16 Flaps Abstract A flap is a unit of tissue transferred from a donor to a recipient while retaining its own blood supply. Their composition, vascularity, and method of transfer constitute the main features by which flaps are classified and named, ranging from small local rotation flaps to large composite free flaps. Nevertheless, regardless of their complexity, all flaps are governed by the same principles, including thorough preoperative planning of the procedure and possible lifeboats, replacement of like with like, meticulous surgical technique, and mindful consideration of the donor area. In flap surgery there is always a trade-off, namely, morbidity of the donor site, that needs to be proportionate to the benefit gained from the reconstruction, and which may range from a small scar to a graft, a noncritical sensory loss, or a mild functional deficit. Finally, the understanding of the blood supply to the skin together with the evolution and refinements in surgical technique have led to the description and development of perforator flaps, which constitute an important part of the armamentarium in reconstructive surgery. This chapter describes the main aspects of flap surgery, including the physiological events that take place after transfer and the main factors affecting flap survival, followed by a thorough description of flap classification and refinements. Lastly, a section on perforator flaps is provided. Keywords: fasciocutaneous flaps, flap, local flaps, perforator flaps, tissue transfer The term flap originates from the 16th-century Dutch word flappe, which means something that hangs broad and loose, fastened only by one side. The roots of flap surgery can be traced back to 600 BC when Sushruta Samhita described a technique for nasal reconstruction using a cheek flap. Later Greek, Hellenistic, and Roman physicians described reconstructive procedures mirroring the Indian methods. In the 16th century, during the Renaissance period, the transfer of tissue to a distant site using tubed and pedicled flaps was reported in Italian literature. One particular application of this technique—reconstructing a nose from a forearm skin flap—was popularized by Tagliacozzi. Despite these early descriptions, flap surgery did not gain wide acceptance during this time. Two centuries passed before skin grafts and flaps reemerged in Europe. During this time there were descriptions of the transportation of skin to an adjacent area through rotation or transposition. The French were the first to describe advancement flaps, but this was done in isolation of any knowledge regarding blood supply, rendering these flaps random pattern. The evolution of flap surgery continued through the First and Second World Wars, when pedicled flaps were used extensively. Despite the meticulous planning that was involved, it is likely that these flaps were designed without an appreciation for their axial vascular supply. These so-called random flaps were designed with a strict length to width ratio under the assumption that the flap would only survive in its entire length if its base were wide enough to allow proper blood supply. Esser, however, in Germany, did note the importance of incorporating a vascular pedicle within the flap; he named his flaps biological or arterial flaps. In the 1960s, Stuart Milton demonstrated that the reliability of a skin flap depended on its vascularization. This led to the work of Ian McGregor and Gwyn Morgan, who made a distinction between axial and random pattern skin flaps in the early 1970s. Also in the 1970s came the introduction of muscle and musculocutaneous flaps. As the use of these flaps gained momentum, the understanding of flap surgery increased, and this eventually led to the birth of free tissue transfer. In 1973, Rollin Daniel and Ian Taylor reported the first cutaneous free microvascular transfer—a groin flap used for lower extremity reconstruction. The ability to perform safe, reproducible, and reliable microvascular procedures dramatically expanded the surgeon’s options to close, replace, or augment areas of deficit and deformity. In the 1980s the tissue types used within flaps increased significantly. Fasciocutaneous flaps, osseous flaps, and osteocutaneous flap were all described during this time. Further advancement came in the 1990s with the introduction of perforator flaps. With this development came mapping of the perforator vessels throughout the body, allowing the design of countless potential flaps. Today free flaps and perforator flaps are a routine part of our armamentarium as reconstructive surgeons. Further innovations and advancements will be influenced by our knowledge of perforator anatomy and by the growth of supermicrosurgery. A flap is a unit of tissue that maintains its own blood supply while being transferred from a donor site to a recipient site. Unlike grafts, flaps are not dependent on the recipient bed for vascularization. Therefore the volume of tissue that can be transferred is significantly greater than that of a graft and can contain multiple types of tissue, including skin, muscle, fascia, nerve, and bone. The term reconstructive ladder has been coined by plastic surgeons to describe the options for management of wounds, starting from the most simple and ascending in complexity. The lowest rung of the ladder is healing by secondary intention. Typically a surgeon would consider options for wound closure in a systematic way so that with any given wound they would work up the ladder until they reached the simplest option that would facilitate closure of the wound in question. This method, though safe and systematic does not necessarily provide the best solution. Nowadays, with the technical advancements and increasing experience in complex reconstructive methods, including microsurgery and free tissue transfer, the reconstructive ladder has been gradually displaced by the “reconstructive elevator,” where the best option is chosen—which may or may not be the simplest. This approach allows the reconstructive surgeon to move up and down freely and select the most appropriate option for any given defect. Flaps rely on the maintenance of adequate perfusion for their survival. An understanding of the physiology as it relates to the metabolic demands of the mobilized tissue allows the surgeon to best plan flaps. In addition, the understanding of flap physiology relies on a detailed knowledge of the normal blood supply to the skin. Cutaneous blood supply provides thermoregulation and nutritional support. Nutritional support is regulated largely by the capillary network, whereas thermoregulation is controlled by arteriovenous shunts, which are mainly located in the dermal vascular plexus within the reticular dermis. A number of factors in the microcirculation contribute to the regulation of blood flow, and these can be broken down into local and systemic factors. Fig. 16.1 The reconstructive ladder. (Reproduced from Janis, Essentials of Plastic Surgery, 2nd edition, ©2014, Thieme Publishers, New York.) Local control, or autoregulation, includes the following: • Metabolic factors: These primarily act as vasodilators and include hypercapnea, hypoxia, acidosis, and hyperkalemia. These factors are more significant in muscle compared with skin because muscle has a higher metabolic requirement. • Myogenic reflex: This reflex triggers vasoconstriction in response to distension of isolated cutaneous vessels, thereby maintaining capillary flow at a constant level independent of arterial pressure. • Local hypothermia: This acts directly on vascular smooth muscle, which causes vasoconstriction. • Increased blood viscosity: This is less significant but may also decrease cutaneous blood flow. Systemic control is facilitated by the following: • Neural regulation: This is the most important system of control in cutaneous blood supply. It acts through sympathetic adrenergic fibers—vasoconstriction is induced by α-adrenergic receptors and vasodilation by β-adrenergic receptors. These act together to maintain vascular smooth muscle control at the microcirculatory level. • Humoral regulation: The cutaneous circulation is extremely sensitive to circulating adrenaline and noradrenaline, which cause vasoconstriction through their action on α-adrenergic receptors. Serotonin and thromboxane A2 may also produce vasoconstriction, whereas histamine and bradykinin cause vasodilation. Another important concept in cutaneous blood supply is the angiosome as described in Chapter 15. Taylor and Palmer coined the term angiosome to refer to the tissue supplied by a named artery and observed that adjacent angiosomes are in communication with one another via choke vessels. This relationship is important when designing flaps. When raising a flap on an axial blood vessel the angiosome supplied by that vessel and the adjacent angiosome can be supported. Incorporating tissue distant to this invites vascular compromise and flap failure. The elevation of a skin flap elicits a number of changes that disrupts the delicate balance in cutaneous blood flow. Success of a flap relies on physiological changes that allow adequate perfusion to be maintained and restored. Initially there is a reduction in arterial blood supply and ischemia. This leads to a dilatation of the arterioles and capillaries with resultant congestion and edema. At ~ 72 hours there is a progressive increase in circulatory efficiency—vascular anastomoses develop between the flap and the recipient bed, the size and number of functioning vessels increase, and functioning vessels are reoriented along the long axis of the flap. These changes plateau about 7 days after flap elevation. Flap failure can be caused by arterial or venous insufficiency. Inadequate arterial inflow causes ischemia and inflammation, with increased edema. The microcirculation narrows, blood flow slows, and clot forms within the microand macrocirculation. Initially these changes are reversible, if the arterial inflow is restored, but as time goes on the changes become irreversible. Inadequate venous outflow can also result in flap necrosis, independent of arterial supply. If both arterial and venous circulation are inadequate the risk of flap failure increases exponentially. The success of tissue transfer depends on several factors, which must be considered by the surgeon and optimized as needed before embarking on a flap reconstruction procedure. Systemic conditions, such as diabetes mellitus, immunosuppression, malnutrition, connective tissue disorders, and peripheral vascular disease, can compromise flap vascularity and result in delayed healing or total flap failure. These conditions should ideally be medically optimized prior to undertaking flap surgery. Smoking is associated with an increased risk of flap necrosis. Nicotine has a number of negative effects—it causes direct endothelial damage and systemic vasoconstriction. These effects significantly decrease capillary blood flow and limit oxygen delivery. Distal perfusion and flap survival are affected by nicotine in a doseand timedependent fashion. Smoking also generally impairs healing due to vasoconstriction, decreased neutrophil function, and decreased collagen synthesis. Successful flap surgery is based on five key principles: 1. Replace like with like. When reconstructing a defect, where possible replace like with like. If this cannot be achieved, use the next most similar tissue substitute available. This assists in producing the most natural result for the patient. 2. Think of reconstruction in terms of units. According to Millard, the body can be divided into seven main areas—the head, neck, trunk and four extremities. Each of these areas can further be separated into units and subunits. Each of these subunits should be considered separately when establishing a reconstructive plan. 3. Always have a plan, a pattern, and a lifeboat. When considering a defect, utilize the reconstructive elevator to establish viable options and compare the advantages and disadvantages of each of these. Sometimes the simplest plan is the safest, but on some occasions a more complex option may provide the best overall result. One should always aim to restore form and function to the best of their ability. In the operating room design the flap and check the markings before starting. The old adage “measure twice, cut once” should hold true no matter how experienced the surgeon. Additionally, anticipate potential difficulties and have a backup plan in mind should the original option fail—“hope for the best but plan for the worst.” 4. Rob Peter to pay Paul. Using the “Robin Hood” principle—rob Peter to pay Paul—but only if Peter can afford it. Everything comes at a price, and with flap surgery we must consider where the tissue is coming from and the potential negative consequences of borrowing that tissue. This goes hand in hand with the fifth and final principle. 5. Never forget the donor area. Both the primary defect and the secondary donor site must be considered equally. We need to ensure that the resultant scars, deformity, and/or disability created by utilizing the donor tissue can be justified. By definition, a flap is a segment of tissue of whatever composition that is transferred from one area to another while maintaining its own vascular supply. This fundamental characteristic is what differentiates a flap from a graft, which depends on vascularization from the wound bed to survive. Any given flap can be classified according to different aspects, though these different classifications are complementary, not mutually exclusive. As a medical student or early surgical trainee, one starts to read and hear about flaps, later participating in flap surgeries, and all these classifications, patterns, and geometrical terms become mixed in a seemingly random manner. Given the different flap classifications, each with its own subcategories, the combinations are multiple, and not all classifications are applicable to all flaps. The problem usually isn’t names and terminology, but instead it is trying to memorize everything instead of acquiring the basic concepts and principles. By incorporating the latter, it will be quite easy to name any flap just by looking at it for a few seconds. However, it is important to note that knowing the “full name” of a flap does not mean that we use the full name every time. Learning the different classifications of flaps is useful for nomenclature and communication purposes among plastic surgeons and other colleagues. Furthermore, knowing a flap’s potential components, its vascularization, and how it can be transferred to the defect has significant clinical implications in preoperative planning, the surgical procedure itself, the management of postoperative complications, and the execution of secondary procedures. Flaps are classified mainly according to their composition, their vascularization pattern, and their method of mobilization. But before going into each classification in detail, the first thing to address is the difference between a pedicled and a free flap. Of course they are both flaps in the sense that they carry their own vascular supply; however, they do so in different ways. Pedicled flaps are those that, in the process of transfer, always remain attached to the body at the point from which they receive their vascular supply. This point can be a broad bridge of skin, a known vascular axis, or even a small, 2 mm perforator vessel. Free flaps, in turn, are those in which the tissue and accompanying vascular pedicle are completely disconnected from the body and transferred to the defect, where the vessels in the flap (donor vessels) are anastomosed to recipient vessels at the recipient site using microsurgical techniques. A flap may be composed of one or several kinds of tissue depending on the reconstructive needs. Furthermore, the various tissue types may be combined in different ways to suit the defect that is being reconstructed. The vast majority of flaps used in reconstructive surgery will include one of the tissues illustrated in If a flap contains only one of these components, then it will usually be named as appears in the figure (i.e., skin flap, fascial flap, fat flap, muscle flap, and bone flap). However, when combined, these components are usually referred to by the following combining forms: • Skin: -cutaneous • Fat: adipo- • Muscle: myo- • Fascia: fascio- • Bone: osteo- • Nerve: neuro- • Cartilage: chondro- In general, the components are named from deep to superficial. Thus examples of correct terminology regarding flap composition would be myocutaneous flap (muscle and skin), fasciocutaneous (fascia and skin), osteomyocutaneous (bone, muscle, and skin). Flaps receive their blood supply through either a random or an axial pattern. These flaps are generally composed of skin and fat and are named because they are based mainly on the subpapillary, dermal reticular, and subdermal plexuses without an identifiable source vessel ( Fig. 16.3 (a,b) Vascular basis of random flaps. (Reproduced from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.) Axial flaps incorporate an identified vessel along their longitudinal axis, usually coursing in the subcutaneous plane. This fact makes these flaps much more reliable from a vascular point of view, and of course less subject to the restrictions already mentioned, thereby allowing larger arcs of rotation and greater versatility in design. However, the need to have a known blood vessel within these flaps poses certain anatomical limitations, as opposed to random flaps, which can be elevated virtually anywhere in the body. Commonly used axial flaps and their vascular source are listed in Because the axial vessel often runs in the subcutaneous plane, these flaps are usually composed of skin and fat. However, for either reconstructive purposes or to enhance their vascularization, the underlying fascia (with its vascular plexuses) can also be included, thus forming an axial fasciocutaneous flap. Fasciocutaneous flaps have their own systems of classification, which are described later in this chapter. Table 16.1 Examples of axial pattern flaps
16.1 Introduction
16.2 Basic Science
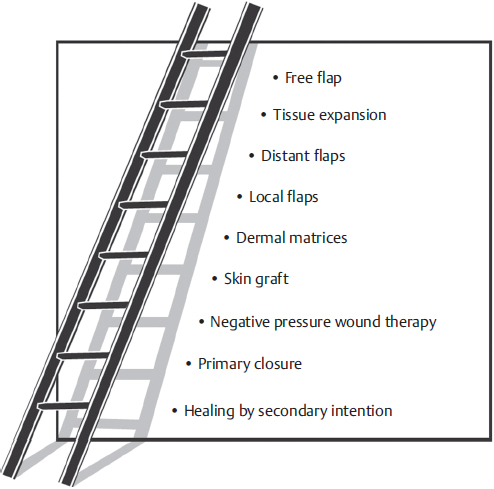
 Fig. 16.1 demonstrates the rungs increasing in complexity until the top of the ladder—free flaps—is reached.
Fig. 16.1 demonstrates the rungs increasing in complexity until the top of the ladder—free flaps—is reached.
16.2.1 Flap Physiology
16.2.2 Risk Factors for Flap Survival
16.3 Principles of Flap Surgery
16.4 Flap Classification
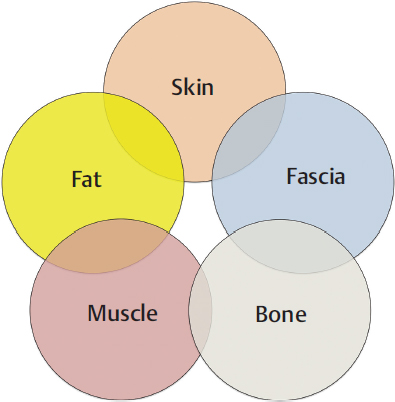
16.4.1 Classification According to Composition
 Fig. 16.2.
Fig. 16.2.
16.4.2 Classification According to Vascularization
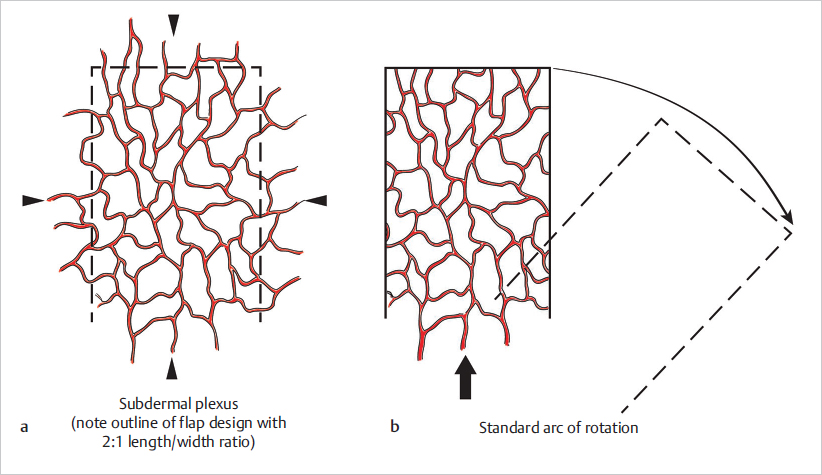
Random Flaps
 Fig. 16.3). This fact poses some restrictions on flap design and elevation, such as a limited arc of rotation and the need for proximity to the defect. Also, given that their blood supply comes from their base, it has been a traditional belief that, in order to survive, random flaps should adhere to relatively rigorous length to width ratios of around 2:1 (or 3:1 on the face given its rich vascularization). Although this has been challenged and is no longer a strict rule to follow, it does illustrate the principle that random flaps do need to have an adequate width to ensure sufficient vascularization.
Fig. 16.3). This fact poses some restrictions on flap design and elevation, such as a limited arc of rotation and the need for proximity to the defect. Also, given that their blood supply comes from their base, it has been a traditional belief that, in order to survive, random flaps should adhere to relatively rigorous length to width ratios of around 2:1 (or 3:1 on the face given its rich vascularization). Although this has been challenged and is no longer a strict rule to follow, it does illustrate the principle that random flaps do need to have an adequate width to ensure sufficient vascularization.
Axial Pattern Flaps
 Table 16.1.
Table 16.1.
Flap | Axial vessels |
Forehead | Supratrochlear |
Retroauricular | Retroauricular |
Deltopectoral | Second and third intercostal perforators from internal mammary artery |
Groin | Superficial circumflex iliac |
Dorsalis pedis | Dorsalis pedis |
16.4.3 Classification According to Method of Movement
In order to reach the defect, the flap must be mobilized in a certain direction. The basic principle of flap mobilization rests on taking tissue from a mobile area and bringing it into the defect without undue tension. Whenever possible, the donor site is closed directly; otherwise a skin graft can be used. There are four basic ways to mobilize a flap into a defect: rotation, advancement, transposition, and interpolation. The method of mobilization depends on a number of factors, including the area of the body where the flap is to be raised; the condition, laxity, and neurovascular supply of the neighboring tissues; and the dimensions and shape of the defect, although the latter can be adapted for the flap to fit properly, even if this means sometimes removing healthy tissue. Rotation, transposition, and interpolation flaps all have a pivot point, around which the flap rotates to reach the defect, and an arc of rotation, which is the area that can be effectively covered by the flap. In addition, the line of maximal tension is a line that runs from the pivot point to the point of the flap that will be furthest away from it after inset ( Fig. 16.4). It should be noted that, when a flap is progressively turned through its arc of rotation, its effective length decreases accordingly (
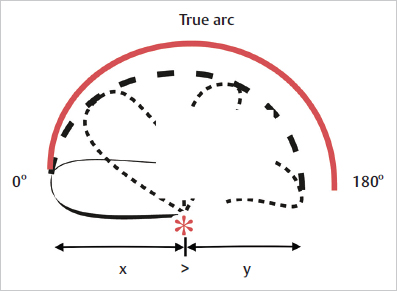
Fig. 16.4). It should be noted that, when a flap is progressively turned through its arc of rotation, its effective length decreases accordingly ( Fig. 16.5). Therefore, careful planning of these flaps keeping these features in mind and making the flap longer than the defect is crucial. Before making any incisions, a good recommendation is to simulate the movement of the proposed flap into the defect by using a piece of cloth or gauze held firmly at the pivot point. This gives a good estimate of the actual reach of the flap and allows redesign if necessary. It is generally better to err on the side of excess, provided that there are no issues with the donor site. A flap that outreaches the defect can be easily trimmed; a flap that does not reach the defect, or does so with excessive tension, is a disaster.
Fig. 16.5). Therefore, careful planning of these flaps keeping these features in mind and making the flap longer than the defect is crucial. Before making any incisions, a good recommendation is to simulate the movement of the proposed flap into the defect by using a piece of cloth or gauze held firmly at the pivot point. This gives a good estimate of the actual reach of the flap and allows redesign if necessary. It is generally better to err on the side of excess, provided that there are no issues with the donor site. A flap that outreaches the defect can be easily trimmed; a flap that does not reach the defect, or does so with excessive tension, is a disaster.
Rotation Flaps
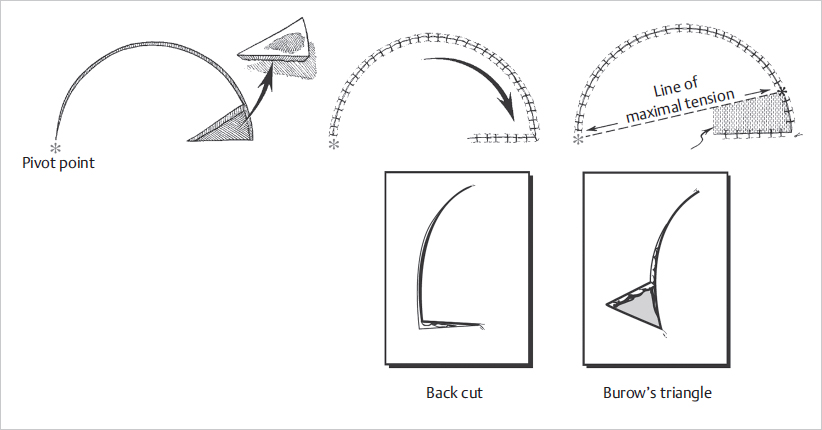
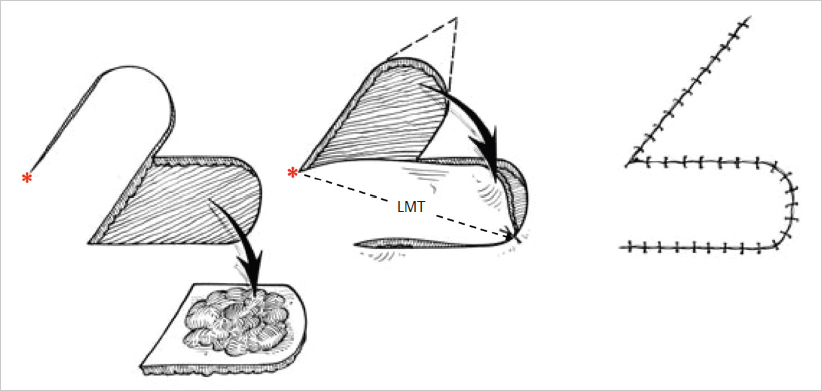
These flaps are one of the most utilized resources in reconstructive surgery. They can be used virtually anywhere on the body, from the scalp to the sole of the foot, with varying dimensions and composition. The defect is tailored to an isosceles triangle that represents the edge of a semicircle, which corresponds to the flap. The circumference of the flap should generally be four to six times the diameter of the defect to allow adequate mobilization onto the defect. Once elevated, the flap is rotated over the defect to cover it ( Fig. 16.4). To facilitate rotation, either a back cut or a Burow’s triangle excision can be performed at the pivot point (
Fig. 16.4). To facilitate rotation, either a back cut or a Burow’s triangle excision can be performed at the pivot point ( Fig. 16.4). Since the back cut is made toward the center of the semicircle, care must be taken to ensure that the flap’s vascularization is not compromised. A Burow’s triangle incision, in contrast, is made away from the pedicle and hence has no influence on the flap’s blood supply. The donor area can usually be closed primarily, although a skin graft might be necessary in some instances.
Fig. 16.4). Since the back cut is made toward the center of the semicircle, care must be taken to ensure that the flap’s vascularization is not compromised. A Burow’s triangle incision, in contrast, is made away from the pedicle and hence has no influence on the flap’s blood supply. The donor area can usually be closed primarily, although a skin graft might be necessary in some instances.
Transposition Flaps
Transposition flaps resemble rotational flaps in that they too rotate about a pivot point to reach the defect; however, whereas rotational flaps are semicircular in shape, transposition flaps generally have a more linear configuration (e.g., rectangular, rhombic). Transposition flaps are based at the edge of the defect and designed anywhere from right next to the defect to 90 degrees from it ( Fig. 16.6). Hence, in most instances, a transposition flap must jump over a segment of healthy tissue to reach the wound. When designing a transposition flap, it must be considered that, as the angle of the flap’s axis in relation to the defect is increased, the flap’s effective length decreases (
Fig. 16.6). Hence, in most instances, a transposition flap must jump over a segment of healthy tissue to reach the wound. When designing a transposition flap, it must be considered that, as the angle of the flap’s axis in relation to the defect is increased, the flap’s effective length decreases ( Fig. 16.5). Closure of the donor site is done either by direct closure or by skin grafting. Rhombic flaps and the z-plasty are common examples of transposition flaps.
Fig. 16.5). Closure of the donor site is done either by direct closure or by skin grafting. Rhombic flaps and the z-plasty are common examples of transposition flaps.
Fig. 16.4 Rotational flap illustrating the position of the pivot point and direction of line of maximal tension. Back cut or excision of Burow’s triangle can be done to increase mobilization. (Reproduced from Janis, Essentials of Plastic Surgery, 2nd edition, ©2014, Thieme Publishers, New York.)
Fig. 16.5 Effective length of a flap. Note the decrease in length (dotted line) as the flap is turned around its pivot point(*). (Reproduced from Janis, Essentials of Plastic Surgery, 2nd edition, ©2014, Thieme Publishers, New York.)
Advancement Flaps
These flaps are designed with their leading edge on one of the borders of the defect, which they close by sliding over it ( Fig. 16.7). Advancement flaps may have different configurations, but the gliding movement remains the same. As can be inferred, advancement flaps must be designed and used in areas with sufficient skin laxity that allows for adequate mobilization. Common advancement flaps include the rectangular and V-Y flaps.
Fig. 16.7). Advancement flaps may have different configurations, but the gliding movement remains the same. As can be inferred, advancement flaps must be designed and used in areas with sufficient skin laxity that allows for adequate mobilization. Common advancement flaps include the rectangular and V-Y flaps.
Interpolation Flaps
By definition these flaps do not share a border with the defect; hence their transfer implies crossing over healthy intervening tissue. They are a useful tool to reconstruct full-thickness wounds with nearby like tissue when adjacent skin is not available or is insufficient. Once the portion of the flap covering the defect becomes vascularized from the wound bed (approximately 2–3 weeks after transfer), a second operation is needed to cut the pedicle and fully inset the flap. Alternatively, interpolation flaps may be tunneled through the subcutaneous tissue to reach the defect, in which case a second procedure would not be needed. The forehead flap for nasal reconstruction, and the heterodigital neurovascular island flap (i.e., Littler’s flap), are good examples of interpolation flaps ( Fig. 16.8).
Fig. 16.8).
Hinge Flaps
Because of the way by which these flaps are mobilized into the defect, hinge flaps are also named turnover flaps. They are tailored right next to the defect with their pedicle based at the flap–defect interface. The flap is then elevated in the subcutaneous plane and turned over the wound like a page in a book. This movement results in the superficialmost surface of the flap facing downward on the wound. These flaps can be raised with or without skin; in the former case, the skin can be used for internal epithelial lining. Additionally, because the flap’s raw surface becomes exposed after hinging, either a second flap or a skin graft is necessary to resurface the wound.
16.4.4 Classification According to Proximity
Based on their spatial relationship with the defect, flaps can be broadly grouped into local, regional, and distant. Minding some inevitable exce ptions, local and regional flaps are almost always pedicled flaps, whereas distant flaps are usually transferred as microvascular free flaps.
Local Flaps
As their name implies, local flaps are obtained from tissues in the immediate vicinity of a defect, providing a relatively simple, straightforward, and reliable solution to coverage of a number of wounds. Nevertheless, in many cases local tissues are either insufficient or inadequate to be used for reconstruction (e.g., due to scarring from previous surgeries or stiffness and poor vascularity from previous radiation). In these cases, regional or distant options must be contemplated to achieve successful coverage and proper wound healing. It is important to note that, although local flaps may comprise one or more of the components listed earlier in this chapter, this does not alter in any way their “local” denomination. In this sense, a small rhomboid skin transposition flap for a defect in the temple area, an advancement pectoralis major muscle flap for a sternal wound, and a large rotational fasciocutaneous flap to resurface a sacral pressure sore are all examples of local (and of course pedicled) flaps, even though their size and composition are quite different.
Fig. 16.6 Transposition flap. *, pivot point; LMT, line of maximal tension. (Modified from Zenn, Jones, Reconstructive Surgery Anatomy, Technique, and Clinical Applications, ©2012, Thieme Publishers, New York.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree