Diagnostic and Therapeutic Procedures
Libby Edwards
The evaluation and diagnosis of anogenital diseases are a process that generally requires only a limited history, careful observation, and minor office procedures. For the majority of patients, the crucial aspects of evaluation are a brief history, careful visual examination, microscopy of vaginal fluid and the scale of some skin conditions, and, at times, a fungal culture or a skin biopsy. The careful inspection of the skin requires a good light, stirrups for women, and, sometimes, simple magnification.
At times, the diagnosis of skin disease on the genitalia is obvious, with classic signs such as the crinkling white skin of lichen sclerosus. However, the typical morphology of skin disease on dry, keratinized skin often is modified in the folds of genital skin. Intertriginous areas are often somewhat red normally, and moisture, heat, and friction obscure scale and change the appearance of dermatoses. For those diseases that display objective abnormalities, the cause is nearly always infection, tumor, or noninfectious inflammation that is often immune mediated. Even when the exact diagnosis cannot be ascertained by examination or biopsy, infection and tumor can be ruled out by cultures and biopsies. Those remaining diseases are categorized by the histologic description (see Appendix) and correlated with physical findings to generate a differential diagnosis. Most visible skin disease that is not tumor or infection is corticosteroid responsive. Sometimes a definitive diagnosis cannot be made, and when easily diagnosed and dangerous conditions have ruled out, a trial of presumptive therapy is reasonable and often beneficial.
Treatment of skin diseases is often time-consuming, because careful and sensitive patient education generally is required, and attention to multifactorial processes such as secondary infection and irritant contact dermatitis is important. Except for radiation therapy and the surgical removal of growths, the therapy of most genital disorders is medical, consisting of self-administered oral and topical medications. There are several office procedures, however, including intralesional therapy, cryotherapy, and the application of topical chemotherapy such as trichloroacetic or bichloroacetic acid, podophyllum resin, or cantharidin.
Diagnostic Procedures
Most diagnostic procedures for anogenital diseases are performed in an outpatient setting or at the bedside. Only careful eyes, a microscope, glass slides and coverslips, 10% to 20% potassium hydroxide (KOH), and normal saline are required. Although these procedures are easy and quick to perform, the interpretation of the microscopic findings requires experience. Unexpected results or poor response to therapy should be followed by cultures or biopsies to corroborate the microscopic findings. Clinicians who care for women with vulvar dermatoses or vulvar pain should invest in a narrow, straight Pederson speculum, which produces far less distention of the introitus and less pain on insertion than does the standard Graves speculum with its bulbous tip. However, visualization of the cervix is more difficult with the Pederson speculum. The use of a speculum allows for visualization of the vaginal walls and sampling of vaginal fluid for microscopic examination and culture.
Cytologic Smears
Fungal Preparations
Fungal preparations are essential to the diagnosis of some anogenital diseases. Men generally interpret all anogenital itching as produced by “jock itch,” or tinea infection, whereas women assume all vulvovaginal itching to result from candidiasis. The confirmation or elimination of these conditions is vital.
KOH 10% to 20% solution is a basic agent that dissolves the keratin of epithelial cells, allowing spores and fungal hyphae and pseudohyphae to be seen more clearly. The reliability of this test depends on the choice of the lesion to be sampled, adequate dissolution of cells so fungal elements are best visualized, and the experience of the examiner in distinguishing fungal elements from artifacts such as hair, fabric fibers, cell membranes, and fractures in crusts.
A fungal preparation of vaginal fluid is much easier than that of scale from skin, because there are fewer
artifacts, and the fragile mucosal squamous epithelial cells dissolve quickly. Vaginal secretions are collected with a cotton-tipped applicator from secretions remaining on the blade of the withdrawn speculum, from a pool of secretions within the vagina (avoiding the cervical os) or by gently rolling the cotton tip along the vaginal walls.
artifacts, and the fragile mucosal squamous epithelial cells dissolve quickly. Vaginal secretions are collected with a cotton-tipped applicator from secretions remaining on the blade of the withdrawn speculum, from a pool of secretions within the vagina (avoiding the cervical os) or by gently rolling the cotton tip along the vaginal walls.
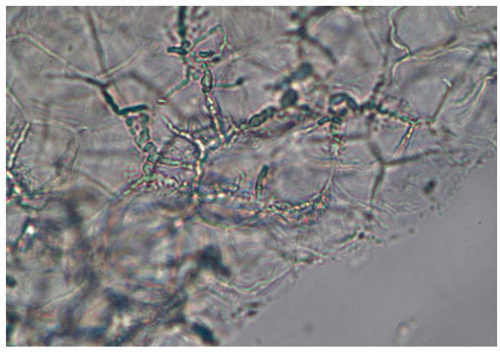
 FIG. 4.1. Dermatophytosis of tinea cruris shows hyphae similar to candidiasis, but there are no budding yeast; the hyphae are long, branching, and cross cell membranes. |
The skin specimens most likely to yield fungi include the peripheral scale from plaques of possible dermatophyte infection, pustule roofs, and white, cheesy material produced by suspected yeast. These elements are removed by scraping with the rounded surface of a number 15 scalpel blade. In the damp areas of the genitalia, the sample usually adheres to the scalpel blade and can be wiped onto the glass slide. Dry, hair-bearing skin can be moistened with water so that the moistened specimen sticks to the blade until it is smeared onto the slide.
Once the specimen is applied to the glass slide, a drop of KOH is placed on the material to dissolve the keratin from cells and to enhance the visibility of fungal elements. A coverslip is applied, and firm pressure to the coverslip with the back of a fingernail or a pencil eraser (to avoid distracting fingerprints on the coverslip) flattens and augments the dissolution of keratin. Although the unkeratinized epithelial cells in vaginal smears deteriorate quickly after exposure to KOH, the scale of keratinized skin requires further attention to dissolve so that fungi and yeasts can be easily detected. The examiner can use KOH mixed with dimethyl sulfoxide to enhance dissolution or can simply allow 10 to 15 minutes for the KOH to disintegrate the cells. In addition, gentle warming of the specimen over an alcohol flame dissolves cells from keratinized skin more quickly.
 FIG. 4.2. A and B. The hyphae and pseudohyphae of dermatophytosis and candidiasis are nearly indistinguishable, but on this higher power, budding yeast often can be seen in candidiasis. |
Fungal elements are best visualized by lowering the condenser and decreasing the light to increase the contrast between fungi elements and epithelial cells. Spores, buds, and hyphal elements appear refractile and sometimes very slightly green, and these are much smaller than common artifacts, such as hair and fibers. Dermatophytes appear as branching, septate hyphae that cross over cell membranes (Fig. 4.1). Candida appears as budding yeasts with or without hyphae or nonseptate, branching pseudohyphae (Fig. 4.2A, B). At times, cell membranes in the process of dissolving resemble hyphae or pseudohyphae (Fig. 4.3). When this is suspected, pressure on the coverslip differentiates between true hyphae/pseudohyphae and these cell membranes by further disrupting the cell membranes. The specific dermatophyte species cannot be recognized from the smear, and Candida species can be divided only into those characterized by the presence of hyphae or pseudohyphae as well as budding yeast forms (C. albicans or C. tropicalis) and yeast forms that exhibit only budding yeast (e.g., C. glabrata or C. parapsilosis, among many other) (Fig. 4.4). A culture is required to confirm the species on the rare occasion speciation is needed. Oil droplets and air bubbles can be confused with yeast buds at times, but the variability in size and very round
shape distinguish these from yeast buds (Fig. 4.5). “Tinea” versicolor (which occurs uncommonly in the genital area) is a misnomer because it is a yeast rather than a dermatophyte or “tinea.” It exhibits short, curved hyphae and spores/buds (“spaghetti and meatballs”) microscopically.
shape distinguish these from yeast buds (Fig. 4.5). “Tinea” versicolor (which occurs uncommonly in the genital area) is a misnomer because it is a yeast rather than a dermatophyte or “tinea.” It exhibits short, curved hyphae and spores/buds (“spaghetti and meatballs”) microscopically.
Saline “Wet Mount” Preparation (see also Chapter 15)
A microscopic evaluation of vaginal secretions using normal saline under a coverslip permits an evaluation of the morphology of cells, a screen for inflammation and estrogen effect, and a crude survey of colonizing and infecting organisms. Information from an examination of vaginal secretions is sometimes extremely important in the evaluation of the vulva and vagina, and recording results in a grid helps ensure that all aspects of the wet mount are evaluated (Table 4.1). Secretions are collected as described above for evaluation for Candida spp. However, the secretions are transferred from the cotton-tipped applicator by touching (if abundant) or gently rolling (if scant) onto the glass slide. Care should be taken to avoid the thick application of secretions, which interferes with visualization. Some clinicians prefer inserting the cotton-tipped applicator into a test tube and adding a few drops of saline to dilute the secretions and separate the cells to better evaluate the morphology. This author has tried both methods but has found that the evaluation of white blood cells is problematic when the vaginal secretions are first diluted. The proportion of white blood cells per epithelial cell, a standard method of measurement, is far lower with diluted specimens compared to undiluted specimens.
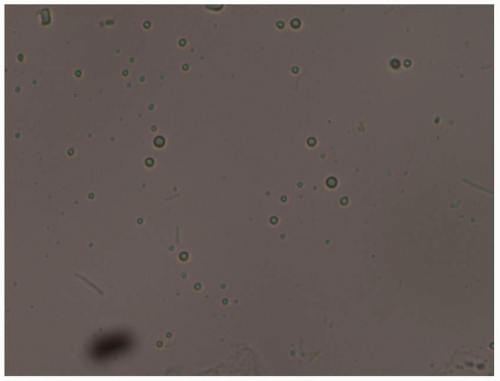
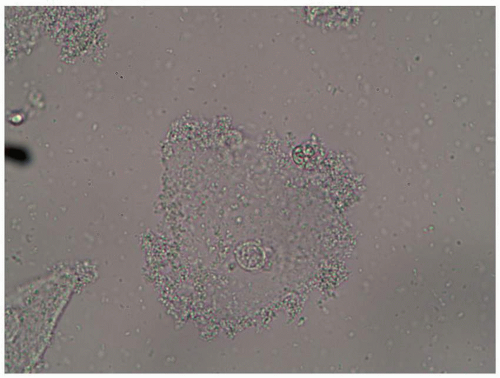
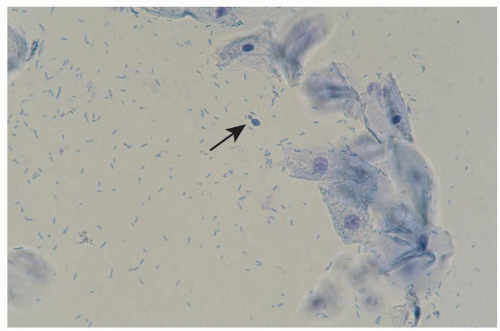 FIG. 4.4. The lack of mycelia on wet mounts of non-albicans Candida can make this diagnosis more challenging. These forms of yeast exhibit only budding yeast (arrow). |
In order to evaluate vaginal secretions for abnormalities, the examiner should be aware of the normal findings beyond the absence of yeast, clue cells, and trichomonads (Fig. 4.6). Mature epithelial cells shed from a well-estrogenized vaginal epithelium appear as large, often folded, polygonal
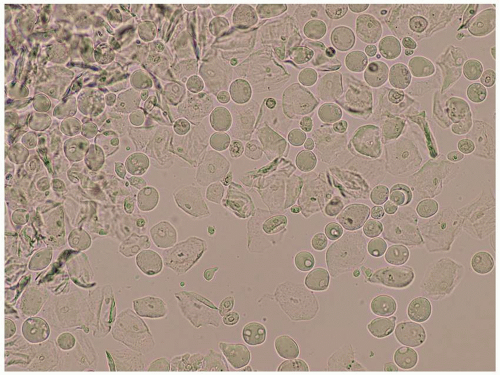
cells with abundant cytoplasm and a small, condensed nucleus. A crude maturation index can be performed, in which the degree of maturation of epithelial cells is estimated. Immature epithelial cells, or parabasal cells, are much smaller and rounder than mature cells, with proportionately larger nuclei (Fig. 4.7). These less mature cells are seen in several settings and serve as a marker for atrophic, estrogen-deficient vaginal epithelium, erosions within the epithelium, and rapidly proliferative inflamed skin. Clue cells are a distinct abnormality of epithelial cells that are pathognomonic for bacterial vaginosis. These clue cells occur when non-Lactobacillus bacteria adhere to epithelial cells and obscure the sharp borders of the cells so that the edge appears ragged and the cytoplasm appears granular (Fig. 4.8).
cells with abundant cytoplasm and a small, condensed nucleus. A crude maturation index can be performed, in which the degree of maturation of epithelial cells is estimated. Immature epithelial cells, or parabasal cells, are much smaller and rounder than mature cells, with proportionately larger nuclei (Fig. 4.7). These less mature cells are seen in several settings and serve as a marker for atrophic, estrogen-deficient vaginal epithelium, erosions within the epithelium, and rapidly proliferative inflamed skin. Clue cells are a distinct abnormality of epithelial cells that are pathognomonic for bacterial vaginosis. These clue cells occur when non-Lactobacillus bacteria adhere to epithelial cells and obscure the sharp borders of the cells so that the edge appears ragged and the cytoplasm appears granular (Fig. 4.8).
TABLE 4.1 Diagnosis of unknown skin disease | |
|---|---|
|
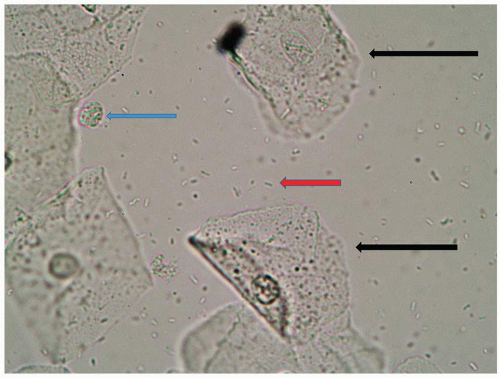
Although many different organisms are normal vaginal inhabitants, lactobacilli are the most common bacteria seen on a saline preparation of normal vaginal secretions of a well-estrogenized woman. These appear as rods of variable lengths. Occasionally, lactobacilli attach to each other from end to end, to form very long filaments, formerly believed to represent Leptothrix (Fig. 4.9). These strands are sometimes confused with the hyphae of C. albicans or C. tropicalis. However, the filaments of lactobacilli are more delicate and smaller in caliber than yeast, and these are nonbranching, when compared to the hyphae and pseudohyphae of C. albicans.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree