9 Essentials of Dermatology for Plastic Surgeons Abstract The skin is the body’s largest organ. It is organized in layers that contain different kinds of cells and structures, which can give rise to a multitude of lesions of varying characteristics. Is it important for plastic surgeons to have a basic knowledge of the clinical features and management of the most common skin lesions, especially those with potential for malignant degeneration and amenable to surgical treatment. Additionally, a number of lesions, including premalignant and some malignant ones, can be treated with nonsurgical modalities, which generally result in less morbidity and which plastic surgeons should be familiar with to avoid overindication of surgical treatment in some cases. Nonmelanoma skin cancers, namely basal cell carcinoma and squamous cell carcinoma, are the most frequent cancers in humans. Their treatment is based mainly on surgical excision and reconstruction (when required), making them an important part of the collaboration between dermatologists and plastic surgeons in a number of institutions. Proper assessment of risk factors is paramount in the therapeutic approach and posttreatment follow-up of a patient with skin cancer. Keywords: basal cell carcinoma, immunosuppression, Mohs micrographic surgery, skin cancer, squamous cell carcinoma, surgical excision The skin is the body’s largest organ, accounting for approximately 15% of the total body weight. It is a dynamic structure, with constant cellular replacement. It is composed of two main layers, the epidermis and the dermis, each of which contains different strata. Adnexal structures, including glands and hair follicles, are embedded within these layers. The skin performs numerous functions, the main ones being to serve as the interphase in our interaction with the external environment; to provide a protective barrier against various forces (mechanical, chemical, thermal), ultraviolet (UV) radiation, and microorganisms; to synthesize vitamin D; and to facilitate thermoregulation. According to coloration and tanning pattern, the skin is classified into six different phototypes ( The most superficial layer of the skin, the epidermis is organized into five strata ( Deep to the epidermis lies a mesoderm-derived layer called the dermis, which makes up for approximately 90% of the skin’s thickness and provides its tensile strength, elasticity, and healing abilities. The dermis is divided into papillary (superficial) and reticular (deep) layers. Importantly, the regenerative properties of the skin depend on the integrity of the papillary dermis—when this layer is affected, wounds often exhibit poor healing and scarring. The cellular compartment of the dermis includes fibroblasts, which produce extracellular matrix components (e.g., collagen, elastin, and structural proteoglycans), immune mast cells, and histiocytes. Table 9.1 Fitzpatrick’s classification of skin phototypes
9.1 Structure and Function of the Skin
 Table 9.1).
Table 9.1).
9.1.1 Epidermis
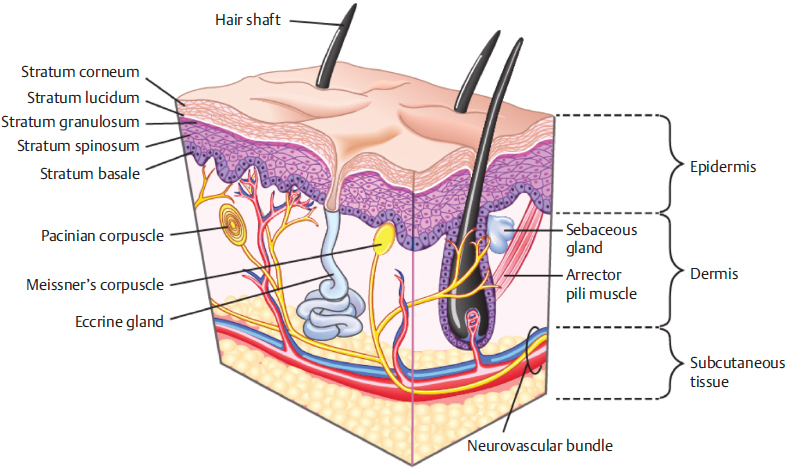
 Fig. 9.1). The epidermis contains two kinds of cells: keratinocytes and nonkeratinocytes. The stratum basale or germinativum contains a pool of proliferative cells that divide and become keratinocytes, which undergo progressive differentiation as they ascend through the stratum espinosum, granulosum, lucidum, and corneum until they are completely detached from the skin’s surface. Nonkeratinocytic cells reside in the basal layer and have special functions, namely antigen presentation (Langerhans’cells); pigmentation and UV protection (melanocytes); and touch sensation (Merkel’s cells).
Fig. 9.1). The epidermis contains two kinds of cells: keratinocytes and nonkeratinocytes. The stratum basale or germinativum contains a pool of proliferative cells that divide and become keratinocytes, which undergo progressive differentiation as they ascend through the stratum espinosum, granulosum, lucidum, and corneum until they are completely detached from the skin’s surface. Nonkeratinocytic cells reside in the basal layer and have special functions, namely antigen presentation (Langerhans’cells); pigmentation and UV protection (melanocytes); and touch sensation (Merkel’s cells).
9.1.2 Dermis
Phototype | Skin color | Sun exposure |
I | White | Always burns, never tans |
II | White | Usually burns, tans minimally |
III | Beige | Burns moderately, tans gradually |
IV | Brown | Burns minimally, tans easily and moderately |
V | Dark brown | Rarely burns, tans very easily |
VI | Black |
Fig. 9.1 Cross-sectional anatomy of the skin. (Reproduced from Papel, Facial Plastic and Reconstructive Surgery, 4th edition, ©2016, Thieme Publishers, New York.)
The dermal–epidermal junction (DEJ) is lined entirely by a basement membrane, which consists of a specialized aggregation of attachment molecules that act both as a barrier and as communicating channels between the two layers. Abnormalities in the DEJ result in rare conditions, such as bullous pemphigoid and epidermolysis bullosa. Also, with aging, the DEJ tends to become flatter, a phenomenon partly responsible for some of the distinctive features of aged skin.
9.1.3 Subcutaneous Layer
Also called the hypodermis, the subcutaneous layer lies beneath the dermis and is composed mainly of adipose tissue. Blood vessels and nerves running in this plane branch out to supply the overlying dermal plexuses (see Chapter 15).
9.1.4 Skin Appendages
Skin appendages include hair follicles, nail units, sweat glands, and sebaceous glands. Although these structures are all derived from embryonic epidermis, they reside in and are supported by the dermis. Sebaceous glands are present throughout the skin except on the palms of the hands and the soles of the feet. They produce sebum that drains into the hair follicles. While small in children, sebaceous glands enlarge and become active at puberty. Apocrine sweat glands reside in the areolae, axillae, inframammary folds, pubis, and anogenital region. Like sebaceous glands, apocrine sweat glands do not become active until puberty. Eccrine sweat glands (true sweat glands) are distributed throughout the whole body, with their highest density found on the palms and soles.
9.2 Basic Lesions of the Skin
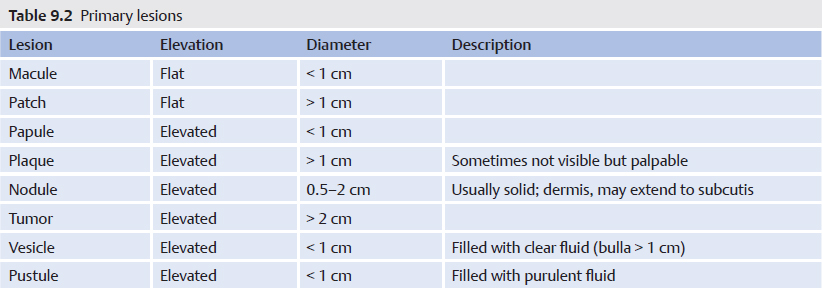
The skin is an area in which a myriad of different lesions can appear. This chapter summarizes the most elemental and common ones ( Table 9.2 and
Table 9.2 and  Table 9.3).
Table 9.3).
Lesion | Description |
Crust | Dried remains of serum, blood, pus, or exudate |
Scale | Accumulation of desquamating layers of stratum corneum |
Ulceration | Full-thickness loss of the epithelium (erosion loss of superficial layers) |
Atrophy | Thinning of the skin (epidermis atrophy thin and wrinkled, dermis atrophy depressed skin) |
9.3 Skin Biopsies
Biopsy specimens are a fundamental part of the diagnostic approach of any skin lesion. Depending on the lesion’s characteristics and suspected diagnosis, different techniques may be used. In order to optimize the result, the selected technique must ensure that sufficient tissue from the area of interest (where the abnormalities are expected) is obtained, while producing the least discomfort and cosmetic disturbance.
9.3.1 Shave Biopsy
Shaving is a simple method of obtaining samples of lesions suspected of affecting only the top layers of the skin (epidermis and superficial dermis). Using a scalpel parallel to the surface of the skin, the lesion is shaved off and the wound left to heal by secondary intention or treated by electrocauterization or topical hemostatic agents (e.g., 20% aluminum chloride, Monsel’s solution [20% ferric subsulfate], 35% trichloroacetic acid, or silver nitrate).
Deep shaving is a variant in which more tissue is obtained by changing the angle of the blade to reach the mid to deep dermis.
9.3.2 Punch Biopsy
This is a convenient and quick method of obtaining a representative full-thickness sample. It is performed with a circumferentially shaped blade with diameters ranging from 2 to 10 mm. Depending on the diameter of the wound, it may be left to heal by secondary intention or closed with a single stitch. Importantly, punch biopsies of < 3 mm may not obtain sufficient tissue to establish an accurate pathological diagnosis.
9.3.3 Incisional Biopsy
Incisional biopsies imply taking a sample of the lesion by standard surgical technique using a scalpel and, in most cases, closure with sutures. This is a good alternative when removal of the entire lesion is likely to leave a significant aesthetic defect. When possible, normal skin should be included in the specimen to allow for comparison on histological examination.
9.3.4 Excisional Biopsy
This technique removes the entire lesion en bloc and represents the ideal method because it allows examination of the whole lesion—its full architecture and depth as well as its relationship with the layers of the skin. In some cases, however, the morbidity of performing a complete excision precludes the use of this technique.
9.4 Nonsurgical Treatment Modalities
Over the last decade, nonsurgical therapies for benign and malignant disorders of the skin have gained increasing popularity, mainly due to their reduced morbidity and superior cosmetic results compared to surgery. Although these techniques are mostly indicated and performed by dermatologists, plastic surgeons need to be familiar with their indications and limitations so as to be able to inform patients on the full range of options for a given lesion and avoid unnecessary surgical resections where possible. Finally, patients treated with these modalities should be closely followed because a number of lesions may respond partially or not respond at all, in which case escalation to a more aggressive therapy is warranted.
9.4.1 Cryodestruction
Also referred to as cryotherapy or cryosurgery, this technique involves destruction of skin lesions by direct application of a cryogenic agent, such as liquid nitrogen. Cryodestruction is effective against actinic keratoses, viral warts, and seborrheic keratosis.
9.4.2 Photodynamic Therapy
This procedural field therapy consists of the application of a topical photosensitizer (5-amin olevulinic acid or methyl aminolevulinate) followed by irradiation with a light source, which induces a phototoxic reaction that destroys abnormal cells. It is effective in the management of multiple actinic keratoses and squamous cell carcinoma (SCC) in situ, as well as some types of basal cell carcinoma (BCC). Secondary effects during and shortly after the application of light include erythema, edema, and a burning sensation, which can be quite painful in some cases. A new modality of photodynamic therapy, daylight–photodynamic therapy, uses exposure to natural daylight instead of an artificial light source.
9.4.3 Laser CO2
This modality is described in Chapter 10.
9.4.4 Topical Treatments
Topical 5-Fluorouracil
This chemotherapeutic agent is a pyrimidine analogue, which acts by inhibiting thymidylate synthase, thus blocking DNA and RNA formation, especially in rapidly dividing cells. 5-Fluorouracil (5-FU) works best as a field treatment for actinic keratoses and SCC in situ, but it is relatively ineffective for BCC. Signs indicative of therapeutic success following application of 5-FU include inflammation, erythema, and erosions in dysplastic lesions. Conversely, undesired effects of this therapy include contact irritant or allergic dermatitis, phototoxicity, and scarring from ulceration. Patients may also experience pain, pruritus, and burning at the site of application.
Imiquimod
Imiquimod (Aldara, Medicis Pharmaceutical or Zyclara, Valeant Pharmaceuticals) is an immune response modifier with antitumoral and antiviral properties, which has shown to be effective in the treatment of genital warts, superficial BCC, and actinic keratosis, among others. Common side effects include skin irritation and erythema. In addition, although rare, flu-like symptoms and lymphadenopathies have also been reported following treatment with this product.
Topical Diclofenac
This nonsteroidal anti-inflammatory drug acts by inhibiting cyclooxygenase-2 (COX-2), which results in a reduction of prostaglandin synthesis and ultimately avoids suppression of the immune system. It is effective as a field treatment for actinic keratoses, but lesions slowly recur upon interruption of application. The most common reported adverse events (> 1%) are reactions at the site of application and irritant, allergic, or phototoxic contact dermatitis.
Ingenol Mebutate
Ingenol mebutate is a new topical therapy, the main advantage of which is the short treatment period (2–3 days) compared with the aforementioned methods. Its dual mechanism of action includes a chemoablative effect, which induces rapid cellular necrosis within a few hours of application and a long-lasting immunostimulatory effect that eliminates residual dysplastic cells through a neutrophil-mediated, antibody-dependent, cellular cytotoxic effect. This therapy is most effective against actinic keratoses and is currently under investigation for SCC in situ and BCC. The most common side effects include erythema, scaling, and crusting.
Deep Chemical Peels
The application of a chemical agent to the skin causes controlled destruction of the epidermis, with or without the dermis, leading to exfoliation and removal of superficial lesions, followed by regeneration of new epidermal and dermal tissues. Indications for chemical peeling include pigmentation disorders, superficial acne scars, ageing skin changes, and benign epidermal growths, such as seborrheic keratosis, actinic keratosis, sebaceous hyperplasia, warts, and milia. According to depth of penetration, chemical peels are classified as superficial, medium, and deep. Deep peels are performed with phenol-based solutions.
Peels may be complicated by hyperor hypopigmentation, erythema, hypertrophic scarring and keloids, infections, and milia formation. In addition, deep peels may produce cutaneous atrophy, cardiac arrhythmias, and, very rarely, toxic shock syndrome. Contraindications are the presence of active cutaneous bacterial, viral, or fungal infection; a tendency to keloid formation; facial dermatitis; and patients taking photosensitizing medications or those with unrealistic expectations.
9.5 Benign Skin Tumors
Benign skin tumors include an extensive list of lesions of varying appearances, the full review of which is beyond the scope of this chapter. Instead, the focus here is on the most frequent tumors and on those of clinical interest to plastic surgeons.
There is currently no formal classification for benign lesions, which sometimes hampers communication and learning.
9.5.1 Epidermal Lesions
Seborrheic Keratosis
Formerly known as senile warts, seborrheic keratoses (SKs) are one of the most common lesions found in the adult and elderly populations. They are benign, sharply demarcated lesions, often with a waxy or verrucous, “stuck-on” appearance and may develop anywhere on the body. They are usually asymptomatic, but some patients may complain of itching. SKs vary in size and clinical appearance, ranging from a few millimeters to centimeters and can present as macular, papular, or verrucous lesions with a tan to black coloration. Moreover, some SKs may simulate melanocytic neoplasms. SKs are mostly removed for cosmetic reasons by shave excision, curettage, and cryotherapy. The rarely occurring sign of Leser–Trélat is associated with an internal malignancy and involves the sudden appearance of multiple SKs.
Keratinocytic Epidermal Nevi (or Verrucous Nevus)
Epidermal nevi can be divided into organoid nevi (e.g., nevus sebaceous) and nonorganoid (keratinocytic) nevi. Keratinocytic epidermal nevi (frequently referred to simply as epidermal nevi) are benign, congenital tan to brown warty papules or plaques. They are either present at birth or become evident during early childhood and usually adopt a linear configuration on the extremities, although they may occur anywhere on the body. Histologically, epidermal nevi show hyperplasia of epidermal structures. They can be treated by surgical excision or laser therapy and other nonsurgical modalities. The epidermal nevus syndrome represents a sporadic association of epidermal nevi (usually extensive) with abnormalities in other organ systems.
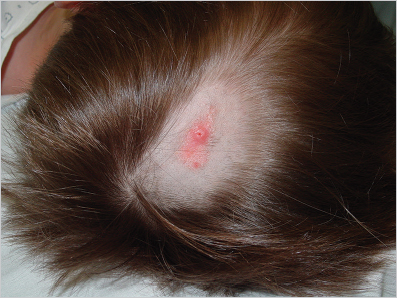
Nevus Sebaceous (Nevus of Jadassohn)
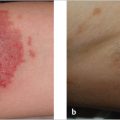
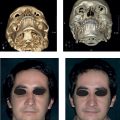
This tumor consists of a hamartoma containing a combination of abnormalities of different skin components (epidermis, hair follicles, sebaceous and apocrine glands). It is usually located on the scalp and other areas of the head and neck. Although it may not be readily noticed, in most cases nevus sebaceous is present at birth or develops during early child hood, presenting as a small, flat, hairless, well-circumscribed yellowish linear or oval lesion. As the child grows, the lesion expands proportionately until puberty, when, as a result of androgen hormones, it becomes a waxy, verrucous, thick plaque ( Fig. 9.2). An episode of local trauma may also trigger this change. Lastly, usually during adulthood, different tumors begin to appear within the lesion. Although most of these are benign (e.g., trichoblastoma, syringocystadenoma papilliferum and trichilemmoma), malignant degeneration into BCC and less frequently SCC can also occur in a small number of patients (< 2.5%). Therefore, nevus sebaceous is a lesion that should be removed surgically. Although there is no real indication during childhood, excision before adolescence is strongly advocated because of its increased risk of malignant transformation and for cosmetic reasons, because these lesions are often located in aesthetic areas and, as already mentioned, they tend to grow during this period of age. Finally, the appearance of multiple nevi sebaceous may be associated with cerebral, ocular, and skeletal abnormalities as part of the epidermal nevus syndrome.
Fig. 9.2). An episode of local trauma may also trigger this change. Lastly, usually during adulthood, different tumors begin to appear within the lesion. Although most of these are benign (e.g., trichoblastoma, syringocystadenoma papilliferum and trichilemmoma), malignant degeneration into BCC and less frequently SCC can also occur in a small number of patients (< 2.5%). Therefore, nevus sebaceous is a lesion that should be removed surgically. Although there is no real indication during childhood, excision before adolescence is strongly advocated because of its increased risk of malignant transformation and for cosmetic reasons, because these lesions are often located in aesthetic areas and, as already mentioned, they tend to grow during this period of age. Finally, the appearance of multiple nevi sebaceous may be associated with cerebral, ocular, and skeletal abnormalities as part of the epidermal nevus syndrome.
Acrochordon
These are benign, small, pedunculated, skin-colored or brownish papules, often located in folds. They have been associated with diabetes and an abnormal lipid profile. Indications for removal of these lesions are purely aesthetic. Shaving is the preferred method of excision.
9.5.2 Melanocytic Lesions (Usually Pigmented)
Ephelides or Freckles
Frequently seen in children and fair-skinned people, freckles are small, pigmented, uniform macules, appearing in sun-exposed areas that typically fade during winter months. Histologically, these lesions show a normal number of melanocytes with increased activity leading to the accumulation of melanin in basal keratinocytes.
Café au Lait Macules
Café au lait macules are sharply demarcated pale brown macules >n 5 mm in size. They are usually present at birth or appear shortly after. Isolated lesions have no clinical significance; however, multiple lesions (> 5) may be associated with underlying conditions, such as neurofibromatosis and other genetic disorders. Histologically these lesions show increased melanogenesis due to an excessive number of melanosomes, with a normal number of melanocytes.
Lentigines
Lentigines are acquired pigmented lesions, larger and better defined than freckles. The most common form, solar lentigines, arise in the middle-aged population and are induced by UV light; thus their preference for sun-exposed areas like the face and hands. Normal solar lentigines should be distinguished from lentigo maligna and lentigo maligna melanoma. Lesions that have appeared recently, showing different colors in their surface or with irregular borders, should be evaluated by a dermatologist to rule out malignancy.
Histologically there is a localized proliferation of melanocytes, specially at the tips of elongated rete ridges.
Nevi
The word nevus (pl. nevi) describes merely a cluster of cells. In order to classify nevi, attention should be paid to the type of cells contained within the lesion as well as their location within the layers of the skin.
Melanocytic Nevus
Melanocytic nevi are the most frequent melanocytic tumors. They are acquired lesions consisting of a concentration of nevus cells (slightly altered melanocytes). Clinically, they appear as round macules or flat papules, homogeneously colored, with regular borders and a diameter < 6 mm. According to where the nests of cells are located, melanocytic nevi are classified as junctional (at the epidermal–dermal junction), intradermal (dermis), and compound (elements of both junctional and intradermal). Junctional nevi usually present as flat brown macules, whereas intradermal nevi appear as fleshy, dome-shaped, skin-color to dark brown papules. The number of common melanocytic nevi depends on age (they usually develop at puberty), skin phototype (higher incidence in Caucasians), genetic background, immunosuppression, and UV light exposure. Contrary to common belief, nevi are rarely premalignant, and, in fact, melanoma typically arises as a new lesion rather than developing from preexisting ones.
Dysplastic and Atypical Nevus
The terms dysplastic nevus and atypical nevus are frequently treated as synonymous; nevertheless, such equivalency is incorrect from a basic conceptual view as well as from a clinical standpoint. Atypia describes the clinical appearance of a nevus, whereas dysplasia refers to the presence of altered cells on histopathological analysis. Thus not all atypical nevi exhibit dysplasia on histological assessment, and, in turn, some common nevi can well be regarded as dysplastic following analysis. Atypia is based on the observation of one or more of the following clinical features, commonly summarized as ABCDE: asymmetry, border irregularity, color variability, diameter > 6 mm, evolution experiencing growth or changes. Conversely, dysplasia is classified as mild, moderate, or severe, depending on the degree of cellular alteration (cellular atypia).
Understanding the association between atypical nevus/dysplastic nevus and melanoma is fundamental for anyone involved in the diagnosis and management of these lesions. In the first place, as already stated, there is robust evidence showing that melanoma develops more frequently de novo rather than from a preexisting lesion, regardless of whether it was a common, dysplastic, or atypical nevus. Furthermore, studies of nevus-derived melanomas show similar proportions arising in dysplastic and common nevi. Secondly, despite widespread belief, most dysplastic nevi in the general population do not progress to melanoma but remain stable or regress. However, the presence of dysplastic nevi does constitute an important marker for an increased risk of melanoma, and in consequence any patient with such a lesion should be referred to a skin specialist for a complete physical examination and close follow-up. Regarding management, the recommendations are not to excise every atypical (irregular) nevus, but only those clinically suspicious for melanoma. Once removed, those showing mild or moderate dysplasia may be observed like common nevi regardless of margin status, whereas reexcision is indicated for those with severe dysplasia or when the pathologist cannot fully distinguish between dysplastic nevus and melanoma.
Blue Nevus
Blue nevi are well-circumscribed, homogeneous, blue-gray to blue-black round papules, often < 1 cm in size. Histologically, blue nevi are composed of a collection of melanocytes in the dermis. Conservative treatment is recommended for this tumor unless changes are observed (e.g., size, color, symptoms, etc.), in which case excision removing the superficial and deep dermis all the way to the subcutaneous tissue is indicated. Of note, primary nodular or metastatic melanoma may clinically and dermoscopically mimic blue nevus.
Halo Nevus
Common benign, asymptomatic lesions characterized by a central melanocytic nevus surrounded by a peripheral halo of hypopigmentation. With time, the central nevus usually disappears. These lesions do not require any treatment, although they should be reviewed in cases of morphological changes or symptoms.
Spitz’s Nevus
This is a benign, usually acquired melanocytic lesion, frequently reported in children and young people and one of the main differential diagnoses of melanoma. Spitz nevus appears predominantly in the head and neck and presents as a solitary, dome-shaped, round to oval, reddish to dark brown papule, as large as 1 cm in diameter. Although Spitz’s nevus has specific dermoscopic features, there are cases in which neither dermoscopy nor histopathology can reliably distinguish a Spitz’s nevus from melanoma. Surgical excision is indicated for all spitzoid lesions appearing after puberty. Lesions without signs of atypia in patients younger than 12 years can be managed conservatively with digital dermoscopic monitoring.
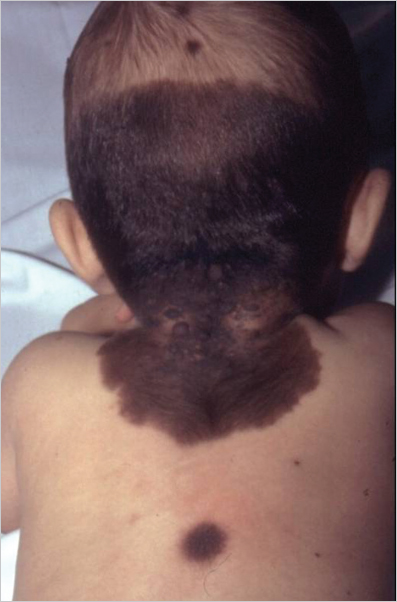
Congenital Melanocytic Nevus
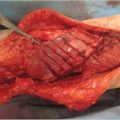
Congenital melanocytic nevi (CMN) appear at birth or shortly after. Based on size, CMN are classified as small (< 1.5 cm), intermediate (1.5–19.9 cm), large (> 20 cm), and giant (> 40 cm). They typically occur on the trunk, presenting initially as a flat, pigmented patch that changes over time, when darker macules and papules and sometimes hair begin to appear, giving the lesion an unsightly mottled and heterogeneous aspect ( Fig. 9.3). The three main concerns about CMN are its potential for malignant transformation into melanoma, its association with neurocutaneous melanosis, and its unaesthetic appearance. Regarding malignant degeneration, the risk of melanoma in CMN depends mostly on size—for small and medium lesions the risk is < 1% risk, whereas for large lesions it is < 5%. Importantly, most CMN-derived melanomas appear before 15 years of age. Neurocutaneous melanosis is associated with giant lesions and other factors, such as satellite nevi or multiple CMN, male sex, and head and neck and dorsal midline location. Neurocutaneous melanosis can affect different regions of the central nervous system and become life threatening in some cases.
Fig. 9.3). The three main concerns about CMN are its potential for malignant transformation into melanoma, its association with neurocutaneous melanosis, and its unaesthetic appearance. Regarding malignant degeneration, the risk of melanoma in CMN depends mostly on size—for small and medium lesions the risk is < 1% risk, whereas for large lesions it is < 5%. Importantly, most CMN-derived melanomas appear before 15 years of age. Neurocutaneous melanosis is associated with giant lesions and other factors, such as satellite nevi or multiple CMN, male sex, and head and neck and dorsal midline location. Neurocutaneous melanosis can affect different regions of the central nervous system and become life threatening in some cases.
Treatment of CMN, especially large and giant, is based on surgical excision, mainly to reduce the risk of malignancy and improve cosmesis. Although highly recommended, whether surgery reduces the incidence of malignant melanoma remains unproven. The unaesthetic appearance of CMN constitutes in many cases the main reason for surgical excision due to the significant psychological impact that these lesions cause, especially large ones. Surgery usually involves serial excision, tissue expansion, and flap coverage. Alternatives to surgical excision have been described, such as dermabrasion, curettage, electrosurgery, and lasers, which could be a good option when morbidity from surgical excision is unacceptable and for cases in which the main concern are the thick hairs growing on the surface of the lesion.
Nevus of Becker
Nevus of Becker typically appears on the shoulder and back of males (5:1) during puberty, presenting as a unilateral, gradually enlarging, and usually hypertrichotic melanocytic lesion (also considered as a hamartoma).
Nevus of Ota
Nevus of Ota is a unilateral, blue-brown facial patch most usually distributed along the territory of the first and second branches of the trigeminal nerve. This type of nevus is more common in Asians and dark-skinned races. Histologically it is characterized by the presence of elongated melanocytes scattered among collagen bundles of the reticular dermis. Treatment usually involves the use of laser therapy, namely Q-switched neodumium:ytrium-aluminum-garnet.
Nevus of Ito
Exhibits the same clinical and histological features as nevus of Ota, differing from it in its area of involvement (posterior shoulder and areas innervated by posterior supraclavicular and lateral cutaneous brachial nerves).
Mongolic Blue Spot (Congenital Dermal Melanocytes)
These lesions are more common in Asian and dark-skinned individuals, presenting as bluish-gray patches with indistinct margins on the lumbosacral area. They typically disappear by childhood and therefore require no treatment.
9.6 Fibrous Tumors
9.6.1 Dermatofibroma
Indurated minimally elevated to dome-shaped papule or nodule with a yellowish to reddish-brown surface that appears in adulthood, more commonly on the lower extremities. Dermatofibromas usually exhibit a positive dimple sign (apparent downward movement of the tumor when pressed at the edges).
9.6.2 Neurofibroma
Neurofibromas are asymptomatic skin-colored, protuberant to pedunculated, soft or rubbery papule-nodules, usually < 2 cm in size. Neurofibromas appear more commonly as solitary lesions; in the presence of multiple lesions neurofibromatosis 1 or 2 must be ruled out. Regardless, they are usually treated by simple excision.
9.7 Miscellaneous
9.7.1 Epidermal Cyst
Usually but incorrectly called sebaceous cysts, epidermal cysts are a proliferation of epidermal cells within a circumscribed dermal space containing an accumulation of foul-smelling keratinous debris with a central pore. Epidermal cysts are located most often on the face, neck, and trunk, where they appear as a fluctuant, flesh colored nodule with a visible punctum (pore), which is usually a useful distinguishing feature from lipomas. They are mostly asymptomatic but may become inflamed or infected, resulting in pain and tenderness. Uninfected cysts can be excised with their surrounding capsule, whereas infected ones are treated by incision and drainage of any purulent material, followed by a course of antibiotics and excision of the remaining capsule/cyst once infection and inflammation have subsided (see Chapter 12 for further details).
Trichilemmal (pilar) cysts are epidermal cysts of the scalp and are managed as already described.
9.7.2 Mucocele
Mucoceles are the most common lesions of the oral cavity and result from mechanical trauma to minor salivary glands. They appear most frequently in the lower lip and cheek mucosa and may be seen at any age, with a concentrated incidence around the second decade of life. Clinically, mucoceles are characterized by a painless, semitranslucent, soft submucosal nodule with multiple cycles of swelling, rupturing, and refilling. Surgical excision is recommended in most cases.
9.7.3 Mucous Cyst
Mucous cysts (also called myxoid cysts) are soft, shiny, viscous-filled nodules appearing in the distal interphalangeal joint in relation to the presence of an osteophyte. In some cases the cyst may compress the nail matrix, resulting in nail plate depression and grooves. Treatment of mucous cysts involves surgical excision, including removal of the underlying osteophyte. Even though they usually appear on one side of the joint, the opposite should be explored during surgery to rule out the presence of occult cysts.
9.7.4 Angiomas
Eruptive angiomas, also known as ruby spots or senile or cherry hemangiomas, are benign tumors of capillary vessels. They appear as isolated or multiple, bright red, dome-shaped to polypoid, sharply demarcated papules up to several millimeters in diameter.
9.7.5 Milium Cysts
Benign, small, white, superficial cysts (< 3 mm in size) appearing most commonly on the face. They are extracted using a sharp instrument, such as a 14-gauge needle or a no. 11 scalpel blade.
9.7.6 Dermoid Cysts
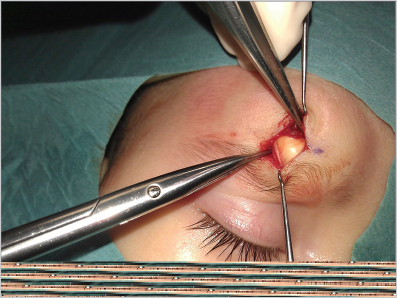
These are congenital lesions developing from the entrapment of endodermal and mesodermal remnants along the lines of embryonic fusion. With time they tend to become progressively larger due to accumulation of debris within the cyst. They are most commonly seen along the supraorbital ridge, lateral brow, or nasal midline ( Fig. 9.4). Lesions along the nasal midline should be investigated by magnetic resonance and/or computed tomographic scan prior to removal in order to rule out intracranial extension as well as glioma, meningocele, and encephalocele. Surgical excision is the recommended treatment.
Fig. 9.4). Lesions along the nasal midline should be investigated by magnetic resonance and/or computed tomographic scan prior to removal in order to rule out intracranial extension as well as glioma, meningocele, and encephalocele. Surgical excision is the recommended treatment.
9.7.7 Pilomatrixoma
These are small, benign tumors derived from the hair matrix, most commonly seen in the head and neck and upper limbs of younger patients. Clinically, they appear as a flesh-colored, white, or bluish papule or nodule, which is firm on palpation due to calcification. Pilomatrixomas are usually solitary lesions; multiple ones have been associated with myotonic dystrophy and Gardner’s syndrome. Despite its benign nature, locally aggressive and even metastatic lesions have been reported, which is why surgical excision is the treatment of choice.