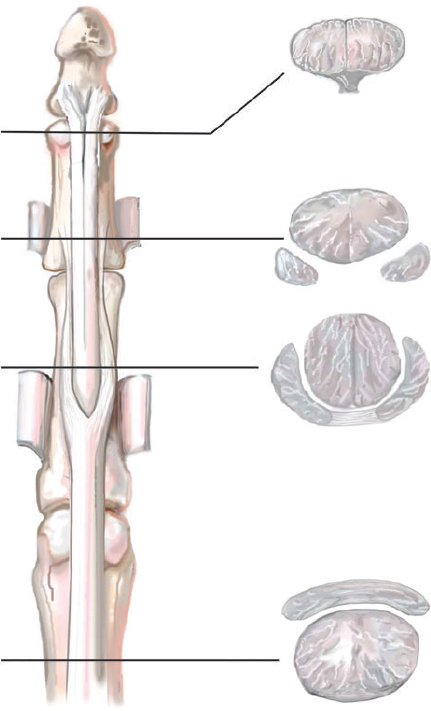
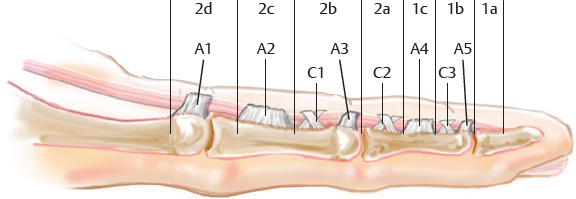
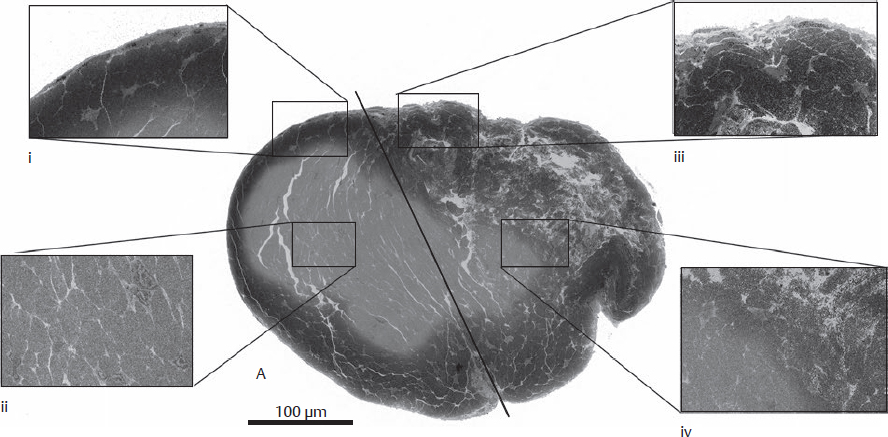
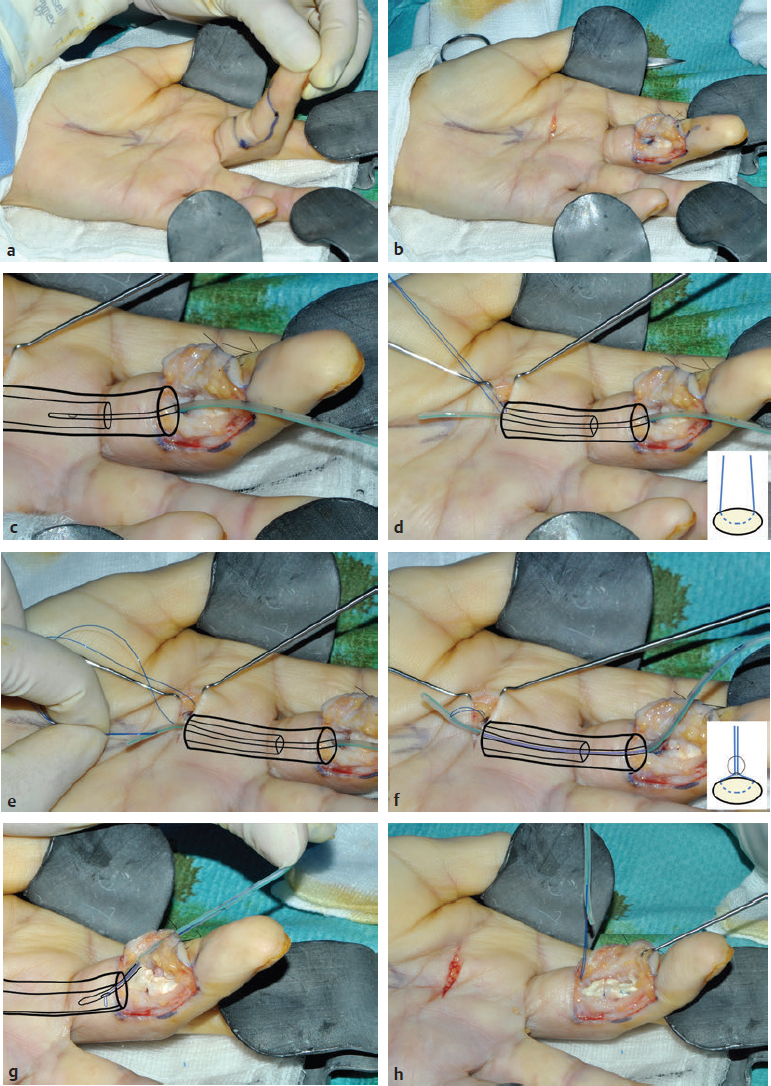
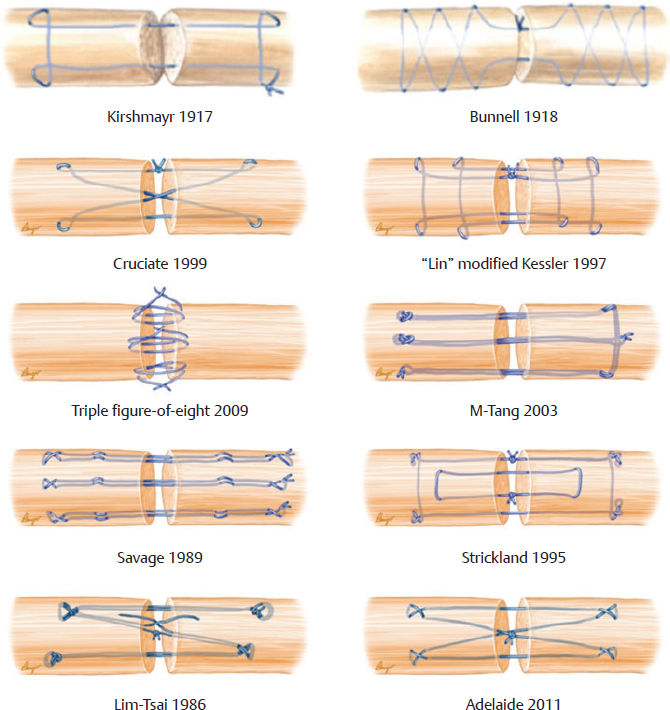
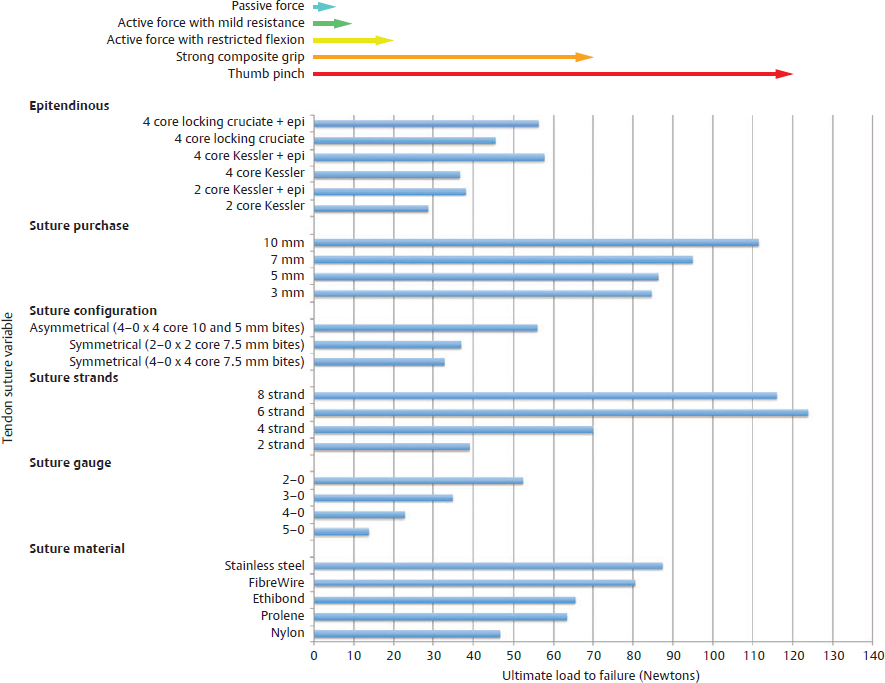
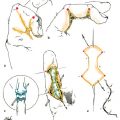
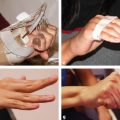
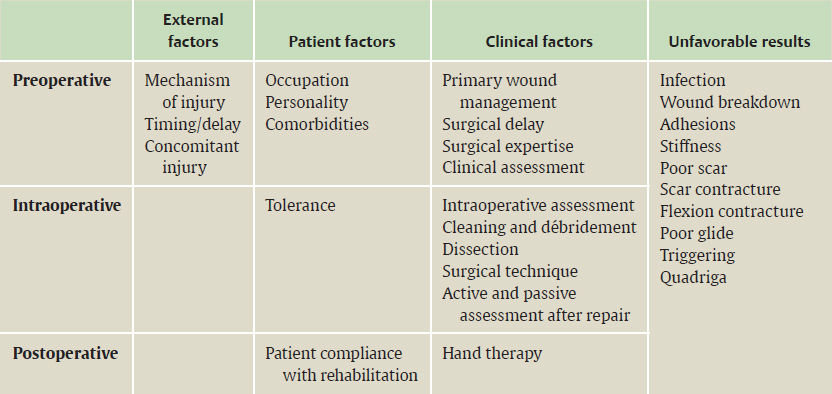
CHAPTER Flexor tendon surgery is a challenging area that has seen modest progress over the years. Comparing the 1977 series by Lister et al1 of two-strand repairs and simple circumferential epitendinous sutures, combined with the Kleinert rehabilitation regimens,1 with the 2011 data from Sandow et al2 on four-strand repairs with early active mobilization regimens reveals the rupture rates and functional results to be almost identical. Our management has completely changed, built on new scientific foundations, but our outcomes have not changed dramatically. Around 25% of patients still have poor to fair outcomes with an approximate 5% rupture rate, even in the best centers. Surgeons seem to have accepted this situation in hand surgery for trauma, whereas it is unlikely to be acceptable in elective surgery. With this range of outcomes, much more needs to be known about why the results are disappointing. Over the years, we have established that failures are related to both the mechanics and the cell biology of tendon repair. The challenge has been to achieve a strong repair in a biologically active structure that is able to withstand friction-free tensile load at the earliest possible time point. It is also important to appreciate that failure occurs in the context of an injured, inflamed, confined environment. Once this concept is understood, it is possible to understand how to avoid unfavorable results. Restoration of functional outcome is a fine balance between strength of the repair to prevent rupture and mobilization to prevent adhesions. Adhesions is a clinical description of loss of range of motion that can be related to true adhesion formation between the tendon and sheath or scarring and fibrosis of the surrounding structures. Early mobilization with good functional glide is designed to prevent adhesions and stiffness, but mobilization carries a risk of rupture. Paradoxically, gapping, elongation, and attenuation may also promote adhesion because of a reduction of glide, secondary healing to the surrounding tissues, and fibrosis between tendons. It is essential to understand the causes of failure to avoid an unfavorable result. This requires a root cause analysis of every case that deviates from a satisfactory outcome and close cooperation between the surgeon, hand therapist, and patient to ensure the best results can be achieved. Summary Box Causes of Unfavorable Results Tendon is an extremely complex tissue that is rarely fully appreciated in anatomical and surgical texts. Given the ubiquitous nature of tendon injuries and pathologies, it is surprising how little attention tendon has received in medical training. Few surgeons realize that tendon has precisely defined areas of anatomical specialization that include vascularity, elasticity, and fibrocartilaginous differentiation, all of which influence how the tendons heal at different points along its length. There are multiple vincula that enter the dorsal aspect of the flexor tendons in the zone II region, which are surgically important for a variety of reasons: • Preventing the tendon from retraction when the tendon is divided • Providing nutrition to the tendon as it heals • Bleeding into the sheath once the tourniquet is released, acting as a source of clot and subsequent adhesion formation In “no-man’s-land” (zone II) of the flexor tendon sheath, the flexor digitorum profundus (FDP) has surface areas of fibrocartilage that resist compression from the flexor digitorum superficialis (FDS) as this tendon splits to surround the FDP between the