Blistering and Pustular Diseases
Libby Edwards
Blisters are skin lesions that are filled with fluid; these can be bullae (large blisters with clear, straw-colored fluid), vesicles (small blisters of clear fluid), or pustules (blisters containing pus). However, when blisters occur on fragile skin such as the genital area, the blister is unroofed quickly, forming erosions so that the various erosive and blistering disorders are often morphologically indistinguishable when occurring on genital skin; this chapter addresses blisters and erosive diseases that originated as blisters or pustules.
Erosions resulting from a blister are usually well demarcated and round, or when blisters coalesce before eroding, arcuate erosions can result. Often, there are blisters on surrounding or extragenital skin that provide a clue to the blistering nature.
Blistering Infections
Herpes Simplex Virus Infection
Herpes simplex virus (HSV) infection is a common sexually transmitted disease manifested by recurrent, painful, grouped vesicles that quickly erode into superficial coalescing erosions.
Clinical Presentation
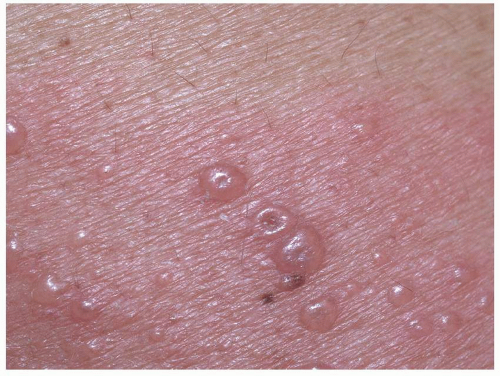
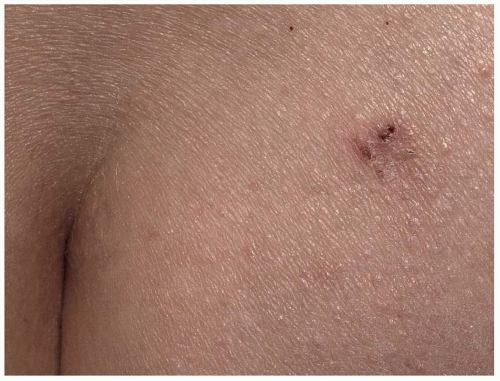
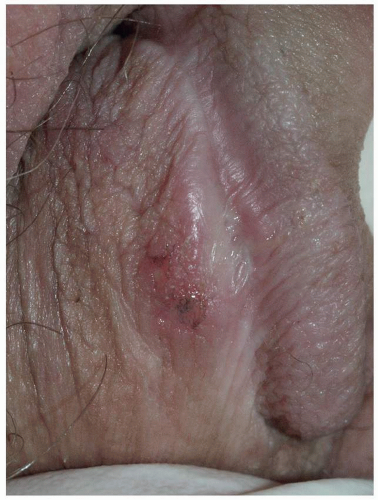
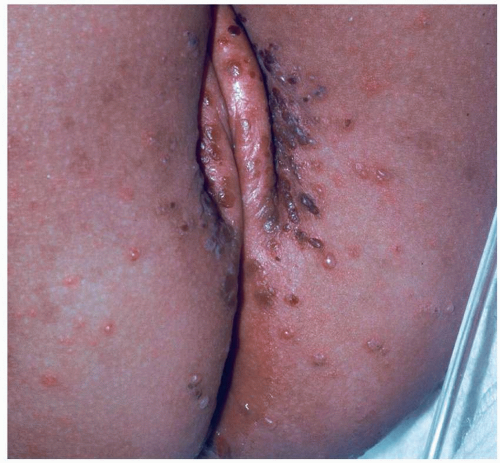
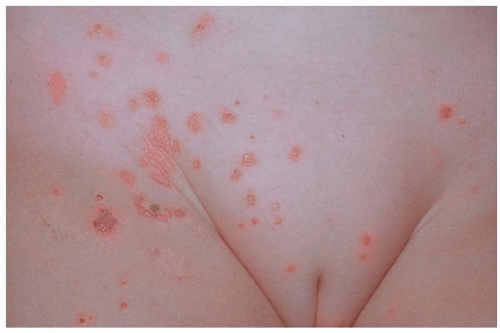
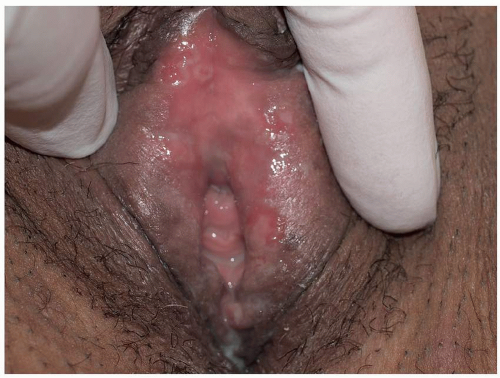
Most common in younger individuals, HSV infection is especially likely in those who have higher risks of contagion due to many lifetime sexual partners. This very common sexually transmitted disease has shown unchanging seroprevalence in the United States between 1999 and 2010 (1). There are estimates of greater than 417 million affected individuals worldwide, with the highest numbers in Africa, with large numbers also in Southeast Asia and the Western Pacific (2). HSV produces both primary and recurrent infections. Primary infection with HSV is often subclinical and unrecognized, but the classic primary infection by HSV is much more severe than recurrent episodes. Primary genital HSV infection follows exposure by 2 to 7 days and often exhibits associated fever, malaise, headache, and other constitutional symptoms. Regional lymphadenopathy is usual; pain and edema may cause urinary retention. The lesions initially occur as small (1- to 3-mm) scattered and grouped vesicles occurring typically on the glans or shaft of the penis or the mucous membrane and modified mucous membrane portion of the vulva, sometimes extending to keratinized skin (Figs. 10.1, 10.2, 10.3, 10.4, 10.5, 10.6, 10.7, 10.8 and 10.9). Because the vesicles are fragile and easily break, well-demarcated, discrete, round, and arcuate erosions (which indicate coalescing of the round erosions) often are seen in addition to intact vesicles, especially on modified mucous membrane skin. Healing may be complicated by secondary infection and irritation from overwashing or topical agents used empirically by an anxious patient.
After a primary infection, HSV remains latent in neuronal cells located in ganglia. Subsequently, the virus intermittently reactivates, producing recurrent disease that is usually milder, localized, and shorter in duration. Recurrent genital HSV infections also are less often associated with fever, arthralgias, and headache than is primary disease. Most patients experience a prodrome of localized tingling, burning, or dysesthesias before the onset of skin lesions. Recurrent HSV is most often located on the glans and shaft of the penis and the mucous membranes and modified mucous membrane portion of the vulva, but can occur on any epithelial surface, including the buttocks, sacral area, scrotum, perianal skin, and hair-bearing labia majora. Recