Fig. 1.1
Human embryo at stage of limb initiation and presumed Hox positioning. (a) Depiction of an emerging upper limb bud (boxed) in Carnegie stage 12 embryo. (b) Hox genes establish upper limb position and polarity. Courtesy of K.C. Oberg and Loma Linda University
By day 37 of development (Carnegie stage 16), the distal portion of the limb can be recognized as a handplate. At the same time there is progressive mesodermal condensation along the proximodistal axis forming the skeletal elements of the limb. By day 56 the major morphologic features of the limb are complete.
Limb Initiation
After the upper limb fields have been specified, induction of the limb bud occurs. The cells of the LPM located within the limb fields maintain active proliferation, while non-limb field LPM begins to divide more slowly [5]. Initially Fgf10 is expressed broadly along the LPM, but just before the limb emerges, the domain of Fgf10 expression becomes restricted to the limb fields. In chicken, the expression of Tbx5 and Wnt2b in the LPM cells of the limb field are responsible for the induction of Fgf10 in the presumptive limb (Fig. 1.2) [6–8]. Recent studies suggest that Tbx5 expression can be induced and regulated by Hox transcription factors, suggesting a role for Hox genes in both positioning limb fields and initiating limb outgrowth [9]. Fgf10 through its receptor FgfrIIa has been shown to induce Wnt3 and Wnt3a in prospective mouse and chick limb ectoderm, respectively. Concurrently, Bone Morphogenetic Protein (Bmp) signaling in the ventral ectoderm induces β-catenin competency in cells of the presumptive apical ectodermal ridge (AER) at the dorsal–ventral boundary [10, 11]. In turn, Wnt3 or Wnt3a induces Fgf8 in a Wnt/β-catenin-dependent manner in the precursor cells of the AER [6, 12]. Fgf8 secreted from the recently formed AER maintains the expression of Fgf10 in the mesoderm, establishing a positive regulatory loop that maintains proximal–distal growth [6, 12].
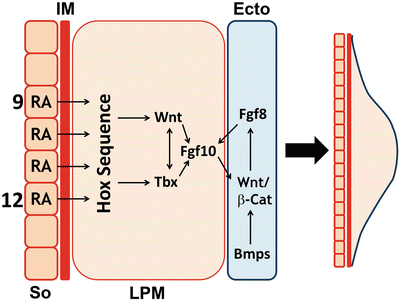

Fig. 1.2
Molecular pathways involved in limb induction. Depiction of the tissues involved in the initiation of the right upper limb bud emerging from lateral plate mesoderm (LPM) at somite (So) levels 9-12. Molecular interactions between LPM and ectoderm (Ecto) are also illustrated. IM-intermediate mesoderm. Courtesy of K.C. Oberg and Loma Linda University
Another signaling molecule that is fundamental to the induction of the limb bud appears to be retinoic acid (RA), the active metabolite of vitamin A. This molecule is produced in the somites of the embryo by the enzyme Raldh2 [13–15]. RA restricts the early expression of Fgf8 within the heart field, which, in turn, permits the expression of Tbx5 in the limb field to initiate forelimb development [16, 17]. Furthermore, RA has been shown to regulate the expression of Hox genes both in vitro and in vivo, which may contribute to limb field induction and/or positioning (see Fig. 1.2) [18, 19].
Signaling Centers
Between the fourth and eighth weeks of development, the limb bud undergoes growth and differentiation to transform it into a fully patterned limb. This process can be described in terms of three coordinate axes: proximal–distal (P–D), anterior–posterior or radial–ulnar (A–P/R–U), and dorsal–ventral (D–V) modulated by three signaling centers [20].
Along the P–D axis, the AER appears as thickened ectoderm overlying the distal edge of the limb bud [21]. The AER is the signaling center that regulates the P–D growth and Fgfs are the signaling molecules that accomplish its function. Excision of the AER in chicken embryos at different stages of limb development results in limb truncations in a progressive fashion; the later the AER removal, the more distal the resulting truncation [22].
The signaling center for the A–P/R–U axis is the zone of polarizing activity (ZPA), a cluster of mesodermal cells located at the distal posterior (ulnar) margin. The ZPA directs A–P/R–U patterning and Shh is the signaling molecule that mediates its function. Both mice (Shh knock-out) that lack Shh function or mutant chickens (Oligozeugodactyly—Ozd mutants) that fail to have limb-specific Shh expression show marked loss of posterior (ulnar) elements [23, 24].
Dorsal non-AER ectoderm directs D–V patterning with Wnt7a as the signaling molecule that promotes dorsalization. Excision and rotation of the dorsal ectoderm results in the formation of dorsal structures within the ventral aspect of the limb [25].
Patterned Development Along Coordinate Axes
Pattern formation is a process by which the cells are sequentially specified, determined, and then differentiated to form the morphological structures of the limb. In this section we will focus on how the process of patterning is accomplished along each axis as directed by the signaling centers and the associated molecular pathways, recognizing that the molecular cascades of these three axes are operating concurrently and integrated together like a fine-tuned instrument.
Proximal–Distal Patterning (P–D)
The upper limb can be divided into three different segments along the P–D axis (Fig. 1.3): (1) the proximal segment or stylopod where the skeletal elements of the humerus develop; (2) the intermediate segment or zeugopod where the radius and ulna form; and (3) the distal segment or autopod where the carpals, metacarpals, and digits form.
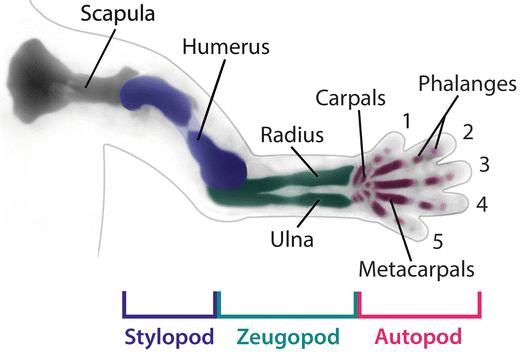

Fig. 1.3
Limb elements. The upper limb consists of a limb girdle or shoulder, and three limb segments known as the stylopod (humerus – colored blue), the zeugopod, which includes the radius and ulna (colored green) and the autopod or handplate (colored magenta). Courtesy of K.C. Oberg and Loma Linda University
Patterning along the P–D axis begins during limb initiation with the formation of the AER, stratified ectoderm at the distal dorsal–ventral boundary of the developing limb bud. The AER secretes fibroblasts growth factors (Fgfs), the molecules primarily responsible for P–D patterning. Fgf8 is the first and functionally most important Fgf secreted from the AER during induction and maintained until the AER regresses, when the drafts of the last phalanges are formed. Fgf4, Fgf9, and Fgf17 are activated sequentially in the posterior AER and expand to the anterior aspect as the limb develops [26, 27]. Classical experiments in chick embryos showed that AER removal abated distal limb outgrowth and resulted in truncations that corresponded to the timing of AER removal; in other words, the later the AER removal, the more distal the structures that were present [22]. Moreover, FGF-soaked beads were able to restore limb bud outgrowth and patterning after AER removal, indicating that Fgfs were the functional signaling factors of the AER [28, 29].
Among the different Fgfs expressed, Fgf8 is thought to be the main AER signal, while Fgf4, Fgf9, or Fgf17 are considered secondary or redundant [30, 31]. This concept is supported from experiments with Fgf8 knock-out mice that showed smaller AERs, delayed limb bud outgrowth, and loss of some skeletal elements [26, 32]. In contrast, knock-out mice for Fgf4, Fgf9, and/or Fgf17 did not develop limb anomalies. Interestingly, Fgf4 expression in Fgf8 knock-out mice was up-regulated, suggesting that redundant expression may have lessened the phenotype of these mutants. This was confirmed by the removal of both Fgf4 and Fgf8 that resulted in a worse phenotype with notably smaller limb buds [32, 33].
Several models have been proposed for P–D patterning. The progress zone model proposes that mesenchymal cell fate is determined by the length of time spent under the direct influence of the AER in a proliferative region called the progress zone (PZ) [34, 35]. The early specification model [36] postulates that the P–D identities are specified early and the different progenitor pools expand sequentially as the limb grows. The differentiation front model suggests that the AER maintains mesenchymal cells in an undifferentiated state; as the limb expands, the cells that are no longer under the influence of the AER differentiate [37].
However, the accumulating evidence supports an alternative model. The two signal model [30] proposes that two opposing signals pattern the limb along the P–D axis: RA emanating from the flank will specify a proximal fate, while Fgfs from the AER will specify a distal fate (Fig. 1.4a) [38, 39]. In somites, Raldh2 oxidizes Retinol to form RA which can act locally in the proximal limb buds to promote the expression of Meis1 and Meis2. The expression of Meis1/2 defines the proximal limb segment and where the humerus (stylopod) will develop. Distally, Fgf signaling induces 5′Hoxa genes (Hoxa11, Hoxa13) and limits distal Meis1/2 expression. Although the mechanism for this repression is not fully understood, it is known that Fgf8 signaling induces the expression of Cyp26b1 in the distal mesenchyme of the limb bud; the product of this gene oxidizes RA into a non-active form, thus clearing the distal region of active RA (see Fig. 1.4c) [40]. Some have questioned RA role as a proximalizing agent [16], and further investigations are warranted to clarify whether RA or another factor influenced by RA is the proximalizing signal.
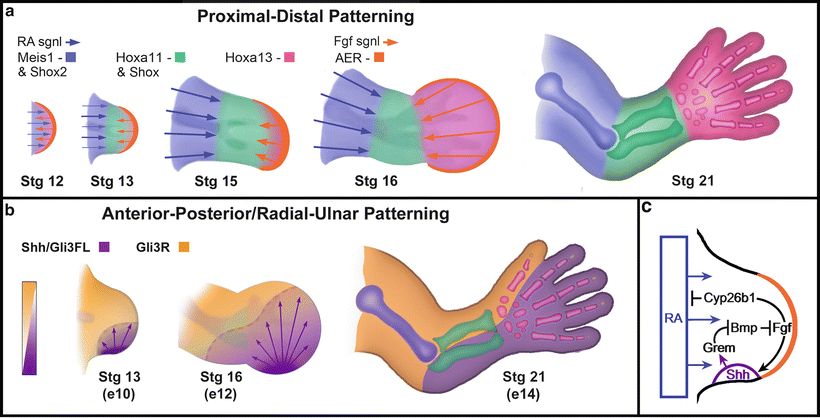

Fig. 1.4
Molecular pathways regulating proximal-distal & anterior-posterior/radial-ulnar axes. (a) Progressive segment specification along the proximal-distal axis based on the two signal model with RA-related proximalizing signals countered by distalizing Fgf signals. Outgrowth separates the signals and an intermediate zone is formed. Mies1 and Shox2 are restricted to the stylopod, Hoxa11 and Shox are markers for the zeugopod, and Hoxa13 delimits the handplate boundaries. (b) Opposing gradients of Gli3 repressor (Gli3R) and Shh-maintained full length Gli3 activator (Gli3FL) establish boundaries between the radius and ulna in the zeugopod and the thumb and ulnar digits in the autopod or hand. c) Some of the molecular interactions that maintain and terminate Shh expression in the ZPA (→ indicates positive regulation, while –| indicates inhibition). Courtesy of K.C. Oberg and Loma Linda University
Anterior–Posterior/Radial–Ulnar Patterning (A–P/R–U)
The limb along the A–P/R–U axis is divided into two segments: the anterior (radial), comprises the thumb and radius, and the posterior (ulnar) with the ulna and ulnar digits (digits two through five) [41].
Patterned development along the A–P/R–U axis is controlled by the zone of polarizing activity (ZPA), a cluster of mesenchymal cells maintained in the distal posterior/ulnar aspect of the developing limb (see Fig. 1.4b). The ZPA was discovered in 1968 through grafting experiments in chick limb buds [42]. In these experiments, grafts from the distal posterior/ulnar mesenchyme were excised from one group of chicks and then inserted into the distal anterior/radial aspect of another group of chicks. The limbs that developed from these grafts demonstrated mirror image duplication of structures [42–44]. RA was found to be the first molecule that mimicked ZPA grafts when applied to the distal anterior/radial aspect of the limb bud [45–48]. Later it was shown that Shh was the molecule responsible for the phenotype induced by RA [49].
Shh is critical to the correct developmental pattern of the limb, particularly for the forearm (zeugopod) and hand (autopod). This is demonstrated in Shh knock-out mice, which have a correctly developed stylopod, a single skeletal element (radius) for the zeugopod, and a minimal autopod. In the upper limb, the autopod forms as a small cartilage condensation [23, 50], while in the lower limb, the autopod consists of a single digit with two phalanges [23, 50].
A–P/R–U polarity and subsequent Shh expression is initiated by axial Hox gene segmentation [51] followed by several other factors that contribute to Shh induction and the establishment of the ZPA. Hand2, expressed in the posterior/ulnar half of the limb bud, is required for Shh induction [52, 53] and has been shown to directly interact with limb-specific Shh regulatory region [54]. Similarly, distal Hoxd (Hoxd10-13) transcription factors also interact with the limb-specific Shh regulatory region, and evidence suggests that their initial phase of limb bud expression helps to localize Shh expression (Fig. 1.5) [55].
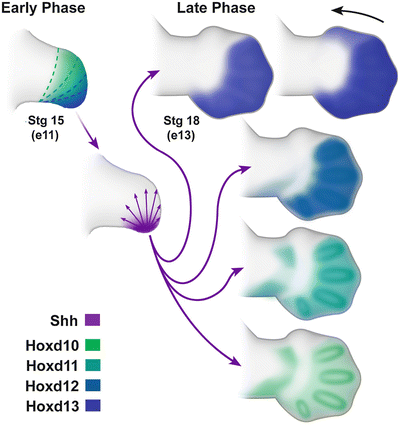

Fig. 1.5
Distal Hoxd genes are expressed in the limb bud in two phases. In the early phase, there is a nested collinear expression pattern. Dotted lines highlight the boundaries of expression, from broadest expression of Hoxd10 (green) to the most restricted of Hoxd13 (blue). In the late phase, Hoxd expressioin demonstrates quantitative colinearity with progressively more robust expression. Courtesy of K.C. Oberg and Loma Linda University
The first or initial phase of distal Hoxd expression in the limb bud occurs in a nested collinear (corresponding to their gene order) fashion along the anterior–posterior axis, with Hoxd10 exhibiting the broadest initial expression domain. The expression of each successively more distal Hoxd gene is nested within the previous gene’s expression domain (see Fig. 1.5). Hoxd13, the terminal transcription factor in the Hoxd cluster, has the most restricted expression domain within the distal posterior or ulnar aspect of the limb bud overlapping the ZPA. This first phase of distal Hoxd expression plays a role in localizing Shh expression and temporally corresponds to specification of the forearm or zeugopod.
The role of Shh in A–P/R–U axis patterning has been characterized largely through knock-out mice for members of the Gli protein family of transcription factors (Gli1, Gli2, and Gli3). Gli3 mutant mice are polydactylous without digit identity while the zeugopod is perfectly formed [56, 57]. Remarkably, the limbs of the double knock-out mice for both Gli3 and Shh were indistinguishable from the Gli3 mutant alone [58, 59], suggesting that the principal function of Shh is mediated through Gli3. Molecular studies demonstrated that Shh signaling prevents the posttranslational processing of full-length Gli3 protein into a short form, which functions as a strong repressor of Shh target genes.
Secreted Shh diffusing from the ZPA establishes a posterior to anterior concentration gradient. A complementary gradient of Gli3R forms with high levels of Gli3R in the anterior zone where Shh signaling is minimal (see Fig. 1.4b) [59]. In the absence of Shh, the level of Gli3R is uniform along the A–P/R–U axis and the elevated levels of Gli3R, unopposed by Shh, are accompanied by an increase in the apoptotic rate of the limb mesenchyme [58, 60]. Thus, the A–P/R–U gradient of Gli3R and its reciprocal full length Gli3 activator are responsible for conveying pattern information along this axis. However, it remains unclear whether the critical patterning signal is the absolute level of GLi3R or the relative levels between the repressor and the activator forms [58, 59]. Collectively, these data help to characterize the role of Shh in A–P/R–U patterning which, at least, in part, is to regulate the form and function of its transcription factor, Gli3.
Dorsal–Ventral Patterning (D–V)
Patterning along this axis is regulated by signals from the non-AER ectoderm that surrounds the limb mesenchyme. The dorsal and ventral areas are defined by the expression of two different genes: Wnt7a in the dorsal ectoderm and En-1 in the ventral ectoderm (Fig. 1.6). Wnt7a signaling defines the dorsal fate of the limb structures [61], while En-1 restricts Wnt7a expression to the dorsal ectoderm, preventing the dorsalization of ventral limb tissues [62, 63]. It is not yet known how Wnt7a is induced in the presumptive limb ectoderm; however, there is evidence that BMP and WNT canonical signaling are responsible for the induction of En-1 in the ventral ectoderm. Knock-out mice have further elaborated their functional roles. Wnt7a mutants have biventral limbs, while En-1 mutants have bi-dorsal limbs [64, 65]. Interestingly, double compound mutant mice for En-1 and Wnt7a display a biventral phenotype, suggesting that the default limb phenotype is ventral and establishing Wnt7a’s role as the dorsalizing signaling molecule of the limb’s D–V axis [64].
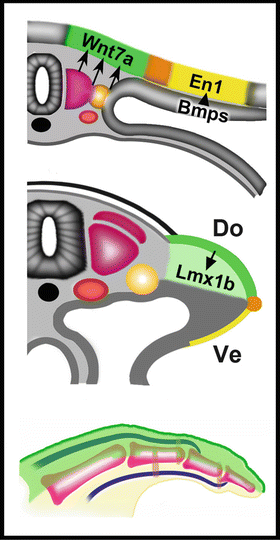

Fig. 1.6
Molecular pathways regulating the dorsal-ventral axis. From top to bottom, unknown factors in somites and/or intermediate mesoderm initiate Wnt7a expression in medial dorsal ectoderm. Bmps induce the expression of En1 in what will become the ventral ectoderm establishing the dorsal ventral boundary where the AER will form (orange). Wnt7a will induce Lmx1b in the underlying mesoderm to dorsalize developing tendons, joints and soft tissues. Courtesy of K.C. Oberg and Loma Linda University
Additional studies demonstrate that Wnt7a manifests its function through the induction of Lmx1b in the underlying dorsal mesoderm. Lmx1b function is both sufficient and necessary for the induction of dorsal fates. In chicken and mice, ventral Lmx1b expression led to bi-dorsal limbs, whereas its inactivation resulted in biventral limbs [61, 64, 66, 67].
Integration of Axis-Related Signaling
The three signaling centers coordinate patterned limb development through interactions between their molecular signaling cascades. One of the most studied interactions is the interaction between the ZPA and the AER. Shh signaling from the ZPA induces the expression of Gremlin in the adjacent mesenchyme that underlies the AER [26]. Gremlin is an antagonist of BMP signaling, repressing Bmp expression in the mesenchyme [68, 69]. Although Bmp signaling is needed in limb and AER induction [70, 71], mesenchymal BMP inhibits the expression of AER-associated Fgfs and increases mesenchymal cell death [70, 71]. Thus, Shh through Gremlin prevents these BMP-associated functions thereby maintaining Fgf expression. Correspondingly, Fgf8 secretion into the mesenchyme maintains Shh expression in the ZPA (through pathways that are not yet characterized) forming a positive feedback loop that supports continued limb growth and patterning. Termination of this reciprocal loop has been proposed as the mechanism that stops limb outgrowth once the appropriate size has been achieved [72].
Integration also occurs between other axes. Wnt7a knock-out mice shows a reduction in Shh expression [61] with a loss of the posterior digits (corresponding to the little finger). In chickens, elimination of the dorsal ectoderm of the limb showed similar results [73, 74]. These findings suggest that Wnt7a signaling from the dorsal ectoderm is capable of inducing or maintaining Shh expression in the ZPA [61]. Although the characterization of pathways that interconnect these three signaling centers is incomplete, it is intuitive that interaction between them is crucial for the proper development of a patterned limb.
Handplate Patterning
The handplate or autopod is the distal-most element of the limb and the last to form. It is composed of digits (fingers) and wrist bones. The axes-related pathways converge to form the most complicated, pattern-rich structures of limb development. Hoxa13, the terminal transcription factor of the Hoxa cluster, is confined to the handplate, demarcating its proximal boundary along the P–D axis (see Fig. 1.4a) [75, 76]. Concurrently, along the A–P/R–U axis, a second “late” Shh-regulated phase of distal Hoxd expression (that corresponds with digit formation) is generated that partially reverses their expression domains, i.e., reverse colinearity (see Fig. 1.5) [77]. More importantly, there is progressive expression intensity, with Hoxd13 exhibiting the most robust expression within the digits and Hoxd10 exhibiting the least intense expression, in what has been termed quantitative colinearity [78]. Along the D–V axis, expression of Lmx1b, the dorsalizing Wnt7a-mediated transcription factor, becomes restricted to dorsal tendons and joint-associated tissues (see Fig. 1.6) [79].
Establishing Digit Number
In addition to regulating the second phase of Hoxd gene expression in the limb bud, Shh-expressing ZPA cells also make a direct contribution to digit development. Fate mapping studies have demonstrated that descendants from Shh-expressing cells of the ZPA populate digit 5, 4 and half of digit 3. The cells of digit 5 have had the longest exposure to Shh and at higher levels, while the cells of the digit 2 are only affected by diffusion of Shh [80, 81]. Moreover, premature arrest in Shh expression causes a reduction in the number of digits corresponding to the stage and duration of arrest. With normal Shh levels, the order of condensation is d4, d2, d5, and d3, and with the premature arrest in shh, the loss follows a predictable order, where digit 3 is lost first, followed by d5, d2, and d4 [82]. Studies of digit duplication in chicken wings by Shh misexpression show that the most posterior/ulnar digits need higher Shh concentrations and longer exposure times than the more anterior digits [83].
Recent experiments in chicken show that Shh integrates both proliferation and specification of digit precursors and that Shh expression is controlled by cell proliferation [82, 84]. These data prompted two models to explain how digit morphology and number are achieved. The biphasic model suggests that an early phase specifies digit number and potential morphology and a second proliferative phase allows for digit growth and final morphologic determination [82]. The growth-morphogen model posits that both Shh concentration and exposure duration progressively expands the limb to specify digit number and morphology [84].
Although a Shh concentration gradient can account for some features of digit morphogenesis, it does not fully explain the repeating digit/interdigit pattern. Experiments out of Marian Ros’ laboratory found that compound gene deletions of Hoxa13 (the terminal Hoxa gene demarcating the handplate), Hoxd11-13 (the Shh-dependent Hox genes of the A–P/R–U axis), and Gli3 (the gene mediating Shh activity along the A–P/R–U axis) exposed an intrinsic self-organizing mechanism in mice involved in digit patterning [85]. Progressive reduction of the Hox gene dosage in the absence of Gli3 progressively increased digit numbers (up to 14 digits) that was not accompanied by a corresponding increase in handplate size; thus, the digits were increasingly thinner and shorter.
Alan Turing developed a mathematical diffusion-reaction model to account for repetitive self-organizing patterns, such as stripes or spots in animal skin and fur [86]. This model considers two molecules, an activator and inhibitor, which diffuse into a field of cells. The activator auto-up-regulates itself and up-regulates its own inhibitor. In contrast, the model’s inhibitor suppresses the activator and auto-inhibits its own expression (Fig. 1.7).
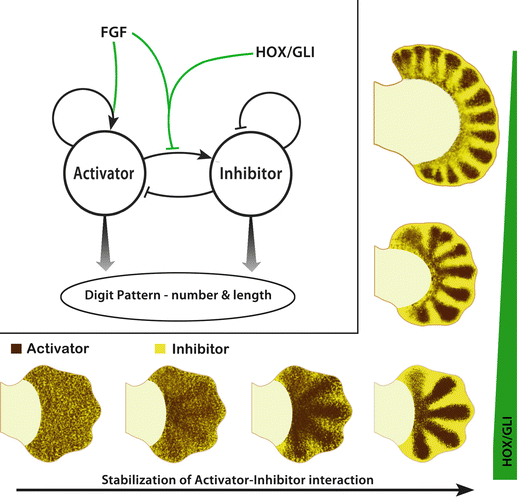

Fig. 1.7
Turing-like patterning in limbs. In the upper left-hand corner is a diagram of the diffusion-driven instability model with an activator and inhibitor modulated by FGF and HOX/GLI. In the model described by Sheth et al. (2012), FGF from the apical ectodermal ridge (AER) promotes a radial stripe pattern from this intrinsic self-organizing mechanism (ISOM) and ultimately regulates digit length, while FGF in concert with distal HOX and GLI transcription factors limit the number of digits. The bottom of the illustration has a series of handplates that show the rapid progression from fluctuating activator-inhibitor interaction (noise) to a stabilized 5-digit pattern. On the right, progressive loss of digit suppressing HOX/GLI transcription factors (green bar) causes an increase in the number of digits patterned by the ISOM. Courtesy of K.C. Oberg and Loma Linda University
Small random molecular variations of activator and inhibitor eventually lead to a stable pattern, typically spots or stripes. The pattern is dependent upon the level of activator and inhibitor as well as their diffusion rates. The evidence suggests that an intrinsic self-organizing or Turing mechanism establishes the initial alternating digit/non-digit pattern in the handplate. Although the identity of the activator and inhibitor are not yet known, the compound Hox/Gli3 experiments indicate that the terminal Hoxa/d transcription factors involved in the P–D and A–P/R–U axes, in concert with Shh/Gli3 regulation, modulate the intrinsic self-organizing mechanism and are critical in establishing the common digit/interdigit pattern of pentadactyly.
Defining Digit-Specific Morphology
Once the number of digits has been established, each digit must then acquire its specific morphology, i.e., thumb and index finger. At the distal end of each digit is a cluster of cells called the phalanx forming region (PFR) or digital crescent that, with progressive digital outgrowth, regulates Sox9 expression and chondrogenesis, thereby shaping phalangeal morphology (Fig. 1.8) [87–89]. The PFR also maintains digit-associated Fgf expression in the overlying AER during digit outgrowth [87].
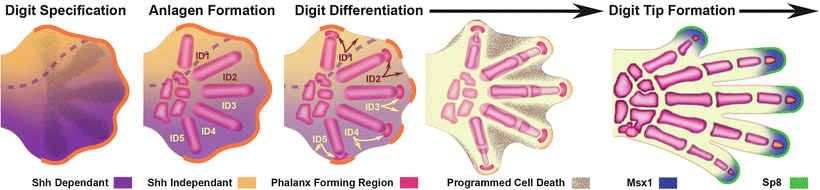

Fig. 1.8
Molecular pathways regulating digit development. After establishing digit number and the Shh dependant/independent domains (boundaries indicated by dashed line), digit morphologies are specified. Interdigital mesoderm as illustrated (ID1–ID5) regulates regression of the overlying AER (orange) and digit morphologies of the adjacent anterior condensing digit via the phalanx forming region (PFR—magenta) capping the distal tip of each anlagen. The PFR, in concert with the AER, determines phalanx size, length and joint position. The interdigital tissue subsequently undergoes Bmp mediated programmed cell death (speckled regions). As the AER overlying the digit regresses the distal or ungual phalanx begins to form and is demarcated by expression of mesodermal Msx1 (blue) and ectodermal Sp8 (green) (Image adapted from Oberg et al., 2010) [209]. Courtesy of K.C. Oberg and Loma Linda University
Although the mechanisms are not fully characterized, evidence suggests that Shh plays a pivotal role in defining digit-specific morphology for digits 2–5 (the Shh-dependent digits). Three Shh-regulated gradients converge to define the appropriate size and number of phalanges. The Shh-dependent Hoxd10-13 transcription factors within the developing digits interact directly with Gli3 [90]. However, the form of Gli3 present at each digit varies based on the Shh-regulated Gli3R/Gli3 activator counter gradients [91]. In addition, Shh induces a Bmp gradient that is also known to regulate digit morphology [89, 92]. The signals that determine digit morphology are conveyed to the PFR through the adjacent posterior interdigital tissue [87, 89] and through Fgf and Wnt proteins secreted from the overlying AER [88, 93–96]. Changes in the interdigital BMP levels or swapping interdigital mesenchyme can transform digit morphology [87, 89].
The thumb is a distinctly different digit in its shape, position, and structure [97]. It is Shh-independent and has a compilation of genes expressed within its domain that is dissimilar from other digits (Fig. 1.9). Hoxa13 is expressed within the entire handplate [75, 76] and overlaps the expression of Tbx5, which extends into the carpal and thumb domains but not into the domains of the ulnar digits (digits 2–5) [98]. Moreover, the thumb domain is accentuated by the lack of Shh-regulated Hoxd10-12 expression [99]. The absence of distal Hoxd gene expression has been used as a marker of “thumbness” across species [100, 101]. Interestingly, the wrist is also a zone with limited Hox protein expression (see the illustration in Fig. 1.9 associated with Hoxd12 expression). Recent experiments with mouse mutants that express low levels of Hox proteins showed transformations of metacarpal bones to carpal-like elements [102]. Thus, Tbx5 and low levels of Hox transcription factors may limit the size of the thumb and carpal bones, while the distal Hoxd transcription factors are thought to elongate digits [75, 98, 103].
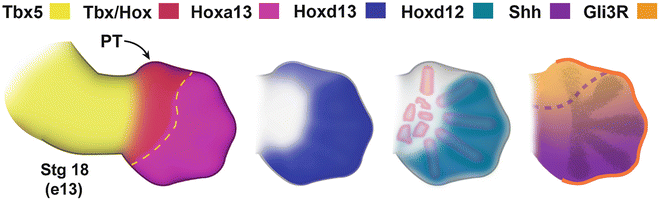

Fig. 1.9
Molecular regulation of thumb patterning. The presumptive thumb domain (PT) is defined by the overlapping expression of Tbx5, Gli3R, Hoxa13, and Hoxd13. The other Hoxd transcription factors (10-12) have overlapping expression domains in presumptive digits 2-5, but are restricted from the thumb domain. Note that the Hoxd genes are also restricted from the developing carpal region (Image modified from Oberg, 2014) [97]. Courtesy of K.C. Oberg and Loma Linda University
The terminal phalanges differ structurally from other phalanges: they are cone shaped and associated with a surface modification at the dorsal tip called the unguis or nail, which is dense keratinized epithelium that protects the tip of the digit. Terminal phalanges also differ in their development, with ossification beginning at the distal tapered tip of the cartilage model rather than forming a collar around the mid-shaft [104]. As the AER regresses, the terminal phalanges begin to form [94, 105]. Sp8, a specificity protein transcription factor that mediates Wnt signaling, is expressed in the distal tip ectoderm [104, 106] and appears to direct dorsal signals to form the nail. In mice with a reduction in Sp8 levels, dorsal dimelia forms (Haro et al., [107]). The distal tip mesoderm also expresses Bambi, a Bmp inhibitor, and Msx1, a transcription factor that is thought to provide regenerative competency to fingertips [104, 108, 109].
Interdigital Cell Death
In the interdigit mesenchyme, BMP signaling also induces cell apoptosis, in part, by repressing Fgf expression in the overlying ectoderm [110]. RA also appears to play a principal role in regulating interdigital cell death. Rdh10 knock-out mice, which fail to convert precursors to RA, show interdigital webbing and a reduction in the expression of Bmp7 [111]. RA beads are capable of inducing Bmp expression and cell death when implanted in the interdigit regions [112]. Weatherbee and coworkers have also suggested that levels of Gremlin, an Shh-regulated factor that inhibits Bmps, correlates with the degree of webbing across species [113]. Thus, Shh and RA signals may work in concert in the interdigital regions to signal digit morphology and interdigital cell death.
Limb Differentiation
While the limb is growing and acquiring its overall shape, cells from both ectoderm and mesoderm begin to differentiate into the various tissues required for limb function. The differentiation process is tightly regulated by signaling molecules of the three axes. Although we will discuss the different tissues separately (vessels, muscle, bone, cartilage, and nervous tissue), these processes are occurring concurrently, with several signaling molecules shared across tissues.
Limb Vasculogenesis
Vascularization begins with the transformation of mesenchymal cells into hemangioblasts [114]. Bmp4 signaling induces the expression of Flk1 (also known as Vegf-receptor 2) [115], the functional marker of hemangioblasts that confers the capacity to respond to vascular endothelial growth factor (Vegf) [116]. Embryos that lack Flk1 die around day 9 without any vascular development [117, 118]. Hypoxia-inducible factor 1 (HIF1alpha), sensing the local demands for oxygen in the growing tissue, induces Vegf [119]. Bmp4 conjointly with Vegf differentiates hemangioblasts into angioblasts (CD31, CD34, Flk1-positive cells), the precursors of vascular tissue [120, 121].
Angioblasts within the developing limb bud are derived from limb mesenchyme and cells that migrate from adjacent somites [122]. In the emerging limb bud, angioblasts aggregate and differentiate into vascular channels to form the primitive capillary plexuses [121, 123, 124] This process, known as vasculogenesis, is under the control of Vegf [125]. New vessels will sprout from these rudimentary vessels in response to local environmental and chemotactic factors, in a process termed angiogenesis. During angiogenesis, Notch-Delta signaling limits the number of sprouting “tip” cells to support directional outgrowth and remodeling [126, 127]. Interestingly, many of the molecules directing angiogenesis are also involved in axonal guidance (Ephrins/Eph receptors, Slit/Robo signaling, Netrins, Semiphorins, etc.) [128]. This may, in part, explain the parallel pathways taken by these tissues to form neurovascular bundles.