bifurcation before rejoining at the Camper chiasm (Fig. 61.1). This careful arrangement of the pulleys allows the FDS to engage and lock the FDP in the hook grip. Fibrocartilaginous differentiation is also found at the insertion/entheses of the FDP and in the region where the pul leys repeatedly come into contact with the tendon. These structures are constantly changing their cross-sectional profile as they glide; hence it is easy to appreciate that any disruption to the anatomy will result in functional impairment (see Fig. 61.1). Tendon injuries by convention have been subdivided into zones originally proposed by Verdan,3 although the junctions between them are not precise. Zone I, for example, is defined as distal to the FDS insertion, and, as it extends all along the middle phalanx, it incorporates many different situations proximal, through, or distal to the A4 pulley. Tang4 realized that zone II could be better subdivided in to zone IIa, IIb, IIc, and IId to highlight the complexities of repair in this zone5,6 (Fig. 61.2). However, no classification system characterizes the effect of whether injuries of the tendon are in flexion or extension, or in tension or relaxation, and this ultimately dictates the degree of retraction through the sheath. Tendon acquires its tensile strength by having a hierarchal arrangement of collagen extending from its ultrastructure through to its macroscopic anatomy6–9 (Fig. 61.3). Collagen has a triple helical arrangement, grouped as periodically staggered fibrils to form fibers, which are then packaged as bundles or fascicles, interspersed with proteoglycan molecules, which have a high water affinity.6 This arrangement is necessary to allow the tendon to flex around corners, deform, and stretch, which can only happen if the individual molecules and fibrils can slide over one another. Critical to tendons’ longitudinal morphology is the epitenon covering, which is a biologically active single layer of epithelial-like cells and meshed collagen, which contains the internal fibers to give the tendon a tubular form.10 This epitenon in intrasynovial tendon is especially important to appreciate, because it is a layer in which sutures place a hold in the tendon substance; this explains the different suture-holding capacities of extrasynovial tendon and intrasynovial tendon. Fig. 61.1 Cross section of the flexor tendon in the digit and the flexor digitorum profundus and flexor digitorum superficialis arrangement. Fig. 61.2 Flexor tendon subzones of the digit. Subzones of injury in the hand are modified from the Tang subclassification of zone II and Moieman and Elliot subclassification of zone I. Image adapted from Langer illustration. (Modified from Langer MF, Oeckenpöhler S, Hartensuer R, et al. Ringbandrekonstruktion an der Hand. Der Orthopäde 2015;44(10):757–766.) The current teaching of flexor tendon healing that refers to extrinsic and intrinsic pathways gives the misconception that these processes occur independently. The concepts of intrinsic and extrinsic healing were borne from experimental studies that showed tendons needed external vascularization through adhesions to heal (extrinsic) but also could heal in an avascular nutrient-rich environment (experimentally in a knee joint) without external factors (intrinsic). The current paradigm acknowledges that these complex cellular processes occur together.11 The tendon, sheath, subcutaneous tissue, and skin are all injured in continuity through actual trauma or surgery. They therefore heal in an overlapping fashion and if immobilized result in a continuous mass of scar tissue. The process involves the classic phases of wound healing, including coagulation, inflammation, synthesis, and remodeling; however, the different tissues heal at different rates. The peak of skin and subcutaneous tissue collagen synthesis occurs around 1 week, whereas tendon generates the most collagen at around 3 weeks and therefore gradually increases in strength. Protection from full active loading is required for 10 to 12 weeks. Normal tissue boundaries are breached and fibrin clot between the tendon and sheath are infiltrated by fibroblasts at around day 5, leading to new collagen deposited as fibrous adhesions. To some extent this is unavoidable, and the role of therapy and motion is to disrupt or lengthen any adhesions that form. Like all wound-healing processes, there will be individual variations in the tendency to fibrosis. Prediction of those at most risk remains elusive, but considering parallels with scar patients, we can assume that individual patient phenotypes, mechanical forces, and inflammation are likely to play a key role. In addition to this already complex biology, sutures grasping the tendon ends create areas of high strain, resulting in acellular tendon zones. We have found that a suture grasping the tendon leads to tenocyte necrosis and prolongs inflammation that can lead to increased adhesion formation12 (Fig. 61.4). The tendon healing process involves a significant recruitment of cells throughout the various phases, which is perceived as tissue swelling. In the early phases of repair the tendon actually softens because of matrix enzymes liberated by the injury process and suturing. The increase of strength above baseline in the tendon repair is only seen after 2 weeks; up to this time it is reliant on the suture strength alone.13 From this point on tendons gain their structural organization and strength by tensile load; hence active motion with some loading is a beneficial factor in the repair process while collagen is being deposited. A careful balance of rehabilitation with the biology is required by allowing motion, with slowly increasing load over the course of healing. Fig. 61.3 Ultrastructure and microstructure of a partially damaged mouse flexor tendon. (a) Transmission electron microscopy cross section of a mouse flexor digitorum profundus tendon. (i) Magnified view of undamaged epitenon, seen one cell layer thick. (ii) Magnified view of undamaged endotenon and collagen fibers made up of fibrils. (iii) Damaged tendon epitenon showing proliferation of the surface cells. (iv) Damaged endotenon, showing loss of organized bundle and fiber architecture. (This image courtesy of Yinhui Lu and Karl Kadler, whose work is funded by the Wellcome Trust.) Fig. 61.4 Cell mapping of a single suture in a tendon (gold) with resultant acellular zone formation (blue) and inflammatory cell influx (red). Ex vivo biomechanical studies have shown that the strength of repair is influenced by the number of core strands, suture caliber, purchase length, and suture material.14 However, it is perplexing to find that meta-analyses of rupture rates worldwide could not identify significant differences between two-strand and multistrand repairs in clinical studies,15 and complication rates over the last 30 years have remained static. Our surgical attempts to join severed ends with multiple strands of suture are simplistic, as attempting to join analogous engineering structures, such as cables, end-to-end is destined to fail. A suture-based repair experimentally can resist load forces in the region of 40 to 80 N, although gapping usually occurs at forces around 10 N, which falls far short compared with normal tendon strength (usually greater than 150 N). The amount of force transmission permissible during active mobilization must function well within these values. However, the tensile strength achievable at the outset will not necessarily reflect the pattern over the next few days as the surrounding tissue swells, tendon softens, and glide characteristics change. The configuration of most suture repairs dramatically alters as soon as load is placed through the repair. Urbaniak16 showed a considerable loss of tensile strength 5 days after repair. This data is currently being overlooked in the search for an ever-stronger suture repair configuration, because what happens to suture tensile strength in clinical practice over time is unknown. The key point is that the immediate suture strength as determined from current cadaver or animal tendon studies is very different to the in vivo strength at 5 days. More studies to determine the dynamic accrual of tensile strength after tendon repair are required to underpin a logical rehabilitation program of motion and loading. Rehabilitation programs need to institute motion to prevent adhesion but avoid any resistance until the tendon has regained functional tensile strength. Better techniques to investigate tendon loading noninvasively would be beneficial to tailoring rehabilitation regimens. Functional outcomes tend to fare better with early repair of flexor tendon injuries compared with delayed interventions. When tendons have retracted and become adherent, muscle fibers have been shown to shorten, and tendon sheaths collapse. It is not clear at what precise point delay will begin to have a negative effect on functional outcomes. We have found that delays beyond 7 days are subsequently related to a lesser total active range of motion.14 All tissues appear nearest to normal on the day of injury. Within hours there are profound biologic changes in both the wound and tendon, with edema and inflammatory infiltrate. Clinical services should ideally be organized to provide surgical repair within 24 hours to reduce the risk of infection from contamination. However, in reality this can be difficult to provide because of delays in referral and availability of acute services. Many tendon injuries are missed by inexperienced emergency department staff, such as closed FDP rupture related to avulsion from the distal phalanx or division of one of the two tendons in the digit. Early accurate diagnosis and treatment are the first line in avoiding unfavorable results, although on rare occasions a primary repair may still be successful even months after the injury if the divided tendon has not retracted. In our experience the longest time delay between tendon division and a primary repair has been 10 years (Fig. 61.5). Domestic injuries to the flexor tendons tend to be related to mishandling of sharp instruments, slips, and episodes of impaired judgment. Work-related injuries often arise from heavy or violent machinery like circular saws. These are associated with high energy transfer and tend to have more concomitant injuries, fractures, neurovascular injury, and skin loss. High-energy saw-type injuries that macerate the tendon ends result in poorer functional outcomes compared with clean-cut blade injuries, which tend to have a better outcome because of a more limited zone of trauma and lesser degree of inflammation.17 For untidy injuries, early intervention, adequate débridement, antibiotic therapy, and additional hand therapy are needed to prevent a poor outcome. Ensuring the injury is clean of debris, necrotic tissue, and environmental contamination minimizes the chance of infection, which is a serious early unfavorable outcome. Studies have shown that the typical demographic for tendon injuries of the hand is young, active adults who are relatively free of comorbidities; hence impairment of functional and working capacity is significant.18 Patients’ behavior and compliance can have a major effect on outcomes, so care must be taken to optimize the management of anxieties, impatience, and motivational aspects of recovery. Surgeons should be wary of a patient whose injury has been associated with drugs or alcohol. Curiously, failed suicide attempts give little indication of motivation, and yet many such patients work hard to reverse self-inflicted damage, and the rehabilitation effort often distracts them from their other life circumstances. Depending on the hand unit organization, some centers manage flexor tendon injuries during day time by consultant supervised operating lists. Alternatively, trauma may be managed outwith routine operating hours by less-experienced surgeons. Some studies have shown that outcomes are poorer with less-experienced surgeons, whereas others have failed to demonstrate a difference.19 It is clear that there is huge variation in how surgeons approach the problem, and it is important that within each center a surgical protocol matches the rehabilitation regimens. Group-wide training and evaluation of repairs in cadaveric or animal models is valuable for junior trainees and should be mandatory. However, nothing is better than supervised clinical experience to allow trainees to make judgments about management of skin incisions, the pulleys, and the associated damaged structures. The location of the tendon ends can be inferred from the history and mechanism of injury. A digit engaged in grasp at the time of injury is likely to have retracted, whereas a tendon relaxed at injury does not retract far from the wound. A meticulous examination commencing with observation of the normal cascade posture is also important in preoperative planning. Several passive maneuvers on the injured patient can provide an indication of which tendons are divided, such as a tenodesis test (on supination the wrist extends and digits flex if tendons are intact) and forearm compression test. Isolated testing of distal interphalangeal (DIP) joint and proximal interphalangeal (PIP) joint movement is also of value but is usually hampered by pain at the wound site. Anesthetic options for flexor tendon surgery are general anaesthesia (GA); regional anaesthesia (RA) (brachial plexus or regional nerve blockade, under ultrasound control); and the wide-awake local anaesthetic, no tourniquet technique (WALANT).20 GA remains a common method of choice for analgesia in upper limb surgery, because most anesthetists are familiar with its use. RA is also contraindicated when there is clear patient preference toward GA or if there is a local infection at the site of RA injection. RA has been shown to decrease postoperative nausea and vomiting, reduce early postoperative analgesic requirements, and result in less time spent in the hospital. Patients receiving RA ambulate sooner and commence normal oral intake quicker. RA requires a longer induction time, but the time in operating theater is no different from GA. If using a separate room to institute the RA blockade, the total anesthetic time can be reduced even further, thereby minimizing the delay between cases. The success rate of RA is dependent on experience and increases with ultrasound control, which also considerably reduces the complications of pneumothorax, hematoma, and postoperative pain from needle damage to the fascicles. Long-standing concerns about adrenaline use in local anesthetic ring blocks of the finger and hand have led to widespread avoidance out of fear of digital necrosis. This is still evident in current textbooks, medical teaching, and surgical practice. However, there is level 1 evidence demonstrating that local anesthetic with adrenaline can be safely used without the risk of digital infarction. The cases of digital necrosis in the 1950s may be attributed to expired batches of procaine before mandatory expiration dates were instituted. Procaine degraded into a harmful acidic solution, which, when injected, would lead to tissue necrosis by chemical damage.21 This has led to the development of wide-awake surgery using adrenaline to control bleeding without tourniquet control21 (Fig. 61.6). Wide-awake anesthesia has been described to be safe and effective for flexor tendon surgery in the hand with acceptably low rupture rates.20 Wide-awake primary tendon repairs enable the surgeon and therapist to assess range of motion and quality of the repair before skin closure. Inadequate repair (poor glide, triggering, gapping, or bunching) can be corrected immediately, and therapy regimens can be adjusted to any intraoperative findings. Although wide-awake surgery may have potential benefits financially and surgically, uptake has proven to be slow. The surgeon requires familiarity with local anesthesia in conscious patients and familiarity with tendon surgery to instill confidence in an anxious patient. Exposure of the tendon and other damaged structures is an important part of the overall outcomes in flexor tendon surgery (Box 61.1). Skin incisions should be carefully designed to incorporate the existing laceration and develop flaps that maintain good perfusion. Ensuring good primary wound healing reduces the incidence of scar contracture, another unfavorable result. Our starting point is usually to incorporate the wound with either a Bruner’s22 or midaxial incision, although variations of these incisions have been described. Generally there will be a traumatic wound, which requires extension. Before mapping out the incision line it is important to assess any associated digital vascular injuries, because basing skin flaps on divided vessels is likely to lead to skin necrosis. Flaps should be broad-based and, as advised by Bruner,22 there should be a linear extension from the wound before forming a zig-zag to prevent narrow-based flaps that lead to necrosis (Fig. 61.7). The planning of incisions requires the surgeon to weigh the surgical exposure for the repair against creating a larger wound with the attendant problems of postoperative swelling and later fibrosis. Large incisions for exposure will lead to increased swelling during the immediate postoperative period, which makes it harder to mobilize the digit and can affect the rate of recovery. Pitfalls for Incisions • Inadequate exposure on primary incision requiring further extension and a large final scar • A midlateral incision that is too far volar and therefore does not confer the safe sites for incision, placing the digital neurovascular bundle at risk • Distal incisions that compromise flap viability beyond the trifurcation of digital vessels • Skin flaps that are too thin, resulting in necrotic flaps, suture pullout, or wound dehiscence • Crossing the skin crease with the incision as a perpendicular line Steps to Avoid These Pitfalls • Appropriate clinical assessment and history to anticipate the position of the distal and proximal stump in relation to the site of skin injury • Planning of the distal incision to allow adequate purchase and pulley venting at the expected distal stump site • Incisions distal to the digital vessel trifurcation planned for the midline to preserve skin flap vascularity • Skin flaps taken with fat in full thickness down to the tendon sheath to preserve vascularity Outcomes can be impaired by excessive dissection just to find the ends. The surgeon should be able to judge where the tendon ends lie and then plan the incisions, exposure, and management of associated fractures, neurovascular injuries, or need for skin cover. Fig. 61.8 Flexor tendon retrieval. (a) Exposure planned over the zone of injury extending distally and a separate incision at the distal palmar crease. (b) The incisions made preserve skin over zone II. (c) A 6-Fr pediatric feeding tube is passed distal to proximal down the flexor tendon sheath. (d) The tube is pulled through into the proximal wound, and a big grasping stitch with 3–0 Prolene is passed through tendon (small image in inset). (e) Needle cut of long suture length and both suture ends are passed into the eye of the feeding catheter until it passes through the distal wound. (f) The suture ends are grasped and the tendon is snared onto the feeding catheter. (g) The tendon is retracted back through the sheath and used as retraction while performing the repair with a new suture. (h) The snare and catheter are removed after the repair has been performed. (Reproduced from Wong J, McGrouther DA. Minimizing trauma over ‘no man’s land’ with flexor tendon retrieval. J Hand Surg Eur Vol 2014;39:1004–1006.) Pitfalls for Tendon Retrieval • Inability to deliver the proximal stump through to distal incision site because of swollen tendon end or contracted pulley • Difficulty locating the proximal stump in the palm and, as a result, inability to snare the tendon end to the feeding catheter • Overzealous tendon handling leading to frayed tendons Steps to Avoid These Pitfalls • Use of the modified retrieval method to deliver the tendon end so there is less bulk passing through the sheath, which minimizes tendon “catching” • Use of anatomical landmarks at the distal palmar crease, which can help identify retracted tendon ends • Use of artery clips to secure the suture ends if required It is therefore desirable to minimize the degree of surgical dissection and damage by retrieving the tendon through minimal-access techniques. We have described methods of tendon retrieval using a pediatric feeding tube, which allows the repair with only distal extension of the wound and minimal handling of the tendon.23 Using retrieval methods enables a smaller incision, thereby reducing local postoperative edema of the skin and subcutaneous tissue at the repair site. To retrieve the proximal stump, a transverse incision is made at the distal palmar crease, level with the A1 pulley. A pediatric feeding tube is passed through the sheath in either direction, preferably from proximal to distal. Care must be taken not to dislodge the tendons any further if passing in a retrograde fashion. This can be prevented by holding the proximal stump at the palmar incision. A suture is passed through the divided tendon or tendons proximal to the A1 pulley to “snare” the tendon. No knot is tied, and the suture ends are passed through a hole cut in the side of the feeding tube until they are seen in the distal wound and pulled tight to lock the tendons side-to-side to the tube. The tendons are then delivered distally to the wound by pulling gently on the feeding tube, which ensures the tendon ends are adequately exposed to facilitate ease of repair. This technique allows the repair to be conducted with a small incision of 2 to 3 centimeters in length at the point of division and preserves the tendon ends for repair23 (Fig. 61.8). In experienced hands, this technique works well, and the repair is greatly facilitated; however, there are pitfalls to retrieval that can be avoided (Box 61.2). If techniques of retrieval are not effective, wound extension may be necessary. Regardless of the technique used, gaining sufficient access to undertake a strong repair is important. Pitfalls for Tendon Repair • Insufficient bites of the tendon • Overtightening of the suture repair, which strangulates the tendon and results in bunching of the repair • Complex repairs that catch on the core suture and compromise repair integrity • Complex repairs that increase suture bulk and final repair bulk • Suture needle passage too close to sutures, cutting or fraying the suture material • Multiple forced suture passes using too big or too blunt a needle, causing the tendon to fray Steps to Avoid These Pitfalls • Use of noncutting needles on epitendinous repairs to avoid compromising core repair integrity • Awareness of tendon size, selecting the right suture and suture size, and adjusting the number of throws appropriately to achieve “smoothing” of the repair site for minimal bulk and maximal glide • Appropriate venting of the pulleys to enable glide with intraoperative stress testing (see the next section) • Using a technique to approximate the back wall first to help approximate the tendon ends for ease of core suture repair24 • Positioning suture bites farther than 1 cm from the tendon ends when possible Any repair can rupture and lead to unfavorable results, especially if it is performed badly or it catches on pulleys, is intrinsically weak, or has been exposed to too much force (Box 61.3). The repair relies on a number of factors to give it a low profile and make it mechanically strong and easy to perform. No technique will restore the balance and finesse of the tendon’s intrinsic architecture. It is therefore unsurprising that so many repair types have been described. The numerous patterns of placing the core suture have several common characteristics (Fig. 61.9). The suture must grasp the tendon material on either side of the divide, and a number of strands must cross the gap. Usually 2, 4, or 6 strands are common, and the suture is tied and knotted, which is important because knots of different materials may unravel in variable ways under load. In addition, there is variability of suture material tensile strength. Each of these characteristics are discussed separately (Fig. 61.10). Fig. 61.9 Classic and current flexor tendon repairs. (Reproduced from Langer MF, Oeckenpöhler S, Kösters C, Herrmann K, Wieskötter B. Nahttechniken für Beugesehnen der Hand. Der Orthopäde 2015;44(10):748-756.) Early tendon repairs (e.g., Bunnell25) just passed through the tendon from side to side with no specific attempt to encircle a few of the fibers in a grasp to lessen the risk of pullout. The Kirchmayer-Kessler pattern grasped at few fibers at the four corners of a box-shaped configuration.26,27 Sav age and Risitano28 achieved a much more extensive grasp by criss crossing the suture in addition to increasing the number of strands. Concepts such as locking and grasping of tendon fibers require awareness of the longitudinal and horizontal configuration of the suture pass, but in reality this detail does not alter the overall pull-out strength of tendons significantly, largely because, as load is applied, the configuration of these repairs changes dramatically. The number of strands has been a focus of attention in recent years since Savage28 described his innovative technique of using a single strand of material, passing between the ends six times and thus potentially increasing load bearing by a theoretical factor of three and assuming better load sharing. In practice the load taken by each strand depends on the clinical judgments on the length of each strand and the amount of bedding-in of the suture into the soft tendon material. If one strand were to become extremely tight, the overall effect would approximate a single-strand repair. One way of allowing the forces in each strand to equilibrate load is to perform the repair with one continuous suture. In addition, compression at the repair site rather than gapping can be introduced by staggering the repair (see Fig. 61.10). Experimental studies have shown that areas of the tendon subjected to high strain become acellular, largely because of cell necrosis, and there is a theoretical argument that multistrand repairs are likely to have more cell death and inflammatory change, leading to swelling. Clinical evidence of this has recently been reported with observations that two-strand repairs typically rupture at 2 weeks from suture failure, whereas multistrand repairs rupture as late as 8 weeks, probably related to failure of the tendon ends to heal.29
61
Flexor Tendon Injuries
Avoiding an Unfavorable Result in Flexor Tendon Repairs
Anatomy
Biology of Healing
A Repair Strong Enough for Early Mobilization
Preoperative Planning
Timing and Delays in Diagnosis and Treatment
Mechanism of Injury
The Patient: Collateral Pathologies
The Surgeon: Training and Experience
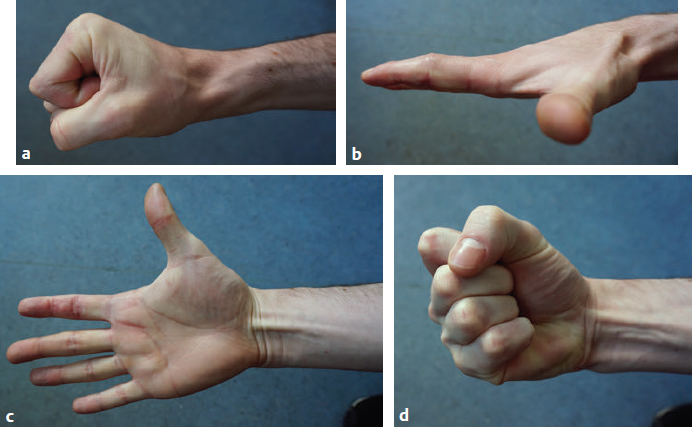
Patient Tendon Examination
Intraoperative Factors that Influence Outcome
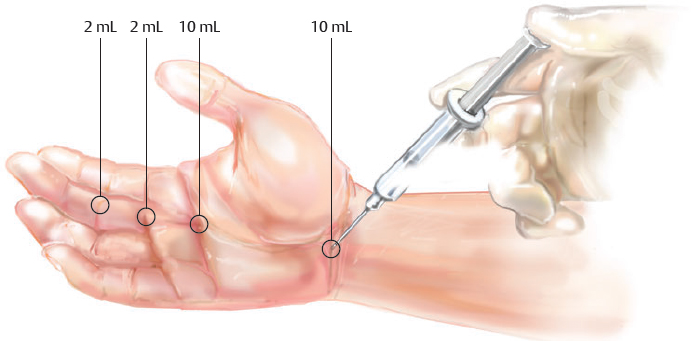
Anesthesia
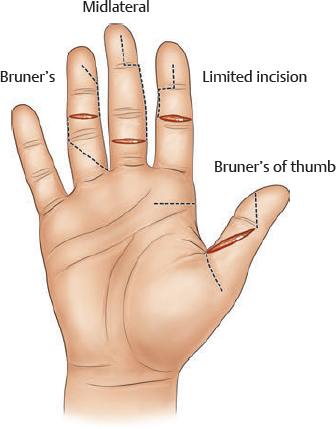
Incisions and Tendon Retrieval
The Tendon Repair
The Core Suture
Grasp
Number of Strands
Plastic Surgery Key
Fastest Plastic Surgery & Dermatology Insight Engine