div class=”ChapterContextInformation”>
32. Urinary Diversion: The Last Resort of Male Urethral Reconstruction
Keywords
Continent urinary diversionIncontinent urinary diversionMonti and Casale techniqueIleocecocystoplastyBladder neck closureIleal conduit32.1 Introduction
Despite its technical difficulties, results from large series and systematic reviews have demonstrated that urethroplasty is the best option for the management of urethral strictures, with around 90% long term success rate in experienced hands [1–3]. For the small number of patients that fail revision urethroplasty, decline repeated urethral surgery, or are unfit for further reconstruction, urinary diversion remains a viable solution for appropriately selected patients.
For this subgroup of patients, urinary diversion options include perineal urethrostomy (Chap. 30) or continent or incontinent urinary diversion, which may require concomitant bladder neck closure and/or bladder augmentation, which will be the focus of this chapter. Urinary diversion surgery has significant early and late morbidity and mortality risks. When performed for quality of life indications such as refractory urethral stricture, it should be considered a last resort and patients should be appropriately counselled.
32.2 Etiology of Failure
Urethroplasty failures have been attributed to inferior tissue health. This may be secondary to extensive fibrosis in the setting of multiple previous endoscopic procedures, radiation therapy, poor vascular supply, prosthesis/ foreign body erosion, infection, and/ or long strictures or multiple previous attempts at urethroplasty. Often the etiology is multi-factorial with patients presenting with more than one risk factor increasing the risk of failure.
For patients that have failed repeated urethroplasties or endoscopic interventions with multiple risk factors for further failure; such as radiation, hypospadias, lichen sclerosis, strictures >4 cm; the most suitable way to manage their urethral stricture and expectations may be to abandon further reconstruction and proceed with definitive urinary diversion.
32.2.1 Pelvic Fracture Urethral Injuries
Failure of an end to end anastomosis after pelvic fracture urethral injuries (PFUI) is related to poor vascular supply and inadequate scar excision. This is made worse with repeated attempted re-anastomosis as multiple surgeries can result in further ischemic injury, due to the loss of vascularity to the bulbar urethra. This lack of vascularity causes ischemia and necrosis of the bulbar urethra. Bulbar urethral necrosis is a devasting outcome, and a major reconstructive conundrum requiring multiple variable techniques to reconstruct as it represents the loss of the entire length of the bulbar urethra and its vascular supply. Outside of major reconstructive urology centers, these patients may be considered for urinary diversion.
32.2.2 Prostate Cancer Treatment
Prostate cancer is the second most common cancer in men worldwide, resulting in 1.3 million new cases in 2018 [4]. Curative treatment options include radiation (external beam radiation (EBRT), brachytherapy (BT), or a combination), radical prostatectomy, or combined therapy. All therapeutic options have potential short term and long-term complications including the development of urethral stricture disease.
The reported incidence of early urethral stenosis or stricture following radical prostatectomy may be higher (8.4%) compared to EBRT or BT (1.7–5.2%) [5], with population-based analysis demonstrating a high 10-year cumulative incidence of 19.3% for prostatectomy, 9.6% for EBRT, 11.94% for BT, and 19.4% for EBRT + BT [6]. While stricture and stenosis rates may be higher following surgery, radiation-induced strictures have high rates of urethroplasty failure (30.3%) [7].
Reconstruction of radiation strictures is associated with higher failure rates even in experienced hands due to the inherently poor tissue effected by the radiation treatment. Given considerations such as age, life expectancy with their prostate cancer, and other medical comorbidities, a urinary diversion may offer a more predictable outcome for these patients, with a better quality of life, and avoid multiple attempts at high risk urethroplasties.
Management of post-radiation strictures by repetitive endoscopic procedures, such as direct vision internal urethrotomy (DVIU), TURP, and laser enucleation, can lead to the creation of difficult and devastating prostatic obliterative strictures, urethra-cutaneous fistulas, recto-urethral fistulas, osteitis pubis, and osteomyelitis of the pubic symphysis. The urethral tissue in these cases frequently is obliterative in nature associated with infection and inflammation. Attempted repairs of these devasting complications of radiation have success rates of 0–86%, dependent on the extent of the radiation damage and surgeon’s experience [8]. Given the high rate of failure and increased complications from attempted repairs, such as fistula, devastation of the bladder neck, erectile dysfunction, and urinary incontinence; urinary diversion is often safer and offers a more predictable outcome.
Patients with radiation-related strictures may also develop a fixed prostatic or membranous urethra such that despite what appears to be an adequate lumen, they suffer ongoing obstructive symptoms similar to that of a tight urethral stricture. These patients may be offered repetitive dilations, DVIU, and TURP with no improvement in outcome due to the fibrotic nature of the urethra. For these patients, urinary diversion is a superior choice of treatment as repeated endoscopic interventions do not result in sustained improvement in symptoms but increases their risk of significant complications. The rigidity of the urethra and often the urinary sphincter can also lead to stress incontinence due to the inability of the sphincter to co-apt. This may be further exacerbated following multiple endoscopic interventions where the sphincter is used as a fulcrum point due to the rigidity of the urethra. These patients may then develop the devastating outcome of incontinence and the inability to completely empty. Urinary diversion with a bladder neck closure could be considered in these instances to optimize their quality of life.
32.2.3 Urinary Prosthesis
Prostheses have been available since the early 1980s for both erectile dysfunction and urinary incontinence. Devices such as synthetic slings, artificial sphincters, or urethral compression devices have been utilized, all of which have been associated with urethral strictures.
The Artificial Urinary Sphincter (AUS) has established itself as the gold standard for post-prostatectomy urinary incontinence. The most recent models have an associated infection and erosion risk of 8–9%. Erosion of the AUS can result in significant urethral tissue loss and subsequent stricture development.
For patients considering reimplantation of a urinary sphincter device following a previous complication, those with a history of radiation therapy, repeated treatment for bladder neck contractures, urethral stent placement, or previous device infection or erosion are at higher risk of repeat erosion and urethral loss. The outcomes of AUS or any newer compressive prosthesis following stricture treatment have not been extensively studied, however, what data there is demonstrates that re-implantation of the AUS does result in a higher rate of erosion than in a virginal implant [9, 10]. To avoid the cycle of urethroplasty, reimplantation, further erosion, and explantation, serious consideration for urinary diversion can be considered in these situations.
Urethral stents for stricture disease , such as the Urolume, have become unpopular due to concerns related to the extension of urethral stricture disease beyond the initial stricture site, fibrosis extension into the stent itself causing stent incorporation into the urethral wall, worsening of the inflammatory process with associated obstruction of the stent, haematuria and poor patient satisfaction. Potential urethral incorporation often requires more complicated procedures for Urethral stent removal, resulting in urethral tissue loss, and increased rates of urethroplasty failure, sometimes requiring urinary diversion for management [11].
32.2.4 Benign Prostatic Hyperplasia Treatment
Urethral stricture and bladder neck contractures are uncommon complications of BPH surgical management. Reported urethral stricture rates vary between energy sources from 1.4% up to 19% [12], while bladder neck contracture rates can be as high at 6.5%.
The traditional open simple prostatectomy has low rates of bladder neck contracture at 3.8% and urethral strictures up to 4.8%. The urethral stricture tends to occur in the urethral meatus which can be easier in the short term to manage.
Both monopolar and bipolar transurethral prostate resection is associated with bulbar and meatal stricture formation, with rates as high as 19% for bipolar TURP [12]. Risk factors that increase the rate of the stricture formation include increased operative time, larger prostates (>70 cc), surgeons experience, mismatch in size between urethral lumen and instruments, and insufficient insulation by the lubricant causing urethral thermal injury.
Bladder neck contractures are also an infrequent (5–9.7% [13, 14]) but bothersome complication of TURP. Factors that increase the risk of bladder neck contracture formation include circumferential resection, mechanical failure with stray current, urinary extravasation, ischemia from large urethral catheters, and infection.
Photovaporisation and Holmium laser enucleation have comparable rates of urethral stricture and bladder neck contracture to TURP, at 0.02% to 3.6% and 0% to 0.8% respectively [15–17].
Management of these complications is fraught with difficulties. One or two attempts at urethral dilation or urethrotomy can be considered in favorable stricture disease. However, patients with long strictures >2 cm and strictures located in the penile or membranous urethra have poor outcomes with repetitive dilation or urethrotomy, so should be considered for urethroplasty or urinary diversion. Many of these patients have had significant lower urinary tract dysfunction prior to their BPH surgical management and would prefer a definitive resolution to their difficulties, and a urinary diversion would offer this immediately for them.
32.2.4.1 Neurogenic Bladder and the Devastated Outlet
Refractory stenosis, urinary incontinence, or both can result from trauma, complications from surgeries or cancer therapies, or sequelae of neurogenic bladder dysfunction, and can be simplistically referred to as the devastated bladder outlet (DBO). Neurogenic bladder (NGB) patients, including those with spinal cord injury or myelomenigocele, are at risk for urethral disease secondary to frequent instrumentation and clean intermittent catheterization. DBO development in the NGB patient is a risk as the disease bladder may exhibit low capacity, high pressure storage, or an incompetent outlet, increasing risk for reconstruction failure. When reconstruction options are exhausted or ill-advised, urinary diversion with or without outlet closure may be indicated to provide continence and urinary quality of life.
32.3 Pre-Operative Evaluation
32.3.1 History and Examination
Male patients with an unreconstructable urethra are a heterogeneous group, with various etiologies represented.
A thorough history should be obtained, including characterization of both previous and current patient symptoms. A complete record of all previous surgical interventions may not be possible but will help with delineation of possible remaining salvage surgical techniques and consideration of risk factors for any diversion.
Patients with a history of urogenital malignancy should be evaluated for possible disease recurrence and life expectancy prior to undertaking any diversion. It is important to establish whether there has been previous radiation for any possible pelvic malignancy including rectal, cervical and urogenital as this will influence the choice of bowel segments for diversion, the possible need for cystectomy and the rate of complications following diversion. Reviewing comorbidities including simultaneous bladder or urethral fistula, ureteral stricture, hydronephrosis, chronic pelvic pain, or renal dysfunction preoperatively will influence whether a patient is a candidate for a bladder conserving approach to diversion.
Careful attention to rectal health, including the presence of simultaneous rectal pain, incontinence, or fistula, is an important consideration as this may affect the patient’s management goals and options.
An evaluation of a patient’s performance status, including ability (dexterity and cognition) and willingness to perform intermittent catheterization or irrigation, is essential when deciding between a continent or incontinent diversion. A discussion of the potential effects of diversion on sexual health and fertility should be completed preoperative so patient expectations are clearly understood.
Abdominal examination should assess for previous abdominal scars and to determine appropriate stoma placement based on the patient’s body habitus.
For patients with recent urethral reconstruction, dilation, or clean intermittent catheterization, a period of urethral rest with a suprapubic catheter to determine if urethral stenosis or stricture will redevelop should be considered. If complete urethral obliteration develops, the patient may not require bladder neck or urethral closure at the time of diversion.
32.3.2 Laboratory Evaluation and Imaging
Routine blood work including renal and liver function should be obtained as the use of bowel in bladder augmentation or urinary diversion may be contraindicated in patients with renal or liver dysfunction.
For patients with a history of or at risk for prostate cancer, PSA testing should be considered in order to ensure no latent or recurrent cancer is present.
Upper tract imaging should be obtained to determine the presence or degree of hydronephrosis or unilateral renal deterioration.
Cystourethroscopy can be utilized to determine if bladder tumors are present, especially in patients with a history of bladder cancer or previous pelvic radiation. Additionally, patency of the urethra and quality of the bladder can be reviewed by cystoscopy, as the presence of radiation necrosis or dystrophic calcifications may necessitate more extensive debridement or cystectomy at the time of diversion.
Urodynamic testing is essential in order to determine the patient’s bladder capacity, continence status and the presence of detrusor overactivity. This essential information may indicate the need for simultaneous bladder augmentation, incontinent diversion or a bladder neck closure in the same surgical setting.
32.4 Continent Catheterizable Urinary Diversion
In motivated patients with good performance status who are free of contraindications such as, pelvic pain, fistula, renal dysfunction, hepatic dysfunction, or concurrent bowel disease (such as inflammatory bowel disease), continent urinary diversion can provide improved quality of life by allowing continence without the need for indwelling catheters or external drainage bags.
- 1.
a reliable continence mechanism
- 2.
an adequate volume
- 3.
low pressure reservoir that avoids the use of synthetic materials
- 4.
technically reproducible in construction.
If required, augmentation cystoplasty should be completed in the same operative setting.
Continent vesicostomy with an ileal channel is typically preferred in the adult patient for whom bladder preservation and continent catheterizable channel (CCC) is indicated, as other options may be limited by anatomic considerations.
While generally the preferred technique in children, Mitrofanoff appendicovesicostomy is usually impossible as the adult appendix is often surgically absent or inadequate secondary to limitations in length, lumen, or mesentery, especially in the obese patient.
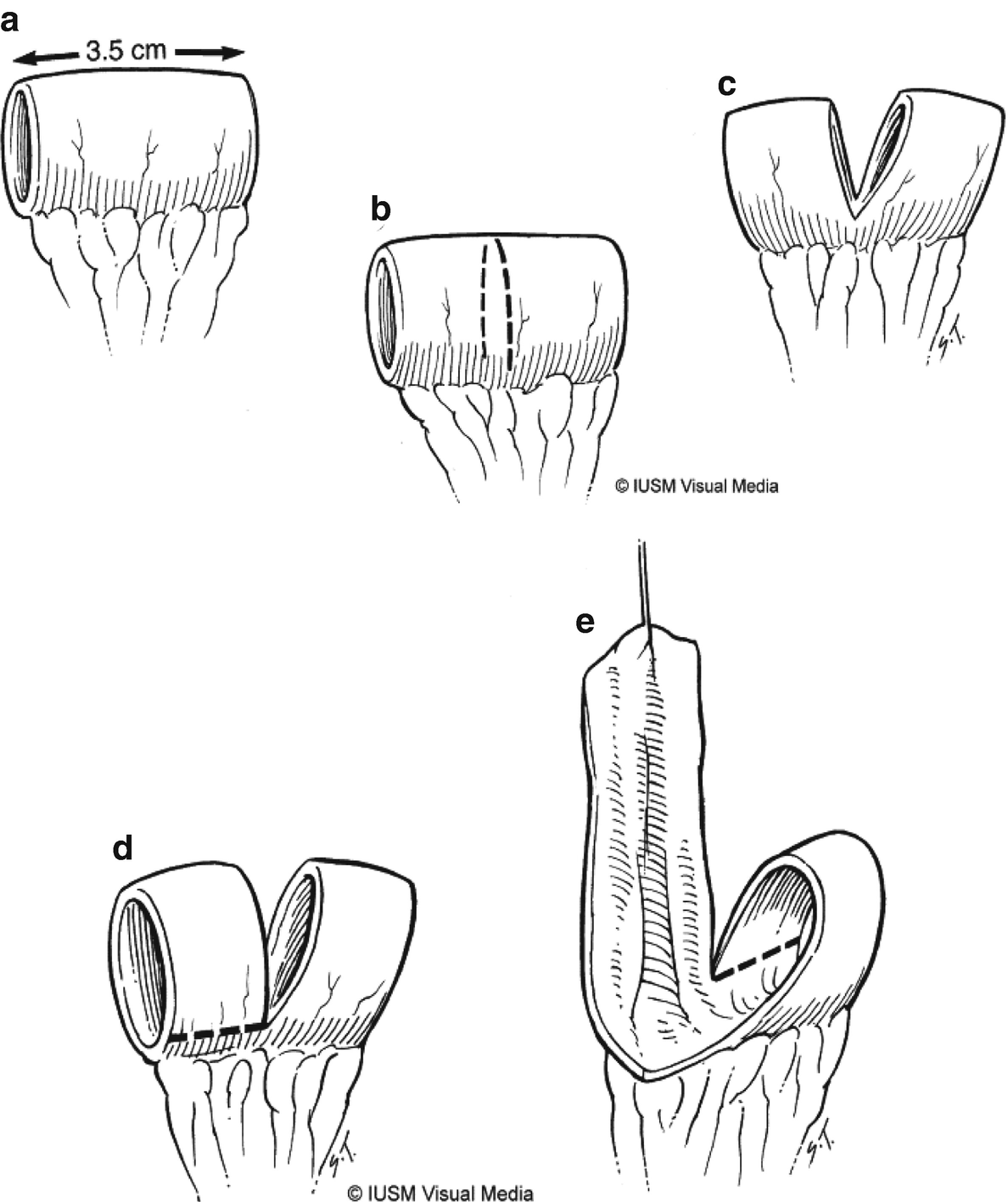
Casale or Spiral Monti catheterizable channel
Tubularization of spiral loop to construct the catheterizable channel
32.4.1 Monti and Casale Techniques
A 4 cm segment of ileum is isolated 10–15 cm proximal to the ileocecal junction. A slightly longer segment can be harvested if additional reach is required. If the patient’s body habitus is such that length will not be an issue, a smaller segment can be harvested, detubularized along the midline length, and tubularized as a standard Monti channel. In the more typical Casale technique, the segment is marked in the midline and divided for approximately 80% of its circumference. Each of the two bowel rings are then incised just at the mesentery on opposite sides, leaving two strips of ileum. This Z-shaped strip is then tubularized with absorbable suture over a 14 French catheter with the resulting channel typically measuring 10–14 cm in length. The tube is tunneled in the bladder distally and a stoma is matured in the umbilicus or, if the distance is too far, the right lower quadrant.
Advantages of this technique include less concern about post-operative issues with gastrointestinal transit or malabsorption as the terminal ileum and ileocecal valve are left in continuity when compared with an ileal cecocystoplasty. Tunneling within the bladder is critical to facilitate continence of the tube, and the ends of the Casale or Monti tube are free to mesentery, facilitating easy tunneling and stoma maturation. However, care needs to be taken with thick hypertrophic bladders, as they tend to cause obstruction or kinking at the tunnel , making it difficult for the easy passage of the catheter.
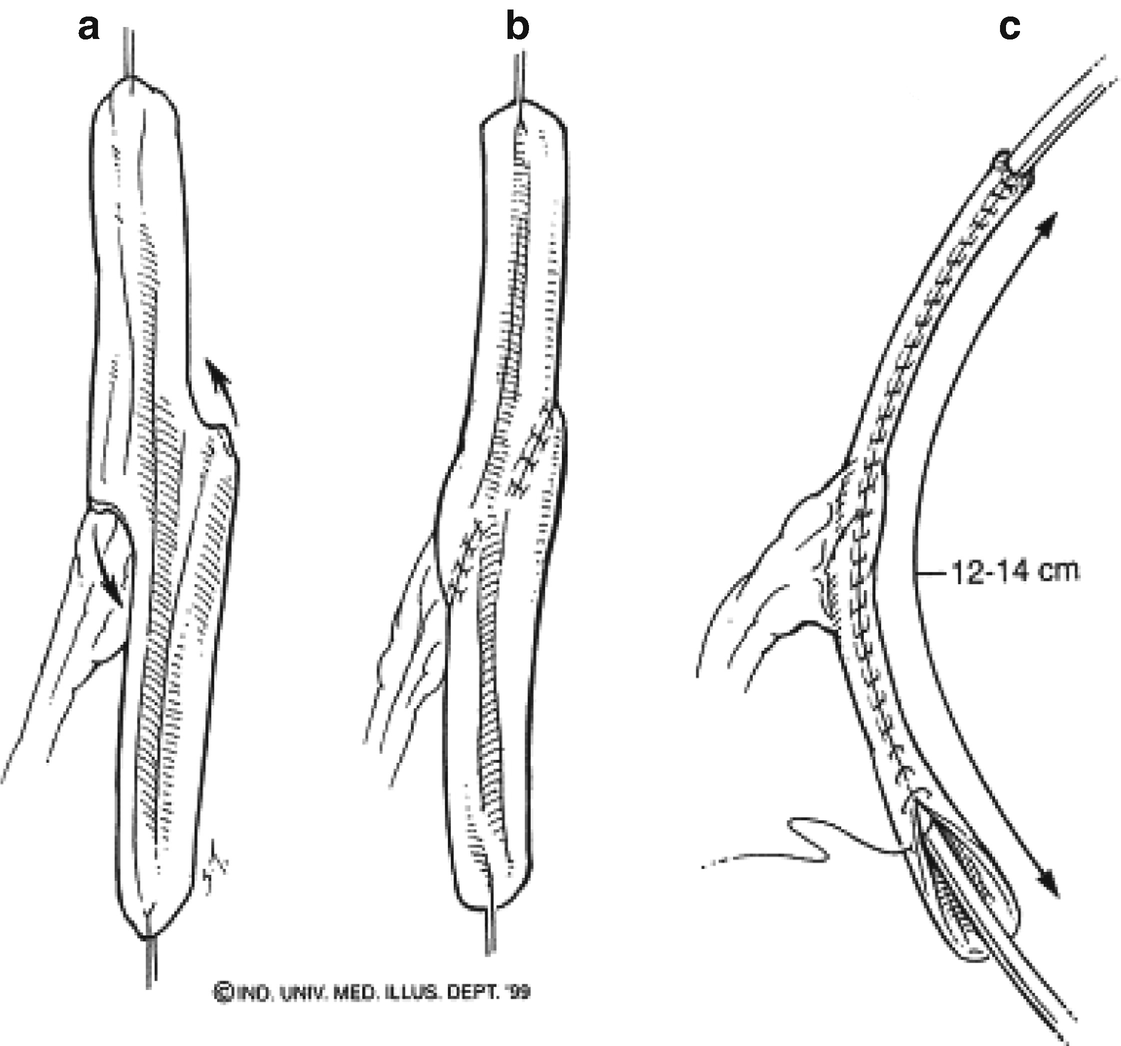
32.4.2 Catheterizable Ileal Cecocystoplasty Technique
When a continent urinary diversion is indicated for patients with unreconstructable urethral disease, the “hemi-Indiana” catheterizable ileal cecocystoplasty (CICC) first described by Sarosdy in 1992 is an ideal option [20, 21]. This operation consists of simultaneous bladder augmentation with detubularized right colon and catheterizable channel creation using tapered terminal ileum. Thus, the continence mechanism is provided by the intact ileocecal valve.
An extended midline abdominal incision is made allowing the cecum and ascending colon to be mobilized. The bladder is divided in a clamshell fashion and 10–15 cm of ascending colon and 8–10 cm of terminal ileum are marked and staple divided. A side-to-side staple anastomosis of the ileum and ascending colon is performed using the GIA-100 stapler.
The colon is next detubularised 1 cm lateral to the taenia. The ileum is tapered over a 14 French catheter using linear staples. The valve may be reinforced with imbricating permanent sutures similar to an Indiana pouch procedure, but we now avoid placing these sutures as it is unclear if they improve continence but may result in difficulty with catheterization or kinking. The appendix is removed. The colon augment is anastomosed to the open bladder with absorbable suture , and the stoma is matured and brought to the skin at the umbilicus.
A minimally invasive approach to this operation has been described which includes ascending colon mobilization first using a hand-assist laparoscopic technique [22, 23]. The hand-assist port is then extended to a Pfannensteil incision for the anastomosis to the bladder and creation of the tapered ileal limb, allowing avoidance of a large midline incision.
A pouchogram can be considered at 10–14 days post-operatively, but we do not find this to be necessary. The indwelling catheter is removed, and patients may begin self-catheterization at 3 weeks. A suprapubic catheter is removed once efficient catheterization is ensured. A negative culture at this stage is mandatory since catheter removal may produce transient vesicoureteral reflux and febrile urinary tract infection can occur.
32.4.3 Continent Catheterizable Channel Outcomes
For functional and motivated patients with an unreconstructable urethra, a CCC may provide a higher quality of life compared to permanent suprapubic catheterization or incontinent diversion requiring an external drainage bag [24]. Few series have addressed the outcomes of these operations in male patients diverted specifically for urethral pathology.
Reoperation rates in modern series of adult patients requiring CCC ranged from 12.9–50%, with most reoperations performed secondary to stomal complications [25–27]. Hadley et al. reported outcomes for 26 patients (6 male, 4 with a devastated bladder outlet) requiring Monti, double Monti, or Casale diversion for NGB [26]. Three of seven (43%) patients who underwent a double Monti were unable to catheterize secondary to stenosis of the channel, and this operation was abandoned in favor of the Casale technique later in the series. At 64 months follow-up, 19 patients (73%) were catheterizing through their stoma. Stomal stenosis, dehiscence, or necrosis was experienced by 1 patient each (12%).
Redshaw et al. compared 31 patients who underwent CICC to 30 patients with tunneled CCC using Monti, Casale, or double Monti techniques. The indication for surgery was NGB in 51 patients (84%) with the remaining patients requiring diversion for unsalvageable urethral problems or bladder neck stenosis. They found more tunneled CCC patients (50%) required a secondary operation for channel problems compared to the CICC group (13%, OR 6.4, 95% CI 1.8–28) at an equivalent 16 months of follow-up [27]. A difference in stomal revision remained significant when CICC was compared to the subgroup of tunneled CCC patients who underwent simultaneous bladder augmentation (46.7%, OR 4.2, 95% CI 1.1–19.6). An initial stomal leakage rate of 29% was observed in the CICC group compared to 43% of the tunneled CCC group (p = 0.12), but the authors report leakage was resolved in most patients (93.5% with CICC and 77.7% for other channels) with catheterization regimen adjustment and anticholinergic therapy.
The authors recognize that low rates of stomal stenosis and revision following CICC may be secondary to short follow-up in this group [27]. Khavari et al. reported stomal stenosis in 3 of 34 CICC patients (9%) compared to 1 of 31 patients (3%) in the Redshaw et al. series [25]. A stomal continence rate of 91% was reported in this series which is similar to the final 93.5% reported by Redshaw et al. These rates compare favorably to a 65% continence rate reported by Van der Aa et al. in their series of 34 adult NGB patients who underwent tunneled CCC [28].
Utilization of tapered ileum during CICC allows for a longer channel compared to detubularized ileum, which may be limited to a 10–14 cm length, possibly resulting in tension on the stoma in obese patients. Hadley et al. recognized in their study that stomal complications were more common in patients with higher body mass index. Additionally, there may be risk of devascularization of the ends of the detubularized ileal channels as the mesentery to the segment is small, limited to the original length of bowel that is harvested.
These reviewed series are primarily composed of NGB patients with urethral indications for CCC observed in only 0–16% of included patients, with concomitant outlet procedure with bladder neck closure (BNC) or AUS performed in 24% of the patients in Khavari et al. and 58% in Hadley et al. [25–27] Therefore, the results may be difficult to extrapolate to the male patient with unreconstructable urethral disease typically related to radiation therapy or multiple previous lower urinary tract operations.
Complication rates in series of CCC patients with simultaneous BNC may better represent expected outcomes. Stomal stenosis was reported in 3 of the 12 patients (25%) with a devastated bladder outlet (DBO) requiring BNC and a tunneled CCC in the series by O’Conner et al. [29] Shpall et al. and Spahn et al. similarly reported stomal revision after BNC and CCC in 5 of 19 patients (26%) and 4 of 15 patients (24%), respectively [30, 31]. These results are also consistent with a stomal stenosis rate of 27% reported in the series of 11 patients who underwent BNC and CCC following salvage prostatectomy [32]. Of note, CICC was not performed in any of these patients while appendicovesicostomy was commonly utilized, potentially accounting for a higher stomal complication rate.
Overall, post-operative complication rates were high (31–54%) in these series which included an abdominal operation for a continent catheterizable channel [25–27, 30], with higher complications being reported in patients requiring bladder augmentation (47–54%) compared to CCC alone (33%) [25, 27].
32.4.4 Simultaneous Augmentation Cystoplasty with CCC
Spahn et al. reported stomal incontinence in 3 of 17 patients (18%) who underwent BNC and CCC with Mitrofanoff or ileal intussusception valve [31]. Their report of DBO patients included 13 males, including ten with a history of prior radical prostatectomy. In their series, only patients with a pre-operative bladder capacity less than 300 ml underwent simultaneous ileal bladder augmentation. They noted that following BNC, six of the eight patients (75%) who did not undergo augmentation required anticholinergic therapy, of whom three developed stomal incontinence. They theorized that BNC is associated with a post-operative loss of bladder capacity and reported two of the three patients were noted to have resolution of their stomal incontinence following delayed bladder augmentation. The authors ultimately concluded that augmentation should be considered in all patients requiring BNC for a DBO.
These results are corroborated by De et al. who remarked that previous patients who underwent salvage prostatectomy with BNC and CCC experienced persistent urgency, low capacity, anticholinergic requirement, and a frequent catheterization interval, and as such shifted their practice to performing routine bladder augmentation in all future patients with BNC in this setting [32, 33]. Pre- and post-operative urodynamic studies of the 34 patients who underwent CICC augmentation in the Khavari et al. series revealed a median increase in bladder capacity of 205 ml [25]. This increased capacity may allow for a longer necessary interval between catheterizations and therefore an improved quality of life in these patients, which must be weighed against the increased morbidity and complication potential following augmentation. Preoperative evaluation of the patient’s bladder capacity is essential, with consideration that capacity may be further diminished following BNC.
It is our bias to perform augmentation cystoplasty in all patients requiring BNC and continent diversion for unreconstructable urethral disease, with rare exceptions such as in patients with large capacity bladders noted on urodynamics studies. Failure of the bladder neck closure or diversion can be secondary to imperfect surgical technique, unfavorable local tissues, or high pressure, small capacity bladders, and therefore augmentation in addition to aggressive anticholinergic therapy is the standard approach to these patients.
32.5 Bladder Neck Closure
For patients with the combination of an unreconstructable urethra and confirmed stress urinary incontinence, simultaneous bladder neck closure (BNC) could be considered as a concurrent procedure with bladder-preserving urinary diversion.
BNC permanently removes the ability to obtain bladder drainage via urethral catheterization in the setting of acute urinary retention , a significant risk in patients with a continent diversion who may develop stomal stenosis or occlusion. As such, BNC should be considered a procedure of last resort to improve urinary incontinence in patients who are otherwise without another reconstructive option.
32.5.1 Transvesical Bladder Neck Closure Technique
In patients, without a history of radiation therapy, we prefer the transvesical bladder neck closure (TBNC) technique as described by Reid et al. [34] An infraumbilical midline or Pfannenstiel incision is made. A transverse incision is made in the bladder dome allowing adequate exposure of the trigone.
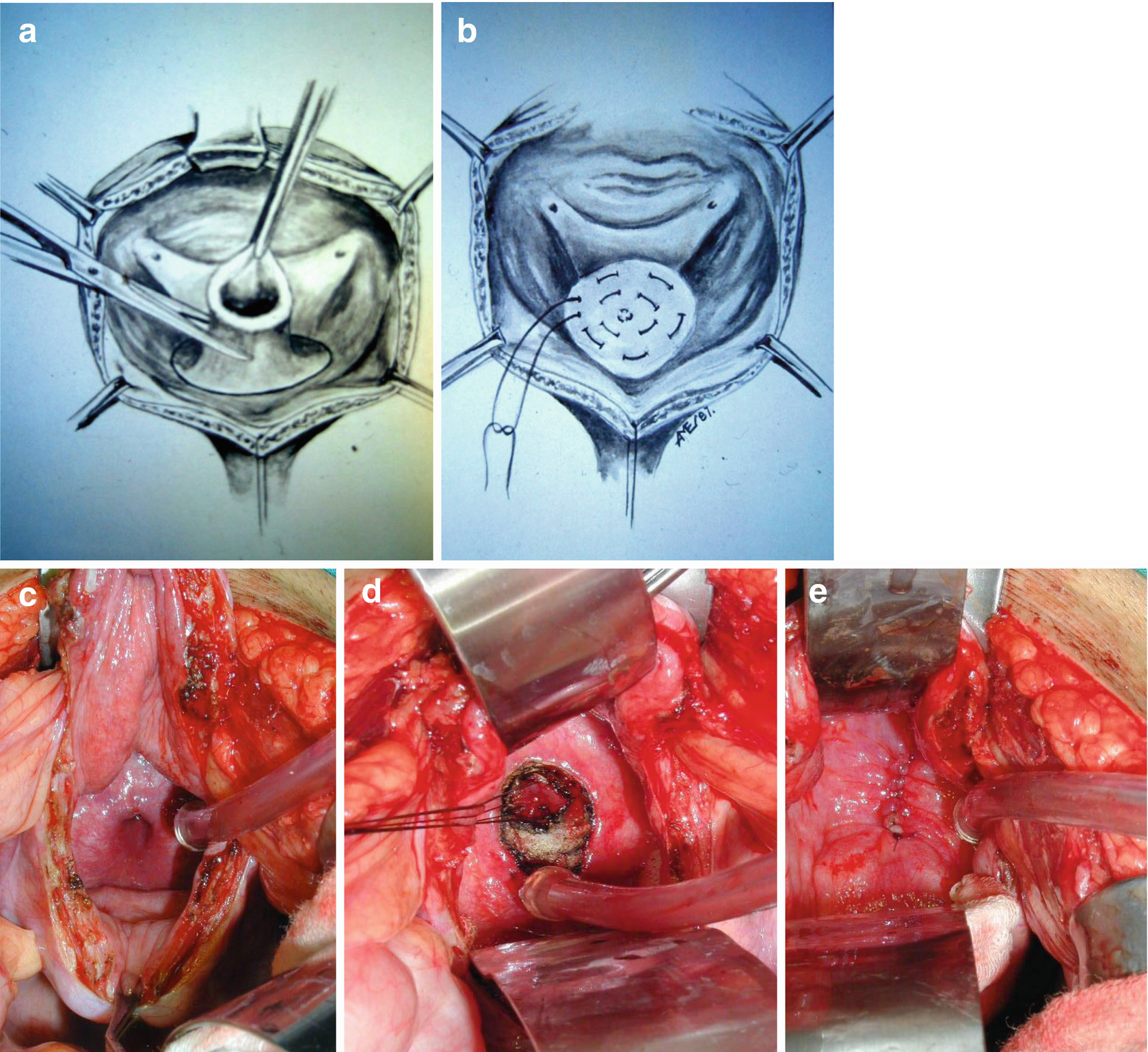
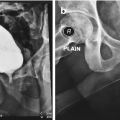
Transvesical bladder neck closure. The bladder is bivalved and the bladder neck excised (a) and closed in 3 layers (b–e). Reid et al. [43]
The benefits of the TBNC compared to a retropubic technique includes significantly less pelvic dissection decreasing operative time, blood loss, and the risk of adjacent organ injury.
32.5.2 Retropubic Bladder Neck Closure Technique
While increasing operative time and potential for blood loss and morbidity, the retropubic (RBNC) technique allows for cephalad mobilization of the bladder from the urethral stump and for tissue interposition and is therefore preferred in patients with previous radiation therapy, prior failed BNC, or other risk factors for failure.
The patient is positioned in a slightly hyperextended Trendelenburg position. The retropubic space is entered via a midline abdominal incision that can be extended cephalad if a diversion or augmentation is also planned. The bladder is mobilized by dividing the urachus and lateral pedicles in an approach similar to cystectomy. The dorsal venous complex, if intact, is a potential source of bleeding and is ligated. A vertical cystotomy is then made 1–2 cm cephalad to the bladder neck to allow visualization of the trigone.
The ureteral orifices are then stented with 5 French feeding tubes to facilitate visualization throughout the closure. The bladder neck is divided using cautery. Grasping the posterior bladder neck edge with Allis clamps allows cephalad mobilization of the bladder. A plane between the bladder and rectum is developed along Denonvilliers’ fascia. Development of this space allows cephalad mobilization of the bladder neck away from the urethra.
The bladder neck is closed with interrupted 2-0 polyglycolic acid sutures with a second layer of closure utilized to imbricate the first closure layer. If feasible, the urethra is closed in an interrupted fashion. In patients with a history of radiation, excellent hemostasis and care not to enter the rectum are essential.
In patients without a history of radiation therapy or previous BNC failure, we do not feel tissue interposition is essential but may be performed to reduce the risk of vesicourethral fistula. If entry into the abdomen is planned for urinary diversion creation, an omental flap is a reasonable source for interposition. A flap of omentum is mobilized along the left or right gastroepiploic artery and draped over the pelvic floor with interrupted suture, separating the urethral stump from the closed bladder neck. If urinary diversion will be achieved with a suprapubic catheter (SPC) alone and entry into the abdomen is not planned, a pedicled rectus abdominis muscle flap can provide a well vascularized interposition [35].
In patients whose prostate remains in situ, dissecting the bladder neck proximal to the prostatic urethra can allow for continued fertility and sexual function in a young male who desires this. If the prostate is planned to be left in situ the patency of the urethra must be investigated. If complete urethral obstruction distal to the prostate is observed then there will be no outlet for prostatic secretions, which can lead to pain or problematic fluid collection over time. Salvage prostatectomy should also be considered at the time of BNC in patients with previous radiation therapy for prostate cancer and may be performed to additionally provide distance between the closed bladder neck and urethra.
32.5.3 Bladder Neck Closure Outcomes
Success of the BNC specifically relies on the technique’s ability to prevent vesicourethral fistula formation. There are few reported series on the success of BNC in male patients, and fewer specifically reporting in those with refractory urethral disease.
Reid et al. first described TBNC reporting success in 4 of 6 male patients (67%) who underwent simultaneous continent vesicostomy for neurogenic bladder (NGB) [34].
Many of the modern RBNC series report male and female success rates of 83–100% [29–31, 36, 37]. Spahn et al. reported a 0% vesicourethral fistula rate following RBNC with omental interposition and continent vesicostomy at 68 months follow-up in 17 patients (13 males) [31]. They attribute their success to meticulous hemostasis, cephalad mobilization of the bladder, and tissue interposition. Kranz et al. similarly reported 100% success in 15 patients (5 with a DBO) following a 2-layer RBNC with omental interposition, augmentation , and continent ileovesicostomy at 24 months follow-up [37].
Vesicourethral fistula is a risk following BNC, and Shpall et al. reported a 15% failure rate in their series of 39 NGB patients at 37 months follow-up [30]. Patients in this series, including 29 male patients and 9 patients with previous urethral surgeries, underwent RBNC with omental interposition and simultaneous ileovesicostomy or continent diversion.
O’Conner et al. reported a 17% failure rate of BNC in 35 complex DBO patients (24 males, 3 with post-prostatectomy complications) at follow-up of 79 months, potentially indicating that the fistula rate may be higher in patients requiring RBNC for urethral indications [29]. All patients underwent RBNC with omental interposition and various forms of urinary diversion, including SPC alone in 15 patients. Of the 6 failures , 4 underwent a successful second transabdominal procedure.
Colli and Lloyd reported a 9% vesicourethral fistula rate in their unique series of 11 male NGB patients, of whom 7 required previous urethral dilations [36]. All patients underwent a RBNC without tissue interposition and permanent SPC diversion. All patients had pre-operative UDS testing revealing a bladder capacity >125 ml and none had previous radiation therapy. The comparable outcomes in this series supports the practice of not routinely providing tissue interposition during BNC in this select patient population.
No injury or deterioration of the upper tracts was observed in follow-up in any series, although this was not consistently reported. Although use of anticholinergic therapy was not routinely discussed in these series, we consider this to be mandatory when BNC is performed for patients with an unreconstructable urethra. A history of radiation therapy or previous surgery to the lower urinary tract was also inconsistently reported, and the success observed in these series may not be representative in the complex male patient with unreconstructable urethral disease.
32.5.4 Simultaneous BNC Following Salvage Prostatectomy
BNC with augmentation cystoplasty and/or urinary diversion is a controversial approach to lower urinary tract reconstruction in the patient requiring salvage prostatectomy. As incontinence rates following salvage prostatectomy are very high, even when AUS is ultimately utilized, this approach may allow for simultaneous treatment for an almost inevitable complication. Reported series involving a complex patient population with a 100% rate of radiation therapy prior to BNC showed acceptably low rates of vesicourethral fistula formation, indicating BNC success can be achieved in even complex patients requiring BNC for unreconstructable urethral disease.
Pisters et al. first reported outcomes for patients who underwent BNC and continent urinary diversion at the time of salvage prostatectomy for local prostate cancer failure following radiation therapy [33]. The initial series of 12 patients underwent BNC following retropubic salvage prostatectomy, which included wide bladder neck and membranous urethral excision, including surrounding skeletal muscle, in order to achieve surgical margins. Omental interposition and continent diversion were performed without augmentation. Four patients (33%) required reoperation including 1 patient (8%) with a vesicourethral fistula who ultimately required cystectomy.
Ullrich and Wessels described a technique following salvage retropubic prostatectomy in which a segment of small or large bowel can be anastomosed to the widely opened bladder neck, allowing simultaneous bladder neck closure and augmentation [38]. This technique may be useful for BNC when extensive necrosis of the prostate is present and significant debridement of the bladder neck is required. The use of bowel interposition allows for tension-free closure of a large bladder neck defect in these cases, and the presence of the bowel mesentery creates its own tissue interposition between the bladder neck and urethral stump. All five men in this series were without vesicourethral fistula formation (100%) at a mean follow-up of 6 months, although 1 patient required exploratory laparotomy for bladder rupture at 4 months post-procedure and another experienced stomal stenosis.
32.5.5 Urethral Closure with Suture Ligation
As an alternative to BNC , permanent urethral closure is an emerging option to achieve continence in patients with an unreconstructable urethra and incontinence. With appropriate pre-operative counseling, this procedure can be considered as an alternative at the time of attempted reconstruction or salvage of a challenging urethral operation. The most likely scenario for urethral ligation to be considered is at the time of AUS explant in a patient with multiple previous urethral operations or cuff erosions who may be a poor candidate for urethroplasty and eventual device reimplantation, potentially avoiding months of urinary incontinence while a definitive operation is planned.
To perform a permanent suture urethral ligation , a perineal incision is made and the bulbar urethra is circumferentially dissected and divided at or near the bulbomembranous junction. The mucosa of the proximal and distal urethra is oversewn with 4-0 poliglecaprone suture, with multiple figure-of-eight sutures used to invert the proximal urethral stump. The stump is secured to deep perineal tissue and layers of tissue, including bulbospongiousus muscle, is interposed.
This procedure is advantageous in that it reduces operative time and morbidity from abdominal surgery in patients who will be diverted with an SPC. Quality tissue interposition is limited, but outcomes may be improved by coverage with spongiosum applied as a rotational flap if the local tissues are of good quality [39].
There exist only limited series reporting outcomes following urethral ligation, however, high success rates have been reported. Higutchi et al. reported on an adult series of 6 urethral ligation patients with 1 reported failure (17%) in a patient with a history of pelvic radiation who experienced post-operative urethral incontinence [39]. A high success rate (100%) was achieved in another series of four young men, all of whom had a history of ileocystoplasty and continent catheterizable channel (CCC) for myelomeningocele [40]. Three of the patients had a history of autologous fascial sling for persistent urethral incontinence following CCC diversion, and success was reported at a follow-up of 49 months.
VanDyke et al. reported outcomes of urethral ligation with concurrent SPC diversion in a series of 10 adult men with end-stage urethral disease [41]. Nine men (90%) had a prior AUS implantation, three (30%) had prior radiation therapy, and a median of 4 prior urethral surgeries was reported among all the men, including 5 patients (50%) with prior urethroplasty. Four patients (40%) developed refractory bladder spasms of whom 2 (20%) developed a urethrocutaneous fistula, and an additional 2 patients (20%) developed a post-operative abscess. Ultimately 70% of patients were dry at a median follow-up of 12 months – one patient required cystectomy, one became dry after a salvage urethral closure, and a third patient had a small persistent fistula despite a second operation.
Complications including persistent urinary incontinence and fistula formation are not uncommon following urethral closure with suture ligation. Therefore, it should be considered a last resort option when an abdominal operation for BNC is ill-advised due to the high risk of intra-operative and post-operative morbidity, in patients with persistent stress urinary incontinence and/or urethral leakage despite previous surgery for urinary diversion.
Aggressive medical management of bladder spasm with anticholinergic therapy is mandatory in all patients undergoing bladder neck or urethral closure. Simultaneous bladder OnabotulinumtoxinA injection may also be considered to help improve outcomes, especially in patients with a small capacity bladder.
32.6 Incontinent Urinary Diversion
For patients with an unreconstructable urethra who are unsuitable for diversion with continent catheterizable channels; incontinent urinary diversion remains an option with good quality of life outcomes. Patients that are not suitable for continent urinary diversion typically are those with pathology which requires concomitant cystectomy, failure of the catheterizable channel or catheterization difficulty, or persistent incontinence following bladder neck closure.
Incontinent urinary diversion options are diverse and include ileovesicostomy or ileal conduit diversion, or less commonly, cutaneous ureterostomy or ureterosigmoidostomy. The benefit of reservoir formation over cutaneous ureterostomy is the reduced risk of stomal stenosis and subsequent urosepsis or upper tract deterioration. This is especially the case for patients with a benign indication for diversion who often have a long life expectancy. Ureterosigmoidostomy was a common technique for urinary diversion in the early twentieth century but is now less commonly utilized due to concerns with recurrent infections, worsening renal function , metabolic abnormalities, or malignancy [42, 43].
32.6.1 Suprapubic Catheter Placement
For patients that would prefer a simple intervention or are not medically fit enough for a larger reconstructive procedure, a suprapubic catheter (SPC) is an acceptable option acknowledging the requirement for a catheter tube and bag.
In patients with no previous abdominal surgeries, a bladder capacity of greater than 300 mls, an SPC can often be placed with endoscopic guidance (cystoscopy and insertion), avoiding the need for an open cystotomy placement. An ultrasound may be utilized to determine if bowel loops are overlying the bladder prior to puncture with an SPC trocar, to decrease the risk of bowel injury. In patients with previous abdominal surgeries, increased abdominal girth/BMI, ultrasound is essential when relying on trocar SPC technique to avoid bowel injury. Open cystotomy is recommended for safe access if the bladder capacity is small or there is bowel overlying the bladder.
Long term suprapubic catheter usage is generally well tolerated. A review by Hunter et al. found that most patients become more optimistic regarding their need for catheterization over time [44]. “Satisfaction” rates of 52–72% are reported in quality of life studies despite the high complications rates associated with long term catheterization [45, 46]. Bacteriuria is inevitable and commonly leads to urinary tract infections in patients with long term catheterization. Katsumi et al. reported long term SPC outcomes for male spinal patients. In their cohort of 46 patients, 11% developed urosepsis and 30% of patients developed >1 UTI per year. 2 (4%) patient deaths resulted from urosepsis [47]. Bladder stone and neoplasia occurrence from long term catheterization increases with the duration of catheter use and study follow up duration. The incidence of bladder stone formation is estimated to be 25% at a mean of 50 months, and close to 50% by 20 years [48]. Rates of catheter-related squamous cell carcinoma are estimated to be 0–2.2% [49].
32.6.2 Ileovesicostomy
Ileovesicostomy provides a relatively simple approach to incontinent diversion that retains the native bladder function and innervation, preserves the native trigone, and maintains sexual function. It is associated with less operative morbidity than other supravesicle diversion approaches as cystectomy, ureter mobilization, and ureteroileal anastomosis is not required. Additionally, the anti-reflux mechanism of the native ureter is preserved. Ileovesicostomy is unique in that it preserves the native urinary tract and is therefore reversible were the patient to experience poor functional outcomes that may later be managed with another diversion technique [49, 50].
Ileovesicostomy was first described by Smith and Hinman in 1955 in canine models [51]. This was shortly followed by the first ileovesicostomies in humans in 1957 by Cordonnier [52]. The contemporary technique stems from the work of Schwartz et al. [50] with several authors (Rivas [53], Mutchnik [54], Gudziak [55], Gauthier [56]) making iterations to the technique since. Principally described for the management of patients with neurogenic bladders who cannot be managed with intermittent self-catheterization , ileovesicostomy is also a recognized option for patients with intractable lower urinary tract dysfunction or a DBO [57].
32.6.2.1 Ileovesicostomy Technique
- (i)
Wide-mouthed vesical anastomosis

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree