Disease of the Breast, Benign to Premalignant; Hyperplasia to DCIS
Michele L. B. Ley
Jeffrey A. Sugandi
Pathology of the breast encompasses a wide variety of disorders, including benign, premalignant, and malignant diseases, as well as disorders of uncertain malignant potential. A variety of infections and systemic diseases may affect the breast. This chapter presents a review of common breast diseases including benign and carcinoma in situ diseases.
The functional unit of the breast is the terminal ductal lobular unit. This is composed of small acini, which drain into a terminal duct. The terminal duct is the first tributary of the ductal system of the breast, which functions to extrude milk from the nipple. The entire lobular and ductal structure of the breast is lined by two layers of cells, the inner epithelial layer and the outer myoepithelial layer, which arises by preferential growth of the inner epithelial layer. A useful corollary of this paradigm is that the demonstration of the absence of myoepithelial cells by immunohistochemistry can be used by the pathologist to determine the malignant potential of a lesion.
Benign Disorders
Fibrocystic change is the pathologic condition that correlates with the term “lumpy, bumpy” breasts. This term is applied to a plethora of benign changes in the breast, which are best categorized based on subsequent risk of development of breast carcinoma. Dupont and Page (1,2) reviewed benign breast biopsies from 3,000 women and used well-defined criteria to categorize them into three groups: nonproliferative lesions, proliferative lesions without atypia, and atypical hyperplasia. This clinically relevant classification has been independently corroborated and sanctioned by a Consensus Conference of the College of American Pathologists (3,4).
Nonproliferative Lesions
This is the most common category of breast disorders and includes cysts, papillary apocrine change, mild hyperplasia of the usual type, and epithelial-related calcifications.
Women with these lesions do not incur a higher risk of development of breast carcinoma than women who had no breast biopsy (relative risk 0.89).
Cystic Breast Mass
In the early 20th century Joseph Bloodgood described blue cysts in the breast (blue-domed cysts of Bloodgood) (5). Haagensen noted that cysts may form a palpable mass, and thus he termed them gross cysts (6). However, most cysts are small and can be visualized only on microscopic examination (Fig. 21-1).
Breast cysts can be diagnosed with physical examination as palpable, ballotable mass. Breast ultrasound can classify whether or not the cyst is simple, complicated, or complex. Table 21-1 summarizes the characteristic findings of cystic breast masses.
Simple breast cysts are benign and no surgical excision is indicated. They are typically a well-circumscribed, anechoic mass with well-defined wall and posterior enhancement and are categorized as BIRADS-2 (benign) on imaging. Fine-needle aspiration (FNA) under ultrasound guidance can be performed if there is concern for inflammation or infection or for symptomatic relief. Turbid or purulent aspirate with concern for infection should be sent for culture. If the cyst does not collapse,
or there is any suspicion of abnormality, for example, frankly bloody aspirate with solid component, then core needle biopsy should be performed instead of cytology to obtain tissue diagnosis. Patients with a simple breast cyst that collapses after aspiration with concordant clinical findings do not need further workup and may resume routine screening (7).
or there is any suspicion of abnormality, for example, frankly bloody aspirate with solid component, then core needle biopsy should be performed instead of cytology to obtain tissue diagnosis. Patients with a simple breast cyst that collapses after aspiration with concordant clinical findings do not need further workup and may resume routine screening (7).
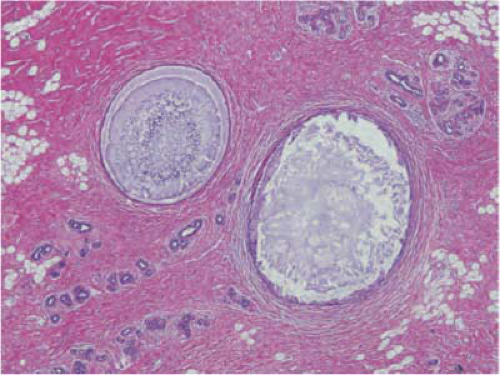 FIGURE 21-1 Fibrocystic change, nonproliferative type. Two cysts are filled with inspissated secretions. Dense fibrosis is seen in the background with normal and atrophic lobules. |
TABLE 21-1 Characteristics Summary of Breast Cysts | ||||||
|---|---|---|---|---|---|---|
|
Complicated cysts are considered a BIRADS-3 (probable benign) on imaging and associated with a ∼2% risk of malignancy (8). Therefore they require a short-term follow-up imaging and physical examination. Core needle biopsy should be performed for any suspicion of malignancy or increase in size.
Complex cysts have both cystic and solid components, thick walls, and/or septa. These cysts are associated with a higher risk of harboring malignancy (14% to 20%) and are categorized as BIRADS 4–5 (suspicious or highly suggestive of malignancy) on imaging, therefore core biopsy is required.
Proliferative Lesions Without Atypia
Women with these lesions have a slight risk of development of breast carcinoma, approximately 1.5 to 2 times that of the general population. This category includes moderate or florid hyperplasia of the usual type, sclerosing adenosis, small-duct papillomas, and fibroadenoma (1,9).
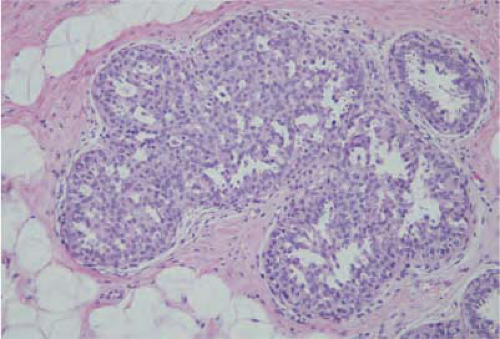
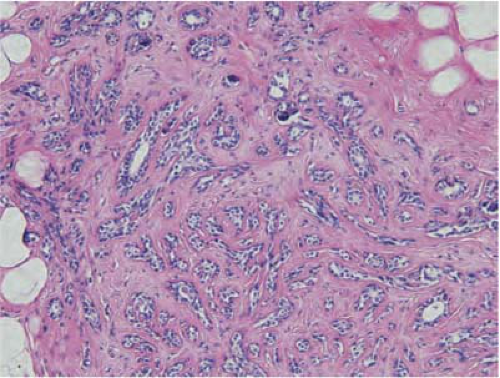 FIGURE 21-2 Sclerosing adenosis. An enlarged lobule has numerous acini in a sclerotic, dense stroma with focal microcalcifications. |
Sclerosing adenosis is the most common lesion and refers to expanded lobular units with a proliferation of both acini and intervening stroma. Microcalcifications are often seen in association with sclerosing adenosis and frequently correspond to “benign calcifications” seen on mammography (Fig. 21-2). These lesions are also often identified on breast MRI mimicking the nonmass enhancement of in situ lesions. MRI-guided biopsy can confirm the diagnosis. Rarely, a clinically palpable mass may be formed by a large number of lobules with cellular sclerosing adenosis and has been referred to as an adenosis tumor. It is considered benign and no treatment or surgical excision is needed for sclerosing adenosis.
Usual ductal hyperplasia, moderate or florid, is an epithelial proliferation characterized by ducts that are more than four cells’ thick and having a slit-like fenestrated pattern (Fig. 21-3). Solid and papillary patterns may also be seen. The cells may overlap, show slight variability in shape and size, and have a swirling or streaming pattern. No additional treatment is required for usual ductal hyperplasia.
Small-duct papillomas arise in the peripheral duct system and tend to be multiple. Microscopically, they are composed of fibrovascular papillae covered with the two
cell types of the ductal system, the inner epithelial and the outer myoepithelial cells.
cell types of the ductal system, the inner epithelial and the outer myoepithelial cells.
Atypical Hyperplasias
Atypical hyperplasias confer a risk of development of breast carcinoma that is 3.5 to 5 times that of the reference population. This category includes both atypical ductal hyperplasia (ADH) and atypical lobular hyperplasia (ALH) (1,2).
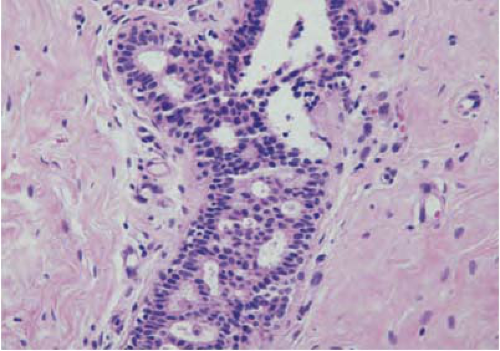
These lesions are difficult diagnostic problems, with variable interobserver agreement (10,11,12). Their morphologic features are midway between those of usual hyperplasia and carcinoma in situ. Page et al. defined criteria for ADH that are widely used and include nuclear monomorphism, regular cell placement, and round, not fenestrated, spaces in part of the duct (Fig. 21-4) (13). ALH refers to proliferation of lobular cells with distention of acini in no more than half of a lobule and may include pagetoid spread into adjacent ducts (14). Earlier studies showed that atypical hyperplasias do not have a linear progression to carcinoma but they increase the risk of development of breast carcinoma in both breasts (2,15). A recent retrospective analysis found that the breast diagnosed with ALH is three times more likely to develop invasive carcinoma than the contralateral breast (16). These data suggest that ALH may lie somewhere between linear progression and generalized risk.
Columnar cell hyperplasia (CCH) with or without atypia is a newly recognized morphologic entity. CCH without atypia is one of many hyperplastic lesions that slightly increase the relative risk of development of breast carcinoma and are grouped under fibrocystic change, proliferative type (17). CCH are frequently associated with microcalcifications and may have mild, moderate, or marked atypia. Severe atypia typically has micropapillary architecture and may be indistinguishable from ductal carcinoma in situ (DCIS). CCH has been noted to be associated with ALH, lobular carcinoma in situ (LCIS), and tubular carcinoma (18). As with other atypical proliferative lesions, there is a high level of interobserver variability among pathologists in diagnosing CCH with atypia (19). Flat epithelial atypia is also a newly recognized morphologic entity (18) where the columnar shaped epithelial cells that layer the terminal duct show low-grade cytologic atypia. Limited available data suggest that it has a very low risk of both local recurrence and progression to invasive cancer (20).
A diagnostic of an atypical lesion of any type as rendered on a core biopsy is an indication for a surgical biopsy. Atypical lesions are diagnosed in 4.3% to 6.7% patients who undergo a core biopsy for a mammographically detected lesion, and the subsequent surgical biopsy reveals intraductal carcinoma in 12.5% to 36% and invasive carcinoma in 0% to 14% of patients (21,22,23,24). Magnetic resonance imaging (MRI) screening has been recommended for women at high risk for development of breast carcinoma (25).
Atypical lesions of the breast are a diagnostic challenge for the surgical pathologist, and there is considerable interobserver variation among pathologists in interpreting these lesions. The Susan G. Komen for the Cure Foundation published a white paper on breast pathology (26) that recommends that the accuracy of diagnosis of breast lesions can be enhanced by specialty training, second opinions, and integration of pathologists into clinical care teams.
Radial Scars and Complex Sclerosing Lesions
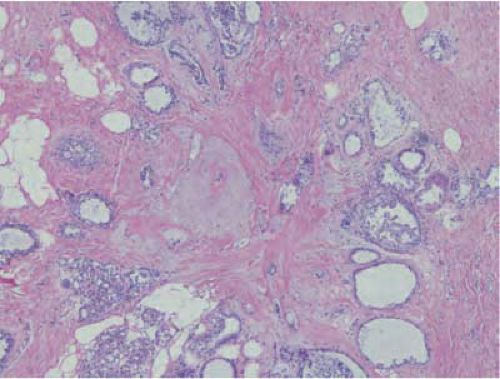
Radial scars are typically small areas of scarring (<1 cm) surrounded by glandular elements. Their stellate morphology mimics a classic scirrhous carcinoma for both the mammographer and the pathologist. Typically they are seen only on microscopy, which reveals an area of intense sclerosis in the center, surrounded by dilated ducts, sclerosing adenosis, and apocrine metaplasia (Fig. 21-5). Glands caught up in the central scirrhous zone may mimic invasive carcinoma on high magnification. The overall architecture of the lesion is critical in avoiding this pitfall, especially on frozen section. Numerous sclerotic lesions do not have the classic morphology of a radial scar but show varying degrees of sclerosis and ductal hyperplasia. These lesions are commonly referred to as complex sclerosing lesions.
In a carefully performed case control study, it was shown that women with biopsy-proven radial scars are at an increased risk for breast cancer. Women with proliferative lesions with atypia associated with radial scar
had a relative risk of breast cancer of 3.0 (27). Surgical excision is recommended when radial scar is diagnosed on core biopsy.
had a relative risk of breast cancer of 3.0 (27). Surgical excision is recommended when radial scar is diagnosed on core biopsy.
Benign Neoplasms
Fibroadenoma
Fibroadenomas (FAs) typically present as painless, mobile, rubbery masses. They are usually solitary but are occasionally multiple (28). They are most often present in the upper outer quadrant and are slightly more common in the left breast.
On gross examination, fibroadenomas are well-circumscribed smoothly lobulated masses. On microscopic examination, fibroadenomas are biphasic having epithelial and stromal components (Fig. 21-6). Occasionally, the epithelial component is insignificant and the stromal component is hypocellular and densely sclerotic. This variant, hyalinized fibroadenoma, is invariably associated with coarse microcalcifications, which may be worrisome to the mammographer.
Fibroadenomas usually present as palpable, well-defined, rubbery mobile mass on physical examination. Imaging findings on ultrasound reveal well-defined, lobulated solid mass. Diagnosis can be confirmed with core needle biopsy and then followed with short-term follow-up within 3 to 6 months. In asymptomatic patients, fibroadenomas can be left alone. If a fibroadenoma is symptomatic or increases in size on follow-up, then surgical excision is warranted. Cryoablation therapy under ultrasound guidance is an alternative approach to management of fibroadenomas, once the diagnosis is confirmed with core needle biopsy. It may take up to 12 months for the progressive resolution of fibroadenomas treated with cryoablation (29,30).
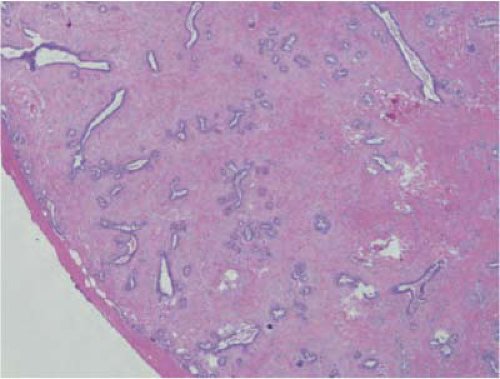 FIGURE 21-6 Fibroadenoma. The tumor is circumscribed and covered by a pseudocapsule. It is composed of numerous ducts in a background of cellular stroma. |
Juvenile fibroadenoma is a variant seen in young girls, and these tumors show both epithelial and stromal hypercellularity (31). They may be multiple, grow rapidly, and cause venous dilation in the overlying skin. Tubular adenoma is a subtype that has an abundance of small tubules on microscopic examination and is also common in young women (32). Complex fibroadenomas contain cysts larger than 3 mm, sclerosing adenosis, epithelial calcifications, or papillary apocrine change. One study showed a higher risk of breast cancer (33) in patients with such lesions.
Lactational adenomas are seen during pregnancy and in the postpartum period and may represent lactational changes superimposed on an underlying tubular adenoma or fibroadenoma. They present a well-circumscribed mass, which on microscopy shows glands and ducts with secretory changes. Infarction, pain, and tenderness may complicate fibroadenomas during pregnancy.
Phyllodes Tumor
Phyllodes tumor is a rare fibroepithelial breast tumor that accounts for less than 1% of all breast neoplasms (35). This entity encompasses tumors that run the gamut from benign to malignant but mostly fall into the category of tumors of uncertain malignant potential (borderline). Conventionally, phyllodes tumors have been referred to as cystosarcoma phyllodes.
On physical examination, phyllodes tumor can be indistinguishable from fibroadenoma. A large breast mass >3 cm with rapid growth should prompt suspicion
for phyllodes tumor and requires core needle biopsy or surgical excision. Microscopically, they have characteristic hypercellular stroma around the ductal elements, which form a leaf-like pattern giving the lesion its name (Fig. 21-7). A high mitotic count, nuclear pleomorphism, infiltrative tumor margins, and stromal overgrowth are the main criteria used to assess biologic behavior. Benign tumors are characterized by increased cellularity, lack of stromal overgrowth, mild to moderate cellular atypia, circumscribed margins, and low mitotic rate (<4 per 10 high power fields). Borderline tumors are characterized by higher degree of cellularity and atypia, mitotic rate of 4 to 9 mitoses per 10 high power fields, and microscopic infiltrative borders. Malignant tumors have marked stromal cellularity, atypia, infiltrative margins, stromal overgrowth, and more than 10 mitoses/10 high power fields (36,37).
for phyllodes tumor and requires core needle biopsy or surgical excision. Microscopically, they have characteristic hypercellular stroma around the ductal elements, which form a leaf-like pattern giving the lesion its name (Fig. 21-7). A high mitotic count, nuclear pleomorphism, infiltrative tumor margins, and stromal overgrowth are the main criteria used to assess biologic behavior. Benign tumors are characterized by increased cellularity, lack of stromal overgrowth, mild to moderate cellular atypia, circumscribed margins, and low mitotic rate (<4 per 10 high power fields). Borderline tumors are characterized by higher degree of cellularity and atypia, mitotic rate of 4 to 9 mitoses per 10 high power fields, and microscopic infiltrative borders. Malignant tumors have marked stromal cellularity, atypia, infiltrative margins, stromal overgrowth, and more than 10 mitoses/10 high power fields (36,37).
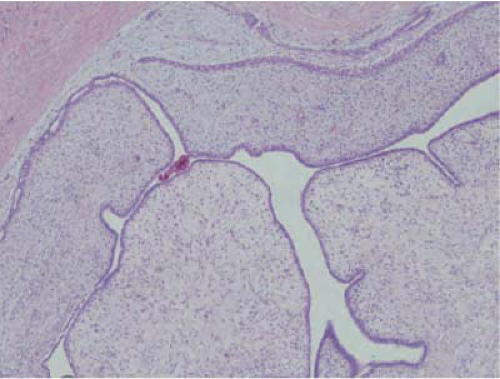 FIGURE 21-7 Phyllodes tumor, benign. The tumor has a frond-like architecture, with compressed ducts lined by ductal and myoepithelial cells. |
As with all stromal tumors, ample sampling is necessary because the sarcomatous component may be small and limited to a small portion of the tumor. Phyllodes tumors tend to have local recurrences and should be widely excised (38,39). Wide excision with at least 1-cm margins is recommended for management of borderline and malignant phyllodes tumor. Benign phyllodes can be excised without additional margins. This recommendation is based on data that showed that tumor size and surgical margins are the prognostic determinant of local recurrence and distant metastases (40). Axillary lymph node involvement is rare in phyllodes tumor, therefore axillary surgery is usually not indicated.
Intraductal Papilloma
These tumors typically arise in a large duct in the subareolar region and present with symptom of unilateral spontaneous bloody nipple discharge. One nested case control study showed that papillomas with areas of ADH within them have a risk of breast carcinoma at that site, which is similar to the risk associated with ADH in the adjacent breast (41). On careful dissection papillomas appear as pearly, small, white nodules. Microscopically, they distend a duct and are composed of a stalk with branching thick fibrovascular cores lined by myoepithelial and ductal cells (Fig. 21-8). The proliferation of both cell types is an indicator of benign nature. Complex growth patterns can make the distinction between a papilloma and papillary carcinoma challenging for the pathologist. Papillomas typically do not show significant pleomorphism, mitotic activity, and necrosis (42,43).
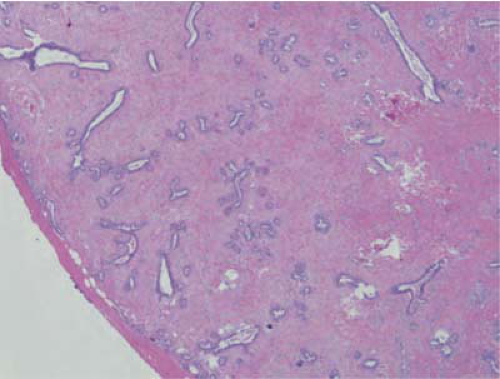 FIGURE 21-8 Intraductal papilloma. A large duct contains a papillary structure with thick fibrovascular cores lined by ductal and myoepithelial cells. |
Papillomas with atypia have been shown to have pathologic upgrade up to 67% at the time of surgical excision, therefore surgical excision is recommended. However, literature on pure papillomas without atypia is mixed with little consensus on management (44,45,46). The available data showed that the relative risk of breast cancer in papilloma without atypia is 2.0. The presence of atypia increases the relative risk to 5.1 (47). Therefore current consensus guidelines issued by American Society of Breast Surgeons recommend individualized decision to excise papillary lesion based on risk: size, symptoms of palpability and nipple discharge, and breast cancer risk factors (Table 21-2). Observed lesions should be closely followed with imaging to establish stability (48).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree