19 Extremity burn reconstruction
Synopsis
 In electrical burn injuries, the patient’s conditions often seems less critical than it really is.
In electrical burn injuries, the patient’s conditions often seems less critical than it really is.
 Always consider compartment syndrome when limbs have extensive burns.
Always consider compartment syndrome when limbs have extensive burns.
 When perform escharotomy on the limbs, pay attention to dangerous sites, such as the basilic or saphenus veins, and the radial or tibial nerves.
When perform escharotomy on the limbs, pay attention to dangerous sites, such as the basilic or saphenus veins, and the radial or tibial nerves.
 The extremites are the most frequent areas affected by post-surgery scar contractures. Correct positioning of articualtions, good management of scars and post-operative care are mandatory.
The extremites are the most frequent areas affected by post-surgery scar contractures. Correct positioning of articualtions, good management of scars and post-operative care are mandatory.
 The Z-plasty technique (in its all variations) remains one of the most effectiveness procedure for post-burn scar retractions.
The Z-plasty technique (in its all variations) remains one of the most effectiveness procedure for post-burn scar retractions.
 Reconstructive surgery of burn sequelae often needs “imagination”. Due to the presence of scars, to the damaged tissues, to the lack of good vessels, it is very hard to follow a single preoperative plan. You need many arrows in your quiver.
Reconstructive surgery of burn sequelae often needs “imagination”. Due to the presence of scars, to the damaged tissues, to the lack of good vessels, it is very hard to follow a single preoperative plan. You need many arrows in your quiver.
Introduction
The majority of burn-related accidents involving the extremities, occur in the workplace and are mainly due to fire and electricity. In a domestic environment the majority of victims are babies, who suffer burns owing to contact with hot liquids.1,2
Extremity burns can be considered “difficult” burns, especially if they are extensive circumferential burns. Deep second- and third-degree burns, if not properly treated, can result in severe compartment syndromes, which can cause the loss of the extremity involved if not treated in a well-equipped burn-specialized facility.3,4
History
The most important progress in limb burn wound therapy occurred during the Second World War, but many concepts such as early excision and grafting of small burns can be traced to Lustgarten in 1891. The major issues addressed during the Second World War era included early excision, skin grafting, splinting, and scheduled mobilization. The work of Allen and Koch in 1942 focused on the concept of closed dressings instead of the exposure technique in the care of burn patients.5 In the same period, many authors (e.g., McCorkle and Silvani) described for the first time the importance of early excision and grafting in order to prevent bacterial infections and to minimize hypertrophic scar sequelae in burns involving the upper limb and hands.6
Tangential excision preserving most of the viable tissue, with immediate skin grafting, was suggested initially by Janzekovic and colleagues in the early 1970s. They recommended performing this procedure on the 2nd to 5th day post-burn as the optimal time for best results, with less scarring and good preservation of function.7
Early tangential excision of deep burns, followed by skin grafting, is probably the most significant breakthrough in recent years, leading to a reduction in the mortality rate in burns >40% TBSA.8,9
Basic science/disease process
Electrical burns
In cases of electrocution, the upper extremities are almost always involved, especially as entry or exit points of what is known as the electric arc.10
Often the most serious damage is to the wrist and ankle. Since the hands and feet are the most frequent entry and exit points, the wrist and ankle are the nearest points of electrical resistance. The patient’s clinical condition on admission often looks less critical than it really is as the lesions may be misleading, with limited charring. In other cases, ample charred areas can be witnessed. These are the electric current’s entry and exit points. However, the underlying damage is always more extensive and may well involve the muscular fasciae. This is because nerves and vessels are very good conductors of electricity, and they may suffer damage a few days after the accident. Venous thrombosis or ischemia can be caused by internal destruction of the vessels catalyzed by the electric current. Peripheral neurological damage may occur either with total destruction of the nerve (neurotmesis), if close to the electricity entry and exit points, or with partial axonal damage (axonotmesis), which is generally resolved within a few months.11,12
In cases of high-voltage electrical burns (over 1000 volts), muscles, bones, and nerve structures can be totally destroyed and, as has been said, complete destruction of the major joints is a common observation.13
Myoglobinuria is closely linked to muscular damage, since the destruction of muscle cell causes the release of myoglobin and in turn catalyses myoglobinemia. The first sign of this pathology is the dark pink color of the urine.14,15
The damage caused by an electric current passing through the body, especially if of high voltage, depends not only on the characteristics of the electricity itself but also on the path it follows and on the characteristics of the tissues affected by the electricity. Even if adequate medical and surgical therapy is available, the risk of amputation of the extremity remains high.11
Vascular and neural reconstruction has to be carefully assessed, since electricity-related damage affecting the neurovascular structures can occur even some days after the surgical operation. This is due to irreversible endothelial damage, which affects small terminal vessels in particular. Hence it is preferable in such cases to postpone the more complex reconstruction procedures until secondary surgical procedures are performed.16 During the acute phase, it is therefore advisable to proceed by performing simple temporary skin coverage with a free skin graft and isolating and preparing the vascular and nervous structures that will be treated in a secondary surgical stage.17
Diagnosis/patient presentation
Patients affected by upper extremity burns often suffer other associated traumas as a result of work-related accidents, which are the most frequent cause of this type of injury. An assessment of potential fractures, amputations, wounds, and blast and crush injuries is essential for planning subsequent reconstruction procedures. The functionality of the shoulder joints must be carefully examined.18 If possible, the nervous functionality of the median, ulnar, and radial nerves should also be evaluated.18
Fluid infusion and edema
These phenomena are caused by endothelial damage and by hypoperfusion, which lead to the liberation of vasoactive substances and cytotoxic free radicals, which in turn are responsible for cellular edema and the generalized inflammatory reaction.12,14
The volume of fluids indicated in the resuscitation of burn patients depends on the gravity of the burn, the age and general condition of the patient, and the presence of associated lesions. In patients with burns in more than 15% TBSA, the quantity of fluid to infuse has to be calculated on the basis of the burned area and body dimensions (ABA Guidelines).3
Compartment syndrome
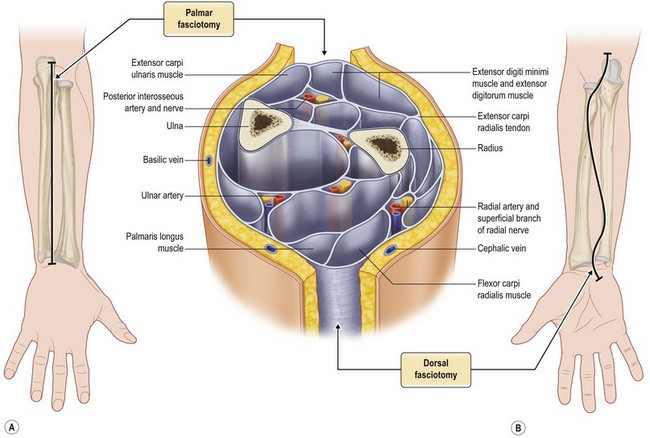
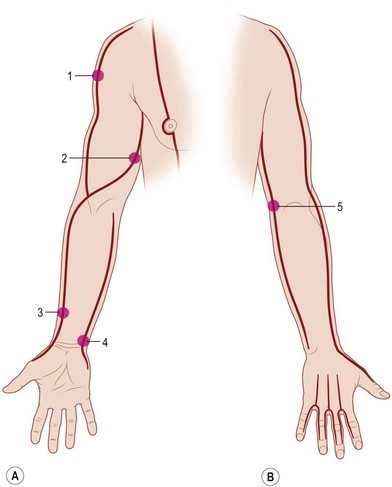
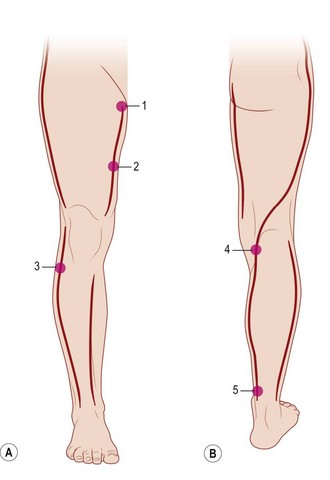
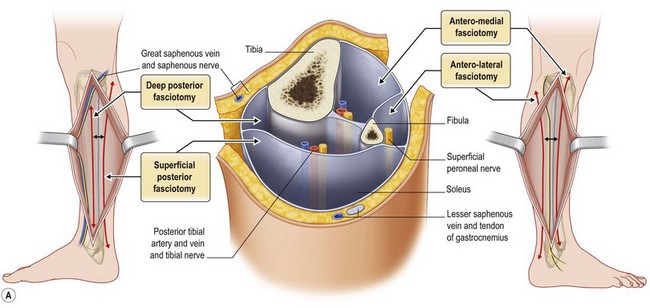
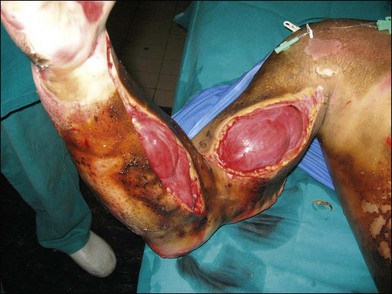
Deep second-degree and third-degree circumferential burns can trigger a serious compartment syndrome (Figs 19.1–19.6, Box 19.1), which if not adequately treated can easily result in the loss of the extremity involved.
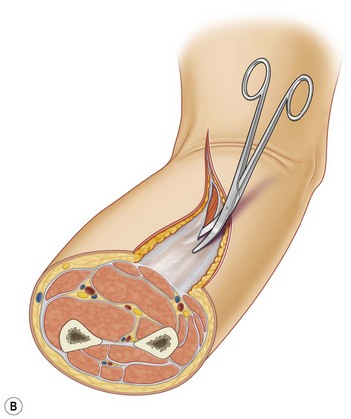
One of the most common devices which allow a precise measurement of compartment pressure is the Stryker Set®. This Device is assembled by a monitoring unit and a syringe with 3 mL of saline solution. After an accurate identification of the muscular compartment we want to check, we proceed with a simple injection of the 0.3 mL to re-equilibrate the pressure with interstitial fluids, and then we get the compartment pressure on the monitoring unit. For measurements exceeding 35–40 mmHg, it is necessary to adopt escharotomy-fasciotomy. In all these cases, it is necessary to perform full-thickness decompressive escharotomy in the very first hours in order to restore normal blood flow.19,20
This procedure must involve all the burned tissues, skin, fat, and fasciae, until release of the compartment is observed. These escharotomies should follow the recommended procedures and be performed before the initial signs of either vascular deficit (frozen hand) or neurological deficits occur. Escharotomies can be performed with an electrosurgical knife or a scalpel, ensuring complete release of the muscles (Fig. 19.4B, 19.5).
When treating the upper extremity, particular attention should be paid to the blood flow through the cephalic and basilic vein and superficial branch of the radial nerve at the level of the wrist. In the most serious cases, it may be necessary to perform complete decompression of the carpal tunnel (Figs 19.1, 19.2).
Escharotomy of the lower limb must be performed avoiding tibial anterior nerve, the medial saphenous vein and nerve, the tibial posterior nerve, the fibular nerve, and the external saphenous nerve (Figs 19.3, 19.4, 19.6).21
Surgery of the burn areas should be performed early, since late recovery leads to a greater incidence of hypertrophic scars, weaker tissues, and delays in prompt mobilization of the extremities.22,23
Related trauma
The same considerations apply in the case of exposed bone, which should also be covered as early as possible, even though resistance to exposure is much higher and more easily resolved than in the case of tendons. Coverage can be performed at a second stage utilizing rotation or free flaps.24
Patient selection
The results of sequelae surgery can be extremely heterogeneous, ranging from scar contraction resolution at the joint level to vascular, nerve, and tendon reconstruction. The main objective of sequelae surgery in these localized areas is to re-establish the extremity’s proper functionality, considering that normal walking, physical autonomy, and self-dependence at work depend on the complete rehabilitation of the areas affected.25
Treatment/surgical technique
With regard to sequelae surgery, the extremities have some unique characteristics, e.g., the majority of scar retractions lead to disabilities, and localized burn-related joint blockages may occur.12
Acute-phase surgery: skin grafting
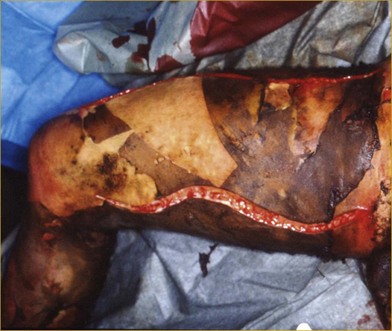
Debridement should be performed up to the vital tissues, after careful removal of all necrotic skin, paying special attention to the flexion-extension areas and the axillary cavity areas. In these areas the necrotomy is more complex to perform. In deep or electric burns, it is advisable to reach up to the muscular fascia in order to avoid the risk of probable but undesirable liponecrosis. Unlike skin, the viable fat underneath is an unstable base for implanting free skin grafts.26–29
During debridement of more superficially burned areas that do however require surgical treatment, instruments like VersaJet® provide useful support as they minimize the waste of vital tissue and also permit careful debridement in nonflat surfaces, as the extremities are.27
Skin grafts must be checked after 4–5 days and treated until full recovery is completed.