A retrospective observational case study and a literature review were conducted to evaluate how anatomic findings, especially those related to the periorbital zone, serve as a guiding compass for injectable implants. Treatment techniques and product selection will be discussed for patients with negative vector, shallow orbit, and deep set eyes. Versatility of injectables will be demonstrated on patients with peanut face, iatrogenically altered anatomy (after surgery), and trauma.
Key points
- •
There are a variety of dermal fillers approved by the US Food and Drug Administration, each with unique properties that make them suitable for volume augmentation at different areas of the face.
- •
Injectables, particularly hyaluronic acid fillers and biostimulant agents, are a useful, minimally invasive alternative for treatment of patients with specific anatomic features.
- •
Negative vector topography can be corrected with tear trough and midface injections with hyaluronic acid, as well as biostimulant agent deposition at the maxilla and about the orbitomalar ligament.
- •
Hyaluronic acid fillers can be used to dissimulate postoperative high upper eyelid crease and volume subtraction.
- •
Injectables can be used to achieve an enhanced final result in patients after surgical reconstruction for trauma or prior cosmetic surgery.
Video content accompanies this article at http://www.facialplastic.theclinics.com .
Introduction
Anatomy is essential for filler injection safety and a guiding compass for treatment design. This article focuses on 4 topics: patient assessment, dermal filler selection, anatomic findings, and injection techniques. It includes visual examples that illustrate signature features that are encountered in oculoplastic practice.
Patient assessment
During patient evaluation, anatomic variations and individual factors are identified, allowing the physician to determine the most appropriate treatment approach. The facial shape must be evaluated from different angles and both at rest and during animation to correctly assess its proportion and symmetry. , It is important to determine the patient’s needs and expectations, discuss the physician’s evaluation with the patient, and combine both to produce an effective and satisfactory treatment plan.
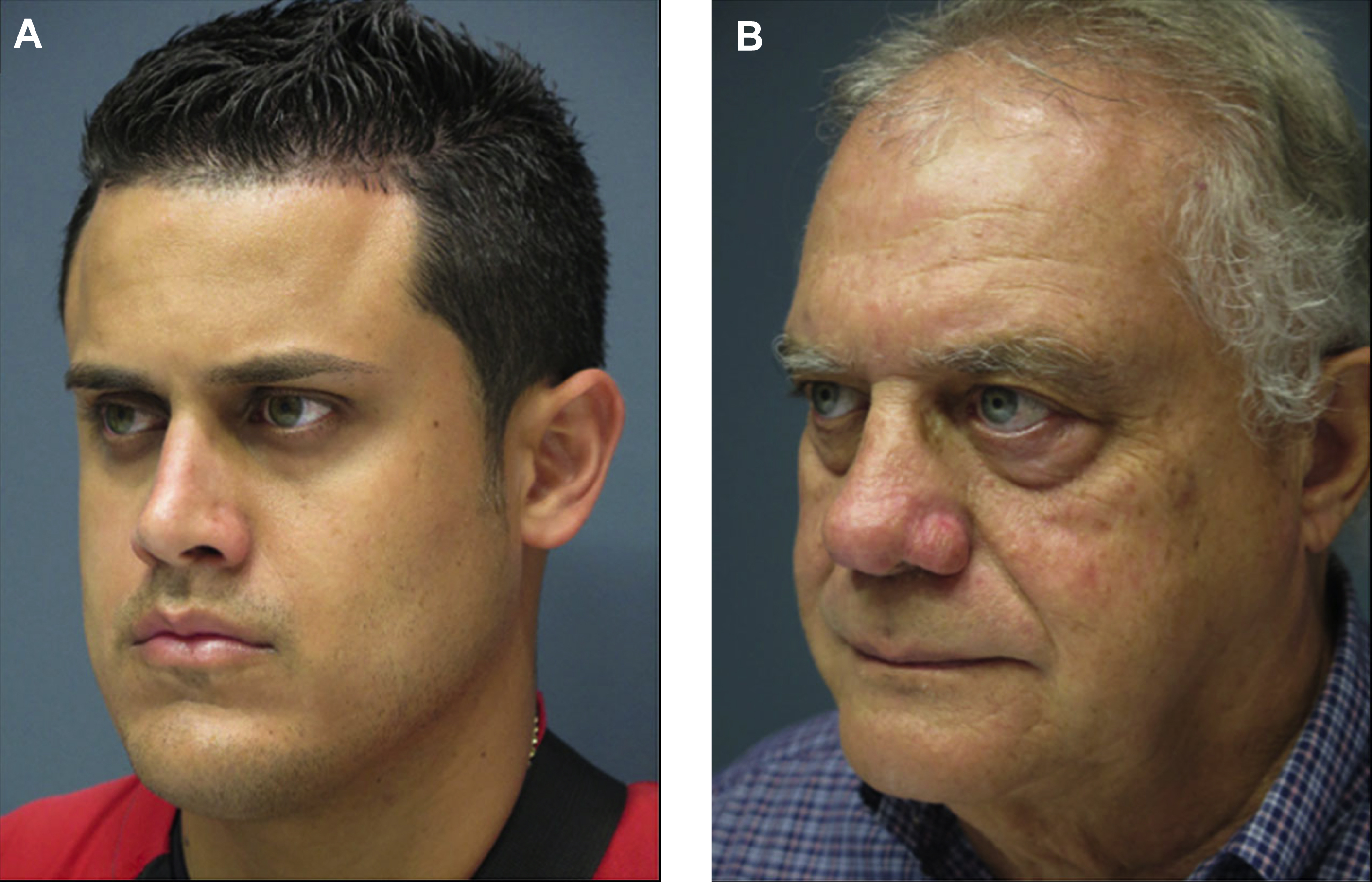
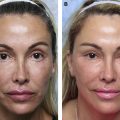
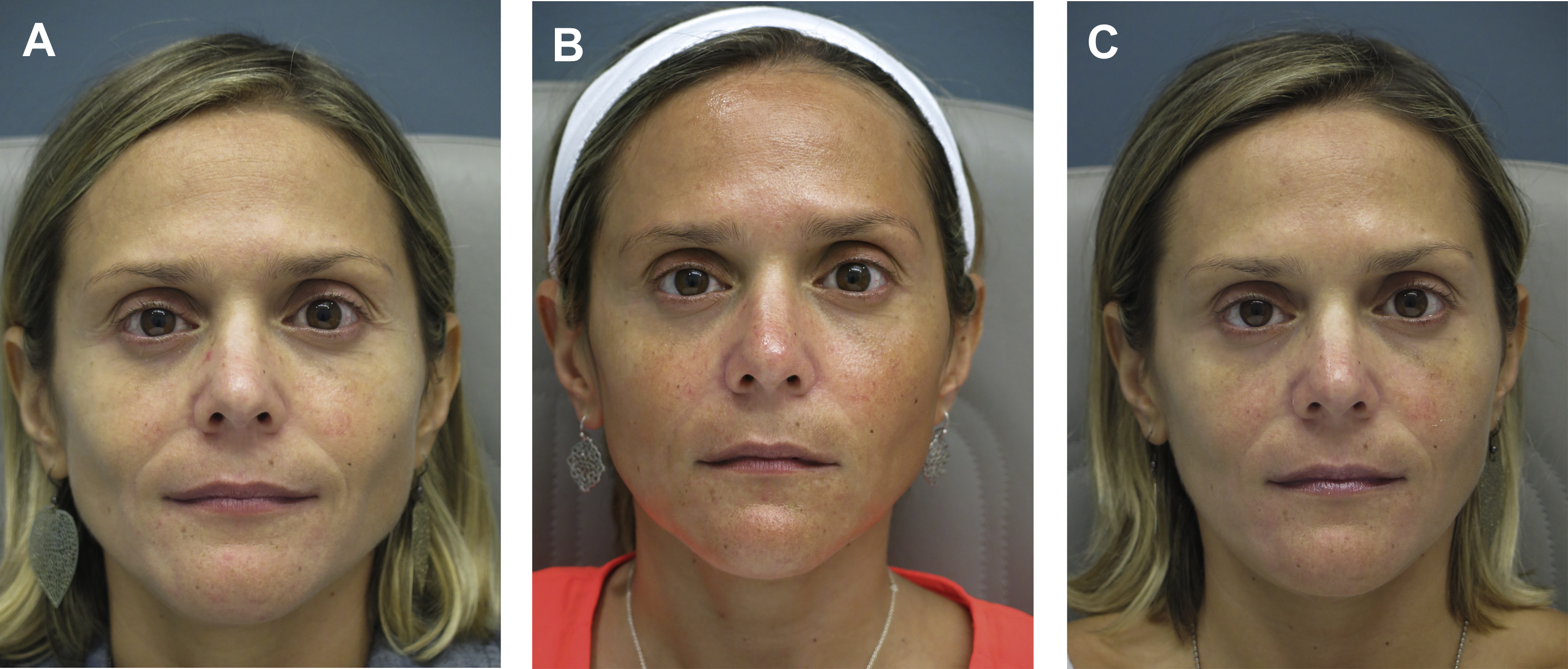
Dr Ava Shamban coined the concept of a “signature feature” and, in theory, we all have one. Shamban highlights the importance of preserving these features during aesthetic enhancement, as they contribute to what makes an individual recognizable to others. Physicians can focus on these salient features and decide whether they can be enhanced or diffused by working around them. For example, in a patient with 2 signature features, a prominent nose and full lips, injectables can be used to enhance the lips and minimize the nose prominence ( Fig. 1 ). The result is the simultaneous balancing and preservation of both features.
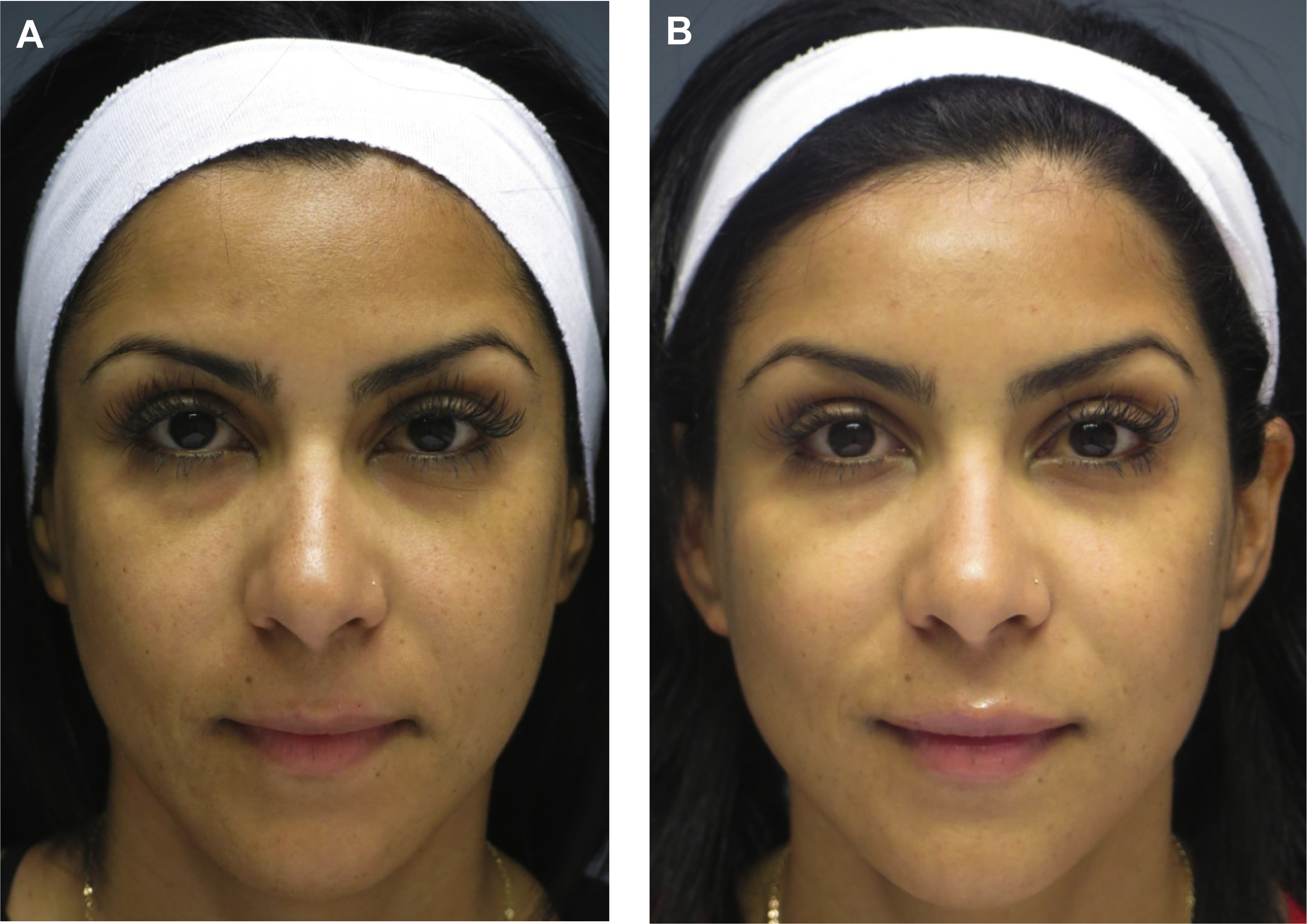
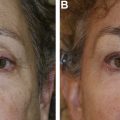
Dr Steven Dayan proposes a 2-fold approach to patient assessment: symmetry as an objective and eyes as the focal point of facial rejuvenation. Asymmetric features require asymmetric patterns of injection to achieve this goal ( Fig. 2 ). A common misconception is that facial shaping with injectables is exclusively about adding volume. However, the possibility of volume reduction plays a role in patient assessment. In a patient with lower face or masseteric hypertrophy, volume can be decreased using a neurotoxin, and deficient midface and temples can be compensated using a biostimulant agent ( Fig. 3 ).
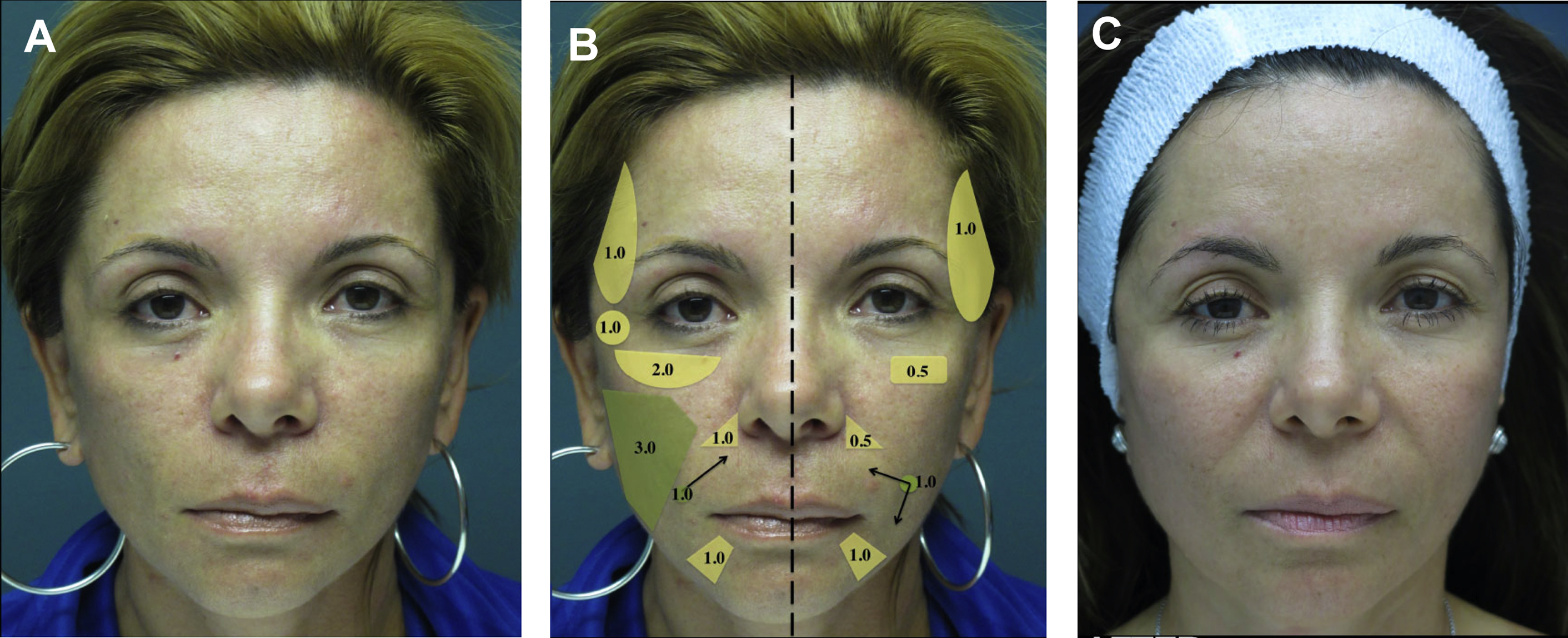
Nonsurgical facial rejuvenation provides a number of advantages for the patient: it is safe, less invasive, and entails a shorter recovery period. Volume restoration is an important part of facial rejuvenation, and dermal fillers are effective methods of achieving this objective. There are a series of dermal fillers that have been approved by the US Food and Drug Administration (FDA), each with unique properties that make them suitable for different procedures and facial regions ( Table 1 ).
| Technology | Product | Cross-Linking (%) | Hyaluronic Acid Concentration (mg/mL) | |
|---|---|---|---|---|
| Hyaluronic acid | Hylacross | Juvederm Ultra | 6 | 24 |
| Juvederm Ultra Plus | 8 | 24 | ||
| Vycross | Volbella | _ | 15 | |
| Vollure | _ | 17.5 | ||
| Voluma | <5 | 20 | ||
| NASHA | Restylane L | 1 | 20 | |
| Restylane Lyft | 1 | 20 | ||
| Restylane Silk | 1 | 20 | ||
| XpresHAn | Restylane Refyne | 6 | 20 | |
| Restylane Kysse | 7 | 20 | ||
| Restylane Defyne | 8 | 20 | ||
| Cohesive polydensified matrix | Belotero Balance | Variable | 22.5 | |
| Thixofix | Revanesse Versa | 10 | 25 | |
| Resilient hyaluronic acid | RHA 2 | 1.9–4.0 | 23 | |
| RHA 3 | 1.9–4.0 | 23 | ||
| RHA 4 | 1.9–4.0 | 23 | ||
| Biostimulant agents | PLLA | Sculptra | N/A | N/A |
| Calcium hydroxyapatite | Radiesse | N/A | N/A |
Dermal fillers
Hyaluronic Acid Fillers
Hyaluronic acid (HA) fillers are commonly used for volume restoration in different regions of the face. The available FDA-approved HA fillers can be grouped into several categories based on their characteristics and the technology used to create them (see Table 1 ). Hylacross technology produces monophasic gels, resulting in a product composed of cohesive molecules with a 24 mg/mL concentration of highly cross-linked HA. These products are long lasting, soft, and easy to use. Their hydrophilic properties lead to a diffuse filling effect on injected soft tissues. Vycross technology combines low and high molecular weight HA. It has a high G′ and low swelling capacity, resulting in the ability to produce a high lift. Their concentration varies from product to product, ranging from 15 mg/mL to 20 mg/mL. Nonanimal stabilized HA (NASHA) uses a sizing technology: it breaks up the cross-linked HAs by passing them through sizing screens. These products have a concentration of 20 mg/mL. Products made with XpresHAn technology have the same concentration of 20 mg/mL, but they vary in their degree of cross-linking. HA fillers made with cohesive polydensified matrix technology have the lowest G′ of the HA fillers, accounting for their lower elasticity and viscosity. In cohesive polydensified matrix technology, non–cross-linked HA is added in the second cross-linking stage, resulting in a product with a variable degree of cross-linking. ,
Biostimulant Agents
Calcium hydroxyapatite (Radiesse) and poly- l -lactic acid (PLLA; Sculptra) are 2 FDA-approved biostimulant agents. Radiesse is made of calcium hydroxyapatite spheres suspended in a sodium carboxymethylcellulose gel carrier. It is more substantial in consistency and has minimal hydrophilic properties in comparison with HA fillers. PLLA functions by stimulating the body to produce its own collagen. It does so by activating fibroblasts, resulting in gradual soft tissue augmentation. Although its effects are not immediate, PLLA has the benefit of producing subtle results that are favored by many patients. , It is indicated for patients who desire global facial voluminization.
Filler Material Selection
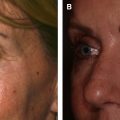
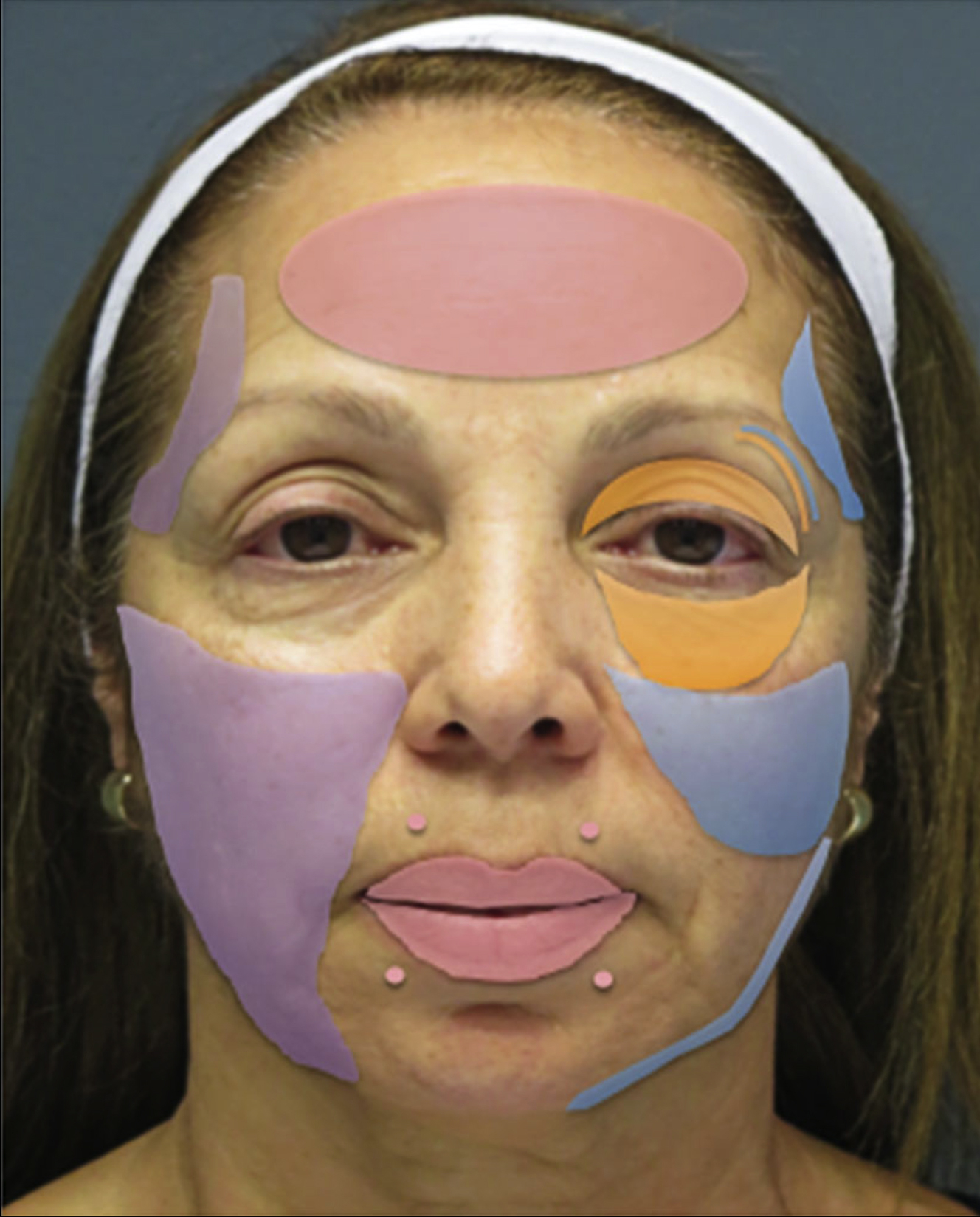
In general, HA fillers with high density, cross-linking, and viscosity are used for deeper volumization in areas such as the midcheek and malar regions. In contrast, products that are softer and less dense are better suited for superficial injections in areas such as the lips ( Fig. 4 ).
Periorbital Area and Tear Trough
HA fillers are suitable for the periorbital area in part because they are reversible with hyaluronidase and temporary. Fillers used in the tear trough and periorbital areas should be made of smaller particles, have less HA concentration, and contain less hydrophilic propensity. Hydrophilic fillers attract water, which can result in a degree of swelling that is undesirable in these regions. Therefore, the use of fillers made with Hylacross technology is not recommended for the periorbital area and tear trough. Fillers with a lower water binding capacity are less likely to result in swelling or bluish discoloration under the eyes. The use of low-viscosity HA is considered safe, whereas injection of high-viscosity HA is not recommended for the tear trough.
HA fillers made with NASHA, Vycross of low HA concentration, and cohesive polydensified matrix technologies are recommended for use in the tear trough and lower eyelid region. NASHA fillers are most commonly used for periorbital volume augmentation. HA fillers with a cohesive polydensified technology have the lowest G′ and allow for greater injection precision. , They are well-suited for injection of superficial regions of the face, as well as atrophic scars. , Because the skin of the tear trough is very thin, small amounts of filler are injected, and additional sessions are performed at another time if necessary.
Temporal Fossa
Both HA fillers and biostimulant agents can be used for temporal fossa augmentation. The choice of dermal filler used for temporal fossa augmentation depends on whether treatment is superficial or deep. Lower G′ products are preferred for superficial treatment of the temporal hollow, whereas products with a high G′ and cohesivity are used for the injection of deep areas. The placement of dermal fillers likewise depends on the dermal filler that is used. HA fillers should be placed in either the subcutaneous plane or the plane between the superficial and deep temporal fascia. When biostimulant agents and HAs with a high G′ are used for augmentation of the temporal fossa, they should be placed deep to the temporalis muscle. If placed within the muscle, their particles may migrate or lump, leading to undesirable results.
HA fillers made with NASHA technology can be used for superficial volume augmentation in the temporal fossa owing to their small particle size and hydrophilic properties. The filler should be injected into the subdermal space at the level of the superficial temporal fascia. Viscous fillers with a larger particle size can be used in the temporal area, because it is thicker than surrounding areas such as the eyelids. PLLA and calcium hydroxyapatite biostimulant agents are suitable for deep temporal fossa augmentation.
Upper Eyelid
Moderately concentrated HA fillers, such as those made with NASHA, Vycross of low HA concentration, XpresHAn with low cross-linking, and cohesive polydensified matrix technologies, are recommended for use in the upper eyelid region. These HA fillers are ideal for use in the thin upper eyelid region, because use of less concentrated fillers may result in diffusion of the product, whereas more viscous fillers can result in a lumpy appearance. Use of the lift, inject, and massage technique is recommended when using HA fillers made with NASHA technology for postoperative upper eyelid hollows. This maneuver elevates the brow adjacent to the hollow above the orbital rim. The HA injected is molded on the rim to diffuse through the tissue planes and smoothly fill the deficit. The biochemical characteristics of the NASHA product allow the injector to achieve a 3-dimensional lift, successfully restoring orbital volume loss secondary to aging or excessive fat removal during upper eyelid blepharoplasty. , , ,
Midface
Fillers with a high HA concentration and G′ are recommended for deep injection at the midface. These properties give fillers a high lifting capability that results in a desired plump appearance in this area. HA fillers made with Vycross (20 mg/mL HA concentration) technology are indicated for augmentation in the cheek region. These products can create a long-lasting lift while maintaining a low swelling capacity. They are malleable, reversible, and can be injected into the subcutaneous and supraperiosteal levels. Specifically, HA fillers with Vycross technology can be used for the lateral, anterior, and medial cheek areas. HA fillers with Vycross and NASHA technologies are recommended for the lid–cheek junction. Additionally, NASHA fillers with large particle size can be used for the midface region.
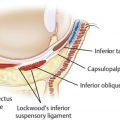
Negative vector patient
Youthfulness and facial attractiveness are usually correlated with a smooth transition between the periorbital zone and adjacent anatomic areas, such as the midface, temples, and brow complex. The negative vector patient lacks support for the globe and orbit from the midface–bony maxilla or soft tissue ( Fig. 5 ). This lack of support leads to lower lid and cheek descent, undereye bags, and scleral show, giving the patient an aged appearance. , For the negative vector patient, infraorbital concavity demands convexity. HA fillers are an effective treatment option for correcting the negative vector. , This treatment modality is less invasive than methods such as fat repositioning and alloplastic implants; additionally, it can be reversed with hyaluronidase if necessary.