Angiogenesis progressively remodels limb vessels from proximal to distal. In addition to Vegf and Notch signaling, Angiopoietin/Tie signaling is involved in this second stage of vessel formation/remodeling [129, 130]. Around Carnegie stage 13 (day 28 post fertilization), remodeling forms a central limb artery (the primitive subclavian artery) that connects with the dorsal aorta (Fig. 1.10a); concurrently, two peripheral veins form to drain into the posterior cardinal system [123, 131]. The endothelial cells from these remodeled vessels secrete platelet-derived growth factor (Pdgf), which recruits smooth muscle cells and pericytes to surround the growing vessels [132]. Arteries and veins differ in the thickness of surrounding smooth muscle and pericytes. In addition, arteries express Ephrin B2, while veins express Eph-B4 receptors [119].
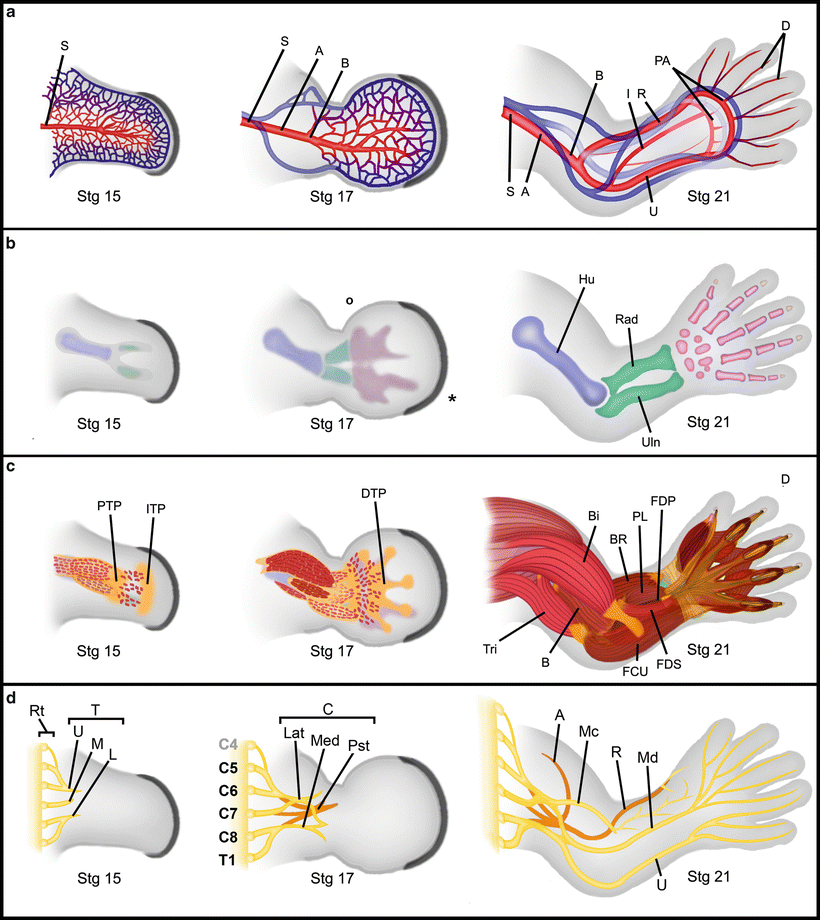

Fig. 1.10
Differentiation of limb tissues. Progressive differentiation of limb tissues from Stage 15 (post fertilization day 35) to stage 21 (day 52 near the end of the embryonic period). (a) Vascular differentiation showing the formation and remodeling of the subclavian (S), the axillary (A), the brachial (B), interosseous (I), radial (R), ulnar (U), palmar arch (PA) and digital (D) arteries. (b) Progressive skeletal differentiation showing anlagen condensation and definition for the humerus (Hu), radius (Rad), ulna (Uln), carpi and digits. (c) Progressive muscle differentiation. Myocyte migration is guided by tendon primoria: First the proximal tendon primordium (PTP) then the intermediate tendon primordium (IPT) and finally the distal tendon primordium (DTP). Secondary myocyte migration and subsequent proliferation within the fascicles defines muscle groups. Triceps (Tri), biceps (Bi), brachialis (B), brachioradialis (BR), flexor carpi ulnaris (FCU), palmaris longus (PL), flexor digitorum superficialis (FDS), and flexor digitorum profundus (FDP). d) Progressive differentiation of limb nerves. The nerve roots (Rt) from cranial 4 through thoracic nerve 1 coalesce to form the upper (U), the middle (M) and lower (L) nerve trunks (T) as they enter the limb bud. Further rearrangements define the lateral (Lat), median (Med), and posterior (Pst) cords (C). As the muscles differentiate and require innervation, major nerves are formed – axillary (A), musclulocutaneous (Mc), radial (R), median (Md) and ulnar (U). (Modified from Tonkin and Oberg, 2012) [198]. Courtesy of K.C. Oberg and Loma Linda University
RA plays an inhibitor role in the angiogenesis process [133, 134]. Experiments with mice lacking Cyp26, an RA degrading enzyme, showed an underdeveloped vasculature that did not progress beyond primitive plexuses [133, 134]. The data suggests that RA can have an inhibitory function on the expression of Flk1 thus halting the development of vessels [133, 134]. Endothelium expresses Cyp26 and may function to limit the presence of RA [Unpublished data, 132] thereby promoting angiogenesis. Alternatively, these early vessels may limit the level of RA accessible to the developing tissues they supply.
Progressive proximal-to-distal remodeling of limb vessels continues as the limb develops with primitive capillary plexuses persisting in the distal limb until about Carnegie stage 19 (post fertilization day 48). By Carnegie stage 21 (post fertilization day 52), the major vessels and general architecture is completed [135, 136]. The vascular network develops arteries, capillaries, and veins. The low pressure venous system is not able to collect all of the fluid distributed to the tissues by the higher pressured arterial system; therefore, a second low pressure vascular system, the lymphatics, forms. The lymphatic vessels also arise from angioblasts that are derived from LPM and somites [122]. Although a unique homeodomain transcription factor, Prox1, distinguishes lymphatics from arterial or venous vessels, the same signaling molecules that direct artery and vein formation likewise appear to control lymphatic vascular development [119].
Limb Skeletogenesis
The limb skeleton is derived from LPM and its development can be described in two steps: (1) chondrogenesis, the process of mesenchymal condensation and chondrocyte differentiation to form endochondral anlagen; and (2) endochondral ossification, the progressive transformation of the cartilage anlagen into the bones of the growing limb. The formation of joints is a related but separate process.
The first indication of chondrogenesis is the up-regulation of Sox9, a high-mobility group transcription factor, in chondrogenic precursors [137]. Sox9 is necessary for skeletogenesis; the lack of Sox9 in animal models results in the complete absence of cartilage and bone, culminating in limb regression [138]. However, Sox9 alone is not sufficient for chondrocyte differentiation. Additional Sox transcription factors (Sox5 and Sox6) are also needed for chondrocyte maturation, i.e., type II collagen production and chondrocyte hypertrophy [139, 140].
Bmp signaling also plays a role in the condensation of cartilaginous anlagen. Studies using constitutively activated and dominant-negative constructs in chicks show that signaling through Bmp receptor 1B (BMPR-1B) is necessary and sufficient to induce cartilage condensation [141]. The induction of Noggin, a potent inhibitor of BMPs, in the limb bud results in the complete absence of mesenchymal condensation [142]. Similarly in mice, knock-out of Bmp receptors 1a and 1b (BmpR1a, BmpR1b) impairs chondrocyte differentiation and Sox5/6/9 expression [143].
The ablation of individual Bmp proteins instead of their receptors does not prevent chondrogenesis in mice but rather delays the process [70]. This finding suggests that Bmps have a redundant function in chondrogenesis and that a threshold level of Bmp is needed to trigger the induction of Sox 5/6/9 and promote anlagen condensation. Despite the delay in cartilage condensation, individual Bmp knock-out mice exhibit normal endochondral ossification [70] (for a full review of the role of Bmp in skeletogenesis and embryonic development see [144]).
In contrast to Bmp, RA limits the expression and activity of Sox9 [145, 146]. Experiments with Cyp26b1 knock-out mice demonstrate impaired RA clearance. The elevated level of RA in the limb arrests or restricts cells to a pre-chondrocytic state and aborts cartilage formation and skeletal progression [147]. Interestingly, Bmp signaling counters this activity by inhibiting Raldh2, a gene that encodes for an RA synthesizing enzyme [148]. Thus, Bmps utilize direct and indirect pathways to promote chondrogenesis.
As with other aspects of limb development, chondrogenesis also progresses in a proximal-to-distal fashion. By Carnegie stage 15 (35 days post fertilization), the humerus, radius, and ulna anlagen are evident as a “Y”-shaped condensation (see Fig. 1.10b) [149]. During the next week of gestation (post fertilization days 36–42), condensations form within the handplate. A consistent order of digital condensations in vertebrates has been demonstrated with digit 4 forming first [150, 151] followed by digit 2, digit 5, digit 3, and finally the thumb or digit 1 [82]. Forming last appears to have put the thumb at increased risk, being the most common digit disrupted in malformation syndromes [97]. By Carnegie stage 21, the cartilaginous pattern is established.
Endochondral ossification is mediated in large part by the Runx2 transcription factor that differentiates precursors into osteoblasts and promotes chondrocyte hypertrophy [152]. In addition, Sp7 (also called Osterix), another specificity protein transcription factor, mediates osteocyte maturation, collagen I production and bone matrix deposition [153]. Sp7 works in concert with another transcription factor, ATF4, to maintain osteocyte function [154]. The ossification of long bones is also characterized by an epiphyseal plate that forms between the diaphysis (shaft) and epiphysis (ends). The epiphyseal plate is a growth center responsible for longitudinal growth. At the epiphyseal plate, cartilage proliferation forms regular columns of chondrocytes. These chondrocytes undergo hypertrophy, maturation, and apoptosis with subsequent ossification. These steps are tightly regulated by Runx2, Twist1, Ihh (and Gli3), Vegf, BMP, and FGF signaling [155].
Endochondral ossification transforms the cartilage models into bone. Primary ossification begins as a collar around the diaphyses of all limb long bones except the distal phalanges. Ossification in distal phalanges starts at the distal tip then progresses proximally over the cartilaginous model [104]. There is a consistent sequence to the formation of primary ossification centers in the upper limb. The first anlagen to begin ossification is the humerus (Carnegie stage 23, post fertilization day 56 or 8 weeks gestation), followed by the radius, ulna, distal phalanges, metacarpals, proximal phalanges, and finally middle phalanges by 10 weeks post fertilization [156]. Notably, George L. Steeter, the embryologist entrusted with characterizing the Carnegie collection of human gestations in the 1940s, regarded humeral ossification as the sine qua non of the beginning of the fetal period. Thus, the initiation of limb long bone ossification with the formation of primary ossification centers is a fetal endeavor.
Ossification of carpal bones does not start until around the time of birth [157]. The initiation of carpal ossification also follows a typical sequence beginning with the capitate and hamate (the ulnar aspect of the distal row) and ending with the trapezium, trapezoid (the radial aspect of the distal row), and the scaphoid (the radial aspect of the proximal row) [157]. Formation of secondary ossification centers within the epiphyses of the long bones also occurs postnatally. The characteristic pattern of hand and wrist ossification is a useful tool in assessing skeletal maturity in children. Prior to puberty, a sex-related difference is evident in hand and wrist ossification; in girls, formation of primary ossification centers is completed at around 6 years of age, whereas in boys, it is completed around 8 years of age [157].
The expression of the distal Hoxa transcription factors, Hoxa10, Hoxa11, and Hoxa13, correlates with the stylopod (arm), the zeugopod (forearm), and autopod (hand), respectively [76]. Synovial joints form within the developing skeletal anlagen at the boundaries between these three skeletal segments. Morphologically, a joint passes through three stages (Fig. 1.11): (1) interzone formation, with condensation of a cell dense region of flattened cells called the interzone; (2) cavitation, the formation of a gap separating the two skeletal elements; and (3) morphogenesis, the process of forming complementary articular cartilage-lined surfaces to facilitate movement. Wnt14 is expressed in the presumptive joint and up-regulates Gdf5 prior to interzone formation [158]. Gdf5 becomes tightly restricted to the interzone as it forms and promotes subsequent joint formation [159]. Centrally the interzone begins to cavitate, becomes hypocellular, and accumulates hyaluronan [160, 161]. Although joint-related muscular contractions are not needed for interzone formation, they are essential for proper cavitation to occur [162]. Integrated axis-related patterning pathways and cell movement work together to form complementary cartilage-lined surfaces on the opposing skeletal ends for appropriate articulation [163, 164]. Concurrently, mesoderm surrounding the developing joint condenses, forming fortifying ligaments and the joint capsule [165, 166].
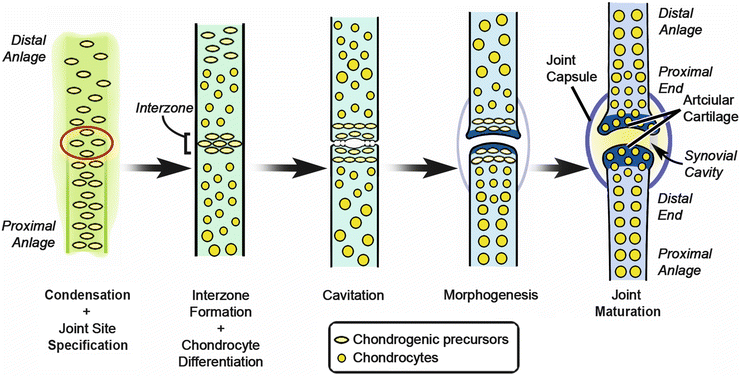

Fig. 1.11
Joint formation. Transformation of a presumptive joint in cartilage anlagen to a joint with synovial cavity and capsule (Image adapted from Pacifici et al., 2005) [164]. Courtesy of K.C. Oberg and Loma Linda University
Limb Myogenesis
Formation of the upper limb musculature is an integrated process involving tendons, myocytes, and nerves. Disruption of any one of these structures results in muscle abnormalities [167]. The arrangement of tendons and their sites of bony attachment establish the framework within which muscles will develop (see Fig. 1.10c). The tendon primordia develop from limb mesenchyme. The first indication of tendon formation is the expression of Scleraxis (Scx), a tendon-specific transcription factor in precursor tenocytes [168]. Subsequently, the cells express the extracellular matrix protein tenascin [169].
Three dorsal–ventral pairs of tendon primordia progressively develop within each limb segment [170]. The tendon primordia, which includes connective tissue cells that will encase and direct the developing myocytes, forms under the influence of axis-related signals and initially is independent from the influence of migrating myocytes [171]. For example, Wnt7a from the dorsal ectoderm regulates dorsal tendon formation and mice that lack En-1, the transcription factor that limits Wnt7a expression to the dorsal ectoderm, develop a symmetrical bi-dorsal phenotype, i.e., dorsal or extensor tendons for both the dorsal and “ventral” aspects of the limb [62, 65]. However, muscle interaction is an absolute requirement for maintenance of the tendon primordia and the final muscle arrangement; in muscle deficient limbs, the tendons form but then degenerate [170].
Muscle undergoes progressive and somewhat overlapping phases of development [172]: (1) an embryonic phase with development of primary mononuclear fibers from migrating myoblasts; (2) a fetal/neonatal phase generating secondary multinucleate fibers from migrating myoblasts; and (3) an adult phase that contributes multinucleated fibers derived from satellite cells.
The first marker of limb-related myocyte differentiation during the embryonic phase of myogenesis is the expression of Pax3, a pair-ruled homeodomain transcription factor, in the dorsolateral cells of the dermomyotome in limb-associated somites [173–175]. Subsequently, the Pax3-positive cells will delaminate and migrate into the developing limb bud. Pax3 knock-out mice show a loss of limb musculature and a loss of cell movements away from the somite [173, 176].
Delamination and migration are also dependent upon scatter factor/hepatocyte growth factor (Sf/Hgf) secreted from the developing limb bud mesenchyme and the corresponding expression of the Sf/Hgf receptor (c-met) in the myocyte precursors [177–180]. Pax3 regulates the expression of c-met in myocytes [181], while AER-associated Fgfs via Fgfr4 signaling control Sf/Hgf expression and thus the migratory routes of myocytes [180]. Mice deficient in c-met or Hgf expression lack migration and show a complete absence of limb musculature [178, 182].
As the myocyte precursors migrate into the limb bud, they split into dorsal and ventral precursors. Lbx1, a homeodomain transcription factor expressed in dorsal myocyte precursors, mediates this segregation. Disruption of Lbx1 disrupts dorsal muscle migration without significantly affecting the migration of ventral myocytes [183].
AER-related Fgfs regulate the expression of SF/Hgf within the limb mesoderm thereby controlling the migration of myocytes as they infiltrate tendon primordia to arrive at their final destination [180]. Within the limb bud, myocyte precursors begin to express MyoD and Myf5, committing them to a myocyte fate [184]. Activation of these myocyte-specific genes is also thought to depend on axis-related signal molecules, such as Wnt7a and Shh [174, 180]. The myocytes elongate and form primary mononuclear muscle fibers.
Progressive proximal-to-distal differentiation also occurs during myogenesis (see Fig. 1.10c). As myocyte precursors extend into the distal primordial tendons, a second wave of myocyte precursors migrates into the proximal limb. These myocyte precursors express Pax7 in addition to Pax3. Some of these precursors will coalesce around primary myofibers and fuse to form secondary multinucleated myofibers [185]. In addition, a population will remain in a precursor state at the periphery as a satellite cell [186]. Adult multinucleated muscle fibers are derived from satellite cells. It is during secondary or fetal myogenesis that motor endplates form and neuromuscular communication begins.
Limb Innervation
Innervation of the limb follows myocyte migration (see Fig. 1.10d). The axons of both motor and sensory neurons from the limb-associated spinal cord aggregate at the proximal limb boundary, forming several thick fascicles. These fascicles differentiate into the upper, middle, and lower trunks of the brachial plexus [187]. The nerve fascicles enter the limb then subdivide into dorsal and ventral branches. The dorsal branches coalesce to form the posterior cord. The upper and middle regions of the ventral branches join to form the lateral cord and the lower branch continues as medial cord. The cords then divide into the terminal branches of mixed motor and sensory axons. These branches follow a predictable pattern within the limb bud that appear to be controlled by variations in the extracellular matrix [188–190]. The initial entrance and distribution of the terminal branches within the limb do not appear to require signals from the final target tissue. However, for terminal sensory branching, the presence of skin is required [191]. Similarly, for fine targeted branching of motor nerves, differentiating muscle bundles are required [192].
The molecular control of axonal guidance and tissue targeting begins prior to axonal outgrowth during motor neuron differentiation. Shh secreted from the notochord and the floor plate of the spinal cord induces motor neuron and pancreas homeobox1 (Mnx1, previously called Hb9), which encodes a transcription factor that transforms the neuroepithelium into motor neurons [193]. Hox transcription factors expressed within the spinal cord organize motor neurons destined for the upper limb into the lateral motor column (LMC), which is also demarcated by the expression of Islet1 and Islet2 (Isl1/Isl2) lim homeodomain transcription factors. The expression of Raldh2, and thus the production of RA, within the lateral LMC induces the expression of lim homeodomain 1 (Lhx1) transcription factor and inhibits the expression of Isl1, further subdividing the LMC into medial Isl1/Isl2-positive neurons that will project into the ventral limb and lateral Lhx1/Isl2-positive neurons that extend into the dorsal limb [194].
A second phase of complex Hox transcription factor expression coupled with the expression of forkhead box P1 (FoxP1) transcription factor is thought to convey axon targeting information to specific partner muscles within the limb defined by axis-related cues [195]. A complex interplay of Ephrins and Eph receptors is involved in the regulation of branching and axonal guidance (see Kao et al. [195] for a comprehensive review). Finally, at the target site, Etv4 transcription factors are required to promote the axonal arborization needed for terminal neuromuscular innervation [196].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree