Key Words
Breast autologous reconstruction, SIEA flap, microsurgery
Introduction
The superficial inferior epigastric artery (SIEA) flap relies on its namesake vessels to perfuse the ipsilateral abdomen skin and fat, generally for autologous breast reconstruction. This type A fasciocutaneous flap was described in 1971 for head and neck reconstruction by Antia and Buch. Grotting introduced the SIEA for use in breast reconstruction in 1991. The advantage of this flap is the lack of fascial and muscular dissection. The SIEA sits in a suprafascial plane, eliminating the kind of donor morbidity seen in the deep inferior epigastric artery perforator (DIEP) or the muscle-sparing transverse abdominis muscle (MS-TRAM) flap.
Despite this, the SIEA flap failed to gain widespread acceptance due to difficulties in harvest and inset, vessel inconsistencies, decreased pedicle size and length, and tendency towards spasm. Historical high rates of take backs (14%–21%), arterial thrombosis (17.4%), short pedicle length, and flap failure (7%–14%) have led some to disavow this flap completely. As such, flaps based on the deep inferior epigastric artery (DIEA) have become the gold standard in autologous-based breast reconstruction.
With the refinement of microsurgical technique and understanding of the SIEA anatomy, the SIEA flap has enjoyed a resurgence, allowing surgeons to take advantage of the inherent donor-site advantages of this flap and the lack of fascial disturbance. Seminal cadaveric work by Taylor and Daniels traditionally indicated that the vessels are present in 65% of patients. Nahai demonstrated in a series of 27 successful SIEA flaps that the vessels are absent in as few as 13%. The senior author described a series of 145 SIEA flaps with a 6% take-back rate and a loss rate of 4.8%, comparable with other breast free flaps. In this series, 500 abdominal-based breast free flaps were performed, 29% of which were SIEA flaps. Accordingly, when anatomically appropriate, the SIEA flap has become a mainstay in our practice and represents about 30% of our flap reconstruction volume, similar to other experienced microsurgeons. In this chapter, we will demonstrate how a SIEA flap can be selected and performed safely, and our strategies to increase success are also delineated in detail.
Indications and Contraindications
The SIEA flap shares the same indications as its DIEP or MS-TRAM counterparts. The vast majority of these cases are performed in breast reconstruction after mastectomy. Other indications include reconstruction in Poland syndrome, unsatisfactory implant reconstruction, or loss of volume after lumpectomy. Other applications, such as use in the head and neck, have been described and are beyond the scope of this chapter.
Our algorithm for superficial inferior epigastric (SIE) vessel selection is based intraoperatively and is as follows:
- 1.
Venous size of 1.5 mm
- 2.
Palpable arterial pulse
- 3.
Pedicle length at least twice the flap size for inset
Relative contraindications to the SIEA flap include prior abdominoplasty, Pfannenstiel incision, or liposuction. Liposuction cannulas can damage the fragile SIEA vessels easily due to their suprafascial nature. In the setting of prior abdominal surgery, careful intraoperative assessment of the SIE vessels is crucial. Successful harvest can be achieved if they are not damaged (see Case 4.1 ).
Other relative contraindications include inadequate flap volume and the lack of an IMA recipient vessel. The use of the SIEA in the unilateral reconstruction is limited due to its ability to only supply a hemiabdomen. If a patient still desires the decreased donor morbidity of an SIEA flap and requires volume, stacking flaps for unilateral reconstruction or discussion of multiple rounds of fat grafting may be required. The SIEA provides 4–7 cm of pedicle length. Due to this shorter length, thoracodorsals or thoracoacromials are generally not used as recipient vessels.
Preoperative Evaluation and Special Considerations
In preparation for surgery, a detailed history and physical examination are performed. It is of utmost importance to obtain a history of prior surgeries, medications, familial diseases and nicotine usage. Most flap patients are generally healthy. Mastectomy and autologous reconstruction may be their first major surgery and stress on their bodies. However, active cancer, use of oral contraceptives, long surgical times, decreased venous return after abdominal plication, and family history of clotting disorders all increase the risk of deep venous thrombosis (DVT) and subsequent pulmonary embolism. The Caprini Risk Assessment score is routinely calculated and documented in the patient’s preoperative chart.
Physical examination entails examining for prior surgical scars and availability of abdominal tissue. High abdomen scars, such as the Kocher, do not preclude the possibility of abdominal-based flaps. Full disclosure of the possible donor-site wound healing complications is necessary. Abdominal tissue excess may be assessed by pinching the infraumbilical skin and fat or performing the classic Diver’s test.
While we do not routinely do so, computer tomography angiography (CTA) from the diaphragm to the lesser trochanter can be used for preoperative assessment of vasculature including the SIE vessels prior to free transfer. Generally, SIEA size is measured at the inguinal ligament; DIEP vessels are measured at the take off of the external iliac artery. SIE vessels of adequate caliber for flap reconstructions are 1.5–2.3 mm on preoperative imaging. Despite this, only about half of vessels found adequate on CTA are usable after intraoperative assessment. Intraoperative assessment is still the gold standard.
Surgical Techniques
Relevant Anatomy
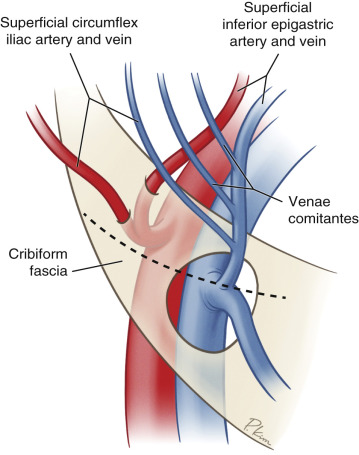
The SIEA originates 2–3 cm below the inguinal ligament: directly from the common femoral artery in 17%, as a shared branch with the superficial circumflex iliac artery (SCIA) in 48% of cases, and absent or hypoplastic in 35% of cases ( Fig. 4.1 ). Recent cadaveric and imaging studies have reliably identified vessels in 65%–75% of cases. At its origin, the SIEA is generally is 1.6 mm, compared to 3.4 mm with the DIEA at the external iliac artery just proximal to the inguinal ligament. A general rule of thumb is that the SIEV arises in the junction between the medial and middle third of a line from the pubis symphysis to the anterior superior iliac spine (ASIS), approximately 4–5 cm from midline. The SIEA arises 3 cm more laterally, and deep to Scarpa’s fascia.
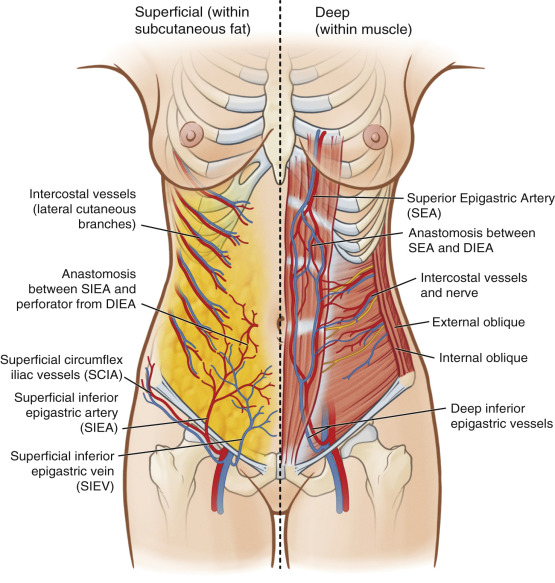
After piercing the deep fascia, the artery travels deep to the cribriform fascia in the superolateral portion of the femoral triangle, piercing the fascia to cross the inguinal ligament at its midway point lying deep to Scarpa’s fascia. Well above the inguinal ligament, the vessel penetrates Scarpa’s fascia to lie in the superficial subcutaneous tissue. Here it runs superomedially toward the periumbilical region progressively more superficial to anastomose with the ipsilateral medial and lateral perforating arteries from the DIEA through the subdermal plexus. Probably due to its lateral nature, perfusion studies have indicated the SIEA acts similarly to lateral row DIEP vessels, without blood flow crossing the midline. In patients with adequate flow and size of the SIEA, the deep and superficial inferior epigastric systems share common vascular channels that can interchangeably supply the lower abdominal skin, providing the anatomical basis of the SIEA flap.
The SIEA often have two closely associated diminutive venous comitantes. These veins are small in caliber and are not often used. They may be used, however, if they are large enough compared to the superficial inferior epigastric vein (SIEV). The SIEV drains into the saphenofemoral bulb before meeting the femoral vein or into the greater saphenous vein as a tributary. Anastomotic ladder-like connections exist between this superficial system and perforating veins from the deep inferior epigastric veins (DIEVs). In the physiologic state, the SIEV has been shown to be the preferential drainage path of the abdominal pannus. In 40% of patients, medial and lateral branches of the SIEV drain variably into different vessels. Both branches, if present, need to be preserved and dissected for adequate venous drainage as many flaps lack overlapping venous territory drainage between branches. If a single venous anastomosis is preferred, functional testing for venous congestion is paramount.
Preoperative Markings
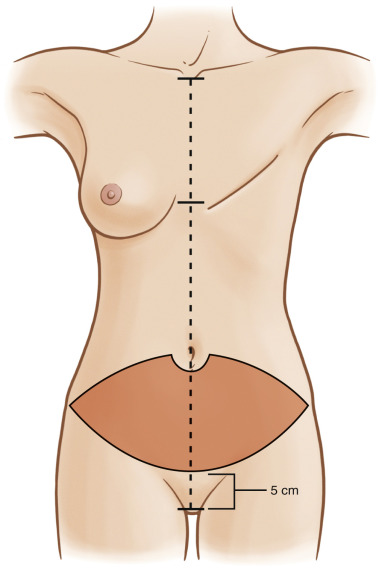
Markings are performed in the preoperative surgical area. The patient’s midline is marked from the sternal notch to the pubic tubercle, including horizontal midline marks at the inframammary fold ( Fig. 4.2 ). Our preoperative markings for all abdominal-based flaps are lower than the traditional DIEP flaps; we do not include the skin bridge immediately cephalad to the umbilicus. Instead we begin our incision half way at the level of and around the umbilicus. The caudal incision is approximately 5–6 cm above the pubis symphysis. The lower incision increases the chance of capturing the point when all the venae comitantes converge, thereby including the more lateral superficial circumflex iliac veins into the flap ( Fig. 4.3 ). This also allows us to visualize and effectively use the SIEA prior to it arborizing into smaller tributary arterioles. We mark this flap with this method in all abdominal-based breast reconstruction for this reason. Periumbilical perforators are preserved with careful technique.
Flap Dissection
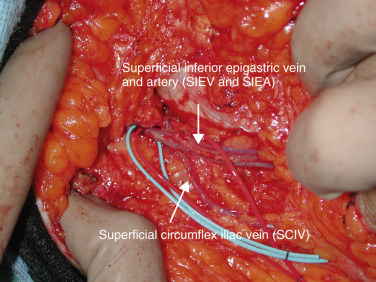
Dissection begins at the lower incision in the lateral to medial direction. This incision should be made judiciously, as in a thin patient the SIEV may lay superficial. Using loupe magnification dissection, the SCIA/SCIV is first encountered. The SCIV is included in the dissection to improve venous outflow whenever possible (see Fig. 4.3 ). The SIEA is next encountered at the midpoint of the inguinal ligament, generally deep to Scarpa’s fascia. We avoid circumferential dissection as this can lead to arterial spasm. The SIEV is identified in a more medial and superficial position in the subcutaneous tissue 2–3 cm medial to the SIEA at a point 4–5 cm lateral from the midline. All branches of the SIEV are included and followed. The vessels are followed for a few centimeters by having an assistant provide gentle cephalad retraction of the flap and superior retraction of the groin skin. The SIEA is dissected in the inferomedial direction until the fascial hiatus is encountered. Fully opening the hiatus at the cribriform fascia affords for maximal exposure ( Fig. 4.4 ).
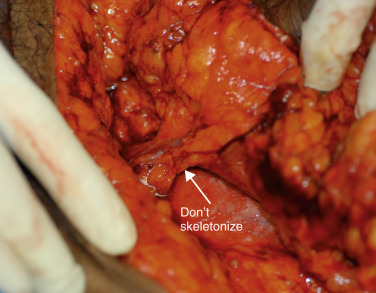
The SIEA and SIEV are dissected to the level of the first appearing lymph node. The lymph node, as noted by Buchel et al., is frequently where the artery and vein separate. Minimizing trauma to the lymphatic channel is key in this area to decrease seroma rates, commonly seen as high as 20% in SIEA flaps (3.4% in our series). However, if nodes are within the bundle, it is counterproductive to attempt to skeletonize and extract this node ( Fig. 4.5 ), as this leads to unnecessary risk, spasm, and possibly irreversible trauma. The artery at this level makes a sharp 90–150 degree turn toward the femoral artery.
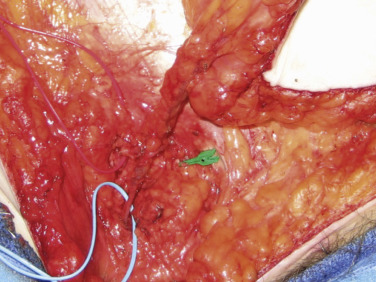
The vein is also followed and will join other comitantes before entering the cribriform fascia. Further dissection at this point will identify the confluence of vessels between the SIEV, SCIV, and other venae comitantes at the saphenofemoral bulb ( Fig. 4.6 ). This single dominant vein is usable for anastomosis providing excellent outflow for the flap. Some authors have cited this bulb to be too large for coupling; we have found clipping the bulb before it excessively dilates effectively controls sizes for anastomosis. The inclusion of all veins is the physiologic basis of our lower, caudal incision.
Our decision to commit to the SIEA flap is based on a simple intraoperative algorithm alluded to above. First we palpate the artery to assess flow characteristics. A strong palpable pulse heralds a large enough diameter for inflow. We do not use size as a measure for the artery and have performed <1-mm anastomosis with successful flaps. Second, we assess the SIEV, and require a 1.5-mm vessel for outflow. Veins of this caliber suggest dominance over the deep system and facilitate coupling. Finally, we generally require pedicle length at least twice the thickness of the flap for insetting purposes, although this is not a hard and fast rule. Careful examination of both sides is necessary if bilateral SIEAs are to be pursued as caliber may not match. Once committed to the SIEA flap, superior and midline incisions are made and the flap is raised off the anterior rectus sheath. The vessels are freed posteriorly, the artery is clipped flush to the femoral artery for maximum diameter, and the vein (or veins) is clipped at a point for sound venous coupling. To decrease traction/spasm issues, we routinely de-epithelialize the flap in situ .
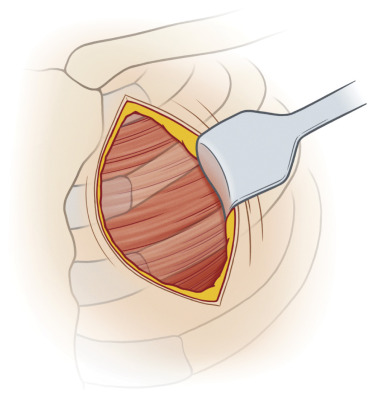
Rib-Sparing Internal Mammary Harvest
Our preferred recipient vessels are the internal mammary artery (IMA) or IMA perforators through a rib-sparing approach. Conventional approaches to the IMA for harvest require removing a costal cartilage from the third or fourth rib. This invasive approach can be associated with intercostal neuralgia and chest wall deformity. A prerequisite to the rib-sparing technique is an interspace that is two fingerbreadths wide, generally at the second or third intercostal space ( Fig. 4.7 ).