findings
In hematoxylin and eosin (H&E) sections, the causative organisms are usually seen as short wavy hyphae in the stratum corneum, associated with clusters of spores. The hyphae can be entrapped within the cornified layer (the “sandwich sign”). They are best seen with PAS or GMS stains, with associated parakeratosis and intracorneal neutrophilia (Figure 20-5). Epidermal spongiosis is variably present, but is often seen in lesions of the feet. In chronic lesions, one may see psoriasiform epidermal hyperplasia with papillary dermal chronic inflammatory cell infiltrates that are predominantly perivascular.
CAPSULE SUMMARY
TINEAS
The forms of tinea are caused by Microsporum, Trichophyton, Epidermophyton, and Malassezia, which are acquired from the soil or animal intermediaries. The specific names of the Tineas depend on the skin regions that they affect or their clinical appearances. The histopathologic image in all of the Tineas is identical, featuring epidermal parakeratosis, intracorneal neutrophilia, and the presence of fungal organisms in the stratum corneum. Chronic lesions may have a psoriasiform appearance.
CANDIDIASIS
Definition and Epidemiology
A spectrum of skin disorders is caused by yeasts in the genus Candida. They represent the commonest mucocutaneous fungal infections in children. Cutaneous candidiasis occurs worldwide, but the organisms especially thrive in a warm, humid environment. Neonates and immunocompromised children are preferentially affected.
Etiology
The most frequently seen Candida species in mucocutaneous disease is Candida albicans, which is dimorphic. Its yeast phase predominates, but the mycelial form is the invasive one. The other species of Candida include C. tropicalis, C. dubliniensis, C. parapsilosis, C. guilliermondii, C. krusei, C. pseudotropicalis, C. lusitaniae, C. zeylanoides, and C. glabrata (formerly Torulopsis glabrata). The dominant species varies with the geographical location. Colonization with C. albicans may occur during birth or infancy. Its mechanism of tissue invasion is unclear, but it is believed that the organism produces enzymes that encourage adhesion to epithelium. The pertinent defect in host defense mechanisms appears to be a loss of integrity in the stratum corneum via maceration or frictional damage. Malnutrition, endocrine dysfunction, and prolonged use of antibiotics are other predisposing factors.
Clinical Presentation
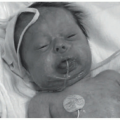
Clinical features of cutaneous candidiasis are variable; they are dependent on patient age, anatomic site, and immune status. Pruritus is common in all forms of cutaneous
candidiasis, and it commonly assumes an “angry” erythematous appearance. In congenital candidiasis, an extensive eruption of pink-red macules and papules is present at birth or within a few hours thereafter. The rash is generalized, and palmoplantar surfaces may be involved. Diaper dermatitis is seen in the genitoperineal skin. Pruritus is common, subcorneal pustules are seen, and irregularly shaped lesions are encountered with satellitosis.
candidiasis, and it commonly assumes an “angry” erythematous appearance. In congenital candidiasis, an extensive eruption of pink-red macules and papules is present at birth or within a few hours thereafter. The rash is generalized, and palmoplantar surfaces may be involved. Diaper dermatitis is seen in the genitoperineal skin. Pruritus is common, subcorneal pustules are seen, and irregularly shaped lesions are encountered with satellitosis.
Vulvovaginitis presents in adolescents who complain of pruritus, vaginal soreness, and a whitish discharge. Vesicles and pustules may extend to the surrounding skin and perineum as well. It is common in girls who have taken antibiotics long term.
Candida balanitis is seen most often in uncircumcised boys, manifesting as transient tiny papules or pustules on the glans penis. They may rupture and leave a peeling edge. The lesions are associated with soreness and burning. In some individuals this condition is self-limiting, but it may also become chronic.
Intertriginous candidiasis is encountered in skin areas that are subject to friction, especially in obese children. Those include the groin, inframammary skin, and the intergluteal cleft. Lesional vesicles and pustules rupture and are associated with maceration and fissuring. The affected skin areas have a white rim that surrounds an erythematous and macerated base. Satellite lesions are commonly seen, and they may coalesce.
Candida paronychia is not common in children. It produces inflammatory, painful lesions of the nail bed that are usually painful. Persons who repeatedly dip their hands in water are preferentially affected. The nail beds are usually swollen and the nail cuticles are absent; a whitish discharge may be expressed from the nail bed. Several fingers may be involved.
Histologic Findings
Candida dermatitis may have an acute vesicular or subacute spongiotic appearance, or it may become psoriasiform if chronic. In acute infections the tissue reaction is purulent, with possible folliculitis. The fungi may be missed in H&E-stained sections, but they are readily identified in PAS or silver impregnation stains (Figure 20-6).
Candida are identified histologically by observing blastospores (yeast forms) in association with pseudohyphae. The blastospores are sharply defined, round, or oval structures, 3 to 4 microns in diameter; they may contain one or more vacuoles. Rarely, candidiasis may produce a granulomatous response, with pseudoepitheliomatous epidermal hyperplasia.
Differential Diagnosis
Contact dermatitis may be readily confused with C. dermatitis. Those conditions are easily separated by demonstrating the causative organisms histochemically.
CAPSULE SUMMARY
CANDIDIASIS
Candidiasis is a spectrum of superficial fungal infections caused by several Candida species. It is the most common mucocutaneous fungal disorder in children and presents with a rash that varies in appearance with patient age and anatomic site; it is almost always pruritic. Causative organisms can be labeled with the PAS method, and their presence separates candidiasis from its differential diagnostic alternatives histologically.
PITYROSPORUM FOLLICULITIS
Definition and Epidemiology
Pityrosporum folliculitis (PF) is inflammatory folliculitis of the chest and upper back of young adults due to infection with Malassezia organisms. PF has a worldwide occurrence, with the highest prevalence being in countries with a warm humid climate. It is seen commonly in adolescents and young adults.
Etiology
PF is caused by Malassezia furfur, a lipophilic yeast that was previously known as Pityrosporum ovale or P. orbiculare. Malassezia organisms are normal commensals in healthy people, but sebaceous gland activity during puberty may cause plugging of follicular ostia and facilitate colonization by the fungi. Malassezia can hydrolyze triglycerides into free fatty acids; this results in a cell-mediated response and activation of the alternate complement pathway with consequent inflammation.
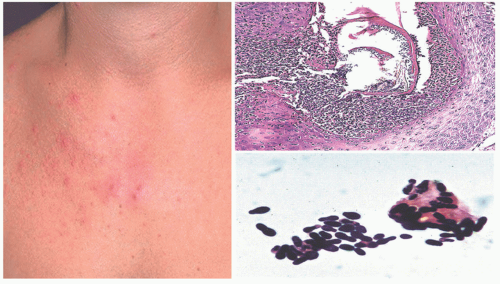
Clinical Presentation
Multiple discrete 2 to 4 mm papules and pustules are seen, which are pruritic, erythematous, and monomorphic. Some may be hyperpigmented. PF favors the chest, upper back, and upper arms; facial involvement is uncommon. The lesions have
a definite follicular pattern, which is best appreciated with the aid of a dermoscope. An absence of comedones helps rule out acne. Many patients have coexisting seborrheic dermatitis.
Histologic Findings
The histopathologic features are those of acute folliculitis and perifolliculitis with dilatation of the hair follicles and formation of keratin plugs. Rupture of the follicles may produce dermal abscesses. The fungi form colonies of yeasts within the follicles and dermal abscesses, surrounded by basophilic debris and neutrophils. The spores often have single buds and measure between 2 and 4 microns; they may be seen in H&E-stained sections, but are best demonstrated with the tissue Gram, PAS, and GMS methods (Figure 20-7).
Differential Diagnosis
The histologic differential diagnosis principally centers on bacterial folliculitis. Tissue Gram, PAS, and GMS stains will allow for a definite diagnosis of PF.
CAPSULE SUMMARY
PITYROSPORUM FOLLICULITIS
PF is folliculitis on the upper torso of children and young adults that is caused by infection with Malassezia species. Skin lesions are papulopustules that are pruritic and erythematous. Histopathologically, acute folliculitis and parafolliculitis are seen with dilation of hair follicles and follicular keratin plugs. Causative organisms can be labeled with tissue Gram, PAS, and GMS methods.
PIEDRA (TINEA NODOSA, TRICHOMYCOSIS NODULARIS, BEIGEL DISEASE, BLACK AND PIEDRA)
Definition and Epidemiology
Piedra is a fungal infection of the hair shafts, characterized by a hardening of the shafts and the development of nodules. There are two types—black piedra and white piedra. White piedra is more common in temperate and semitropical climates, such as Asia, Europe, Japan, and parts of the southern United States, whereas black piedra predominates in South America, East Asia, the Pacific islands, and sub-Saharan Africa. Both forms of piedra could conceivably exist in the same individual. Some reports have suggested a higher prevalence in females. Piedra can occur at any age; the incidence is greatest in young adults, but several reports have also been made of the disease in both preschool and school-age children.
Etiology
Black piedra is caused by infection with Piedraia hortae which is found in soil, stagnant water, and some vegetables. On the other hand, white piedra is caused by Trichosporon asahii, which is present in soil, water, sputum, and vegetables. Although the mode of infection in humans is not clear, the fungi invade hair shafts after colonization of the skin.
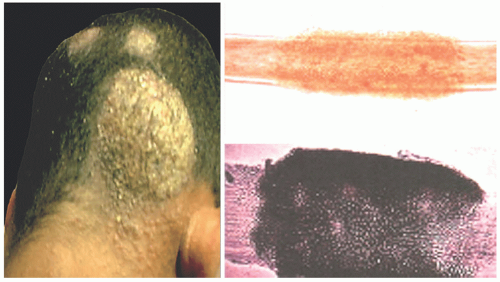
Clinical Presentation
Black piedra is typified by firm, darkly pigmented nodules of variable sizes, especially on scalp hairs. The facial and pubic hair may also be affected. White piedra is characterized by lightly pigmented nodules that are soft in consistency, on hair shafts. It affects facial, axillary, and pubic hair, as well as the eyelashes and eyebrows. When white piedra is seen on the scalp, the disease may be extensive. Hair breakage occurs in both forms of piedra, but the surrounding skin is healthy.
Histologic Findings
There are large nodules along the hair shafts in both forms of piedra (Figure 20-8). Microscopic examination shows that they comprise masses of mycelia (loosely aggregated hyphae) as well as numerous ascospores. The latter forms are dematiaceous and can be labeled with the Fontana-Masson method. Transverse sections of the hair shafts show that they are surrounded by multiple arthrospores with a tendency for linear arrangement.
Differential Diagnosis
There is essentially no differential diagnosis, providing that direct microscopic examination of hairs is performed.
CAPSULE SUMMARY
PIEDRA
Piedra is a fungal infection of the hair shafts, caused by Piedraia hortae and Trichosporon asahii causing black piedra and white piedra, respectively, referring to the formation of dark or lightly pigmented nodules on the hair shafts as seen microscopically. Arthrospores surrounding the hair shafts can be labeled with the PAS or GMS methods, and those in black piedra are also Fontana-Masson-positive.
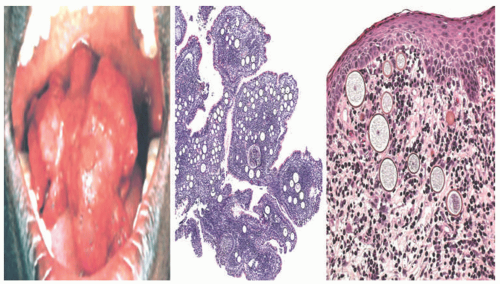
RHINOSPORIDIOSIS
Definition and Epidemiology
Rhinosporidiosis is (RS) a chronic granulomatous infection of the mucous membranes caused by the fungus Rhinosporidium seeberi. It is characterized by the growth of friable polyps on affected mucosal surfaces. RS is endemic in parts of India, Sri Lanka, South America, and Africa. However, it has also been reported in parts of North America. Males are most often affected, and the disease can be seen in either children or adults.
Etiology
Inoculation of R. seeberi occurs with trauma to mucosal surfaces. Local replication of the organism leads to hyperplasia of host tissue and a localized cell-mediated immune response. The commonest sites for RS are the nose, nasopharynx, and palpebral conjunctiva. Other mucosal areas are affected much more uncommonly, and dissemination to other organs is very rare.
RS is sporadic; although some authors have related the infection to contact with infected water, others have suggested a causal exposure to dust. Males are predominantly affected, and most patients are 13 to 40 years old.
Clinical Presentation
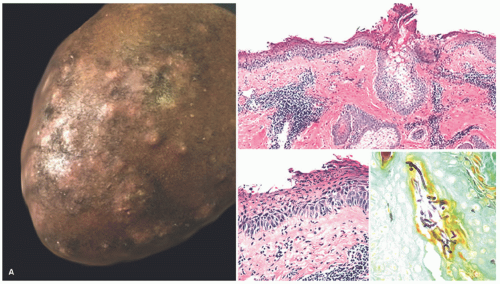
Symptoms of upper respiratory tract infection predate the development of soft polyps with a strawberry-like appearance, typically on the mucosal surfaces of the nose or eyes. The polyps are usually vascular and friable, and they bleed easily.
Histologic Findings
The skin lesions begin as papillomas that become more verrucoid with time and are associated with mucoid surface material. The surface epithelium and submucosa contain numerous globoid cysts that range from 10 to 200 microns in diameter (Figure 20-9). Some may be partially collapsed, assuming a semilunar shape. Spores are released from mature sporangia by rupture of their walls. They develop into small trophic cysts that contain a basophilic karyosome and distinct chitinous walls. H&E-stained sections clearly demonstrate the organisms, and they are stained by the PAS and GMS methods. The inner portion of the walls of larger sporangia may be birefringent.
Differential Diagnosis
The outer walls of the spores and the inner walls of the sporangia are well stained by Mayer’s mucicarmine technique, but R. seeberi shows no morphologic resemblance to Cryptococcus neoformans. Occasionally, small collapsed cysts may contain degenerating small trophic forms that resemble empty spheroids of Coccidioides immitis. Finally, R. seeberi somewhat resembles Emmonsia crescens in size and appearance, but only the former of those organisms is labeled by mucicarmine.
CAPSULE SUMMARY
RHINOSPORIDIOSIS
RS is caused by infection of the mucous membranes of the eyes, nose, and oropharynx by R. seeberi and predominantly seen in India, Sri Lanka, South America, and Africa. Multiple soft polyps appear on the affected mucous membranes that are friable and bleed easily. Verrucoid papillomas are seen histologically, containing globoid cysts that represent sporangia with internal spores. These can be labeled with the PAS and GMS methods but are also easily seen in H&E stains.
Deep Mycoses
The “deep” mycoses are secondary cutaneous manifestations of fungal infections of the viscera. Many but not all use the lungs as the portal of entry.
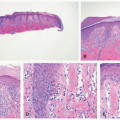
CRYPTOCOCCOSIS
Definition and Epidemiology
Cryptococcosis of the skin is caused by a dimorphous encapsulated yeast, Cryptococcus neoformans. It also has a predilection for growth in the lungs, brain, and meninges. Cutaneous involvement is seen in 10% to 15% of cases. C. neoformans has an international distribution. Spores of that fungal organism have been recovered from avian excreta, soil, and dust.
Etiology
C. neoformans has two variants—var. neoformans (serotypes A, D, and AD) and var. gattii (serotypes B and C). Neoformans is found most often in Europe and North America, particularly in HIV-infected patients. The gattii form is more prevalent in Africa. Both of the organismal variants are carried by airborne particulates that are inhaled. Localized lung disease is the most common, although systemic manifestations are possible depending on the host response, the size of the inoculum, and the virulence of the organism. Disseminated disease is usually seen in immunocompromised individuals. It occurs via hematogenous spread, leading to multiple organ involvement that can include the skin. Much less commonly, direct inoculation of C. neoformans into the skin may result in primary cutaneous disease.
Clinical Presentation
The rare primary lesions of cryptococcosis in the skin are usually single, predominantly situated on the extremities, and are accompanied by regional lymphadenopathy. They may manifest as ulcers, cellulitis, abscesses, or a sporotrichoid pattern of dermal involvement. Secondary skin involvement is the norm; it is associated with systemic (particularly pulmonary) symptoms and may affect any skin area. The lesions are initially papular, but then become pustular or ulcerated. Acneiform papules or pustules, warty or vegetating plaques and ulcers, hard infiltrated plaques or nodules, subcutaneous masses, abscesses, blisters, eczematous plaques, and vasculitis-like lesions are also possible.
Histologic Findings
In H&E-stained sections, Cryptococcus is usually relatively easy to identify. The organisms measure 4 to 7 microns in diameter, and they stain light blue. Their mucinous capsules appear as clear halos. Mucicarmine and colloidal-iron staining labels the capsular material (Figure 20-10), and the organisms may also be positive with the Masson-Fontana technique because they contain melanin precursors (Figure 20-11). The latter feature distinguishes C. neoformans from other yeasts. The surrounding tissue contains a mixture of acute and chronic inflammation, with variable formation of true granulomas. In overtly granulomatous lesions, it may be more difficult to identify the organisms, and histochemical methods will figure more prominently into diagnosis.
Differential Diagnosis
Small forms of C. neoformans may resemble Histoplasma, and, on the other hand, large forms without capsules can mimic Blastomyces. The thick cell walls and multiple nuclei of Blastomyces dermatitidis distinguish it from C. neoformans. Rhinosporidium is regularly carminophilic, but its morphologic appearance is unlike that of Cryptococcus.
CAPSULE SUMMARY
CRYPTOCOCCOSIS
Cryptococcosis is caused by infection with Cryptococcus neoformans presenting with skin lesions that are typically secondary manifestations of distant spread from a primary pulmonary infection, usually in immunocompromised host. Lesions are popular, pustular, or ulcerated with possible progression to plaques, nodules, and vasculitis-like lesions. Yeast forms in the dermis are surrounded by clear halos with a neutrophilic and granulomatous inflammatory response. The organisms can be labeled with the PAS, colloidal iron, and Fontana-Masson technique.
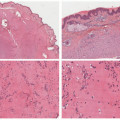
BLASTOMYCOSIS
Definition and Epidemiology
Blastomycosis is a chronic granulomatous and suppurative deep mycosis, caused by infection with B. dermatitidis. This disease is seen globally, but it is commonest in northern parts of the United States and in sub-Saharan Africa. Blastomycosis is most prevalent in adult men, but it can be seen in children as well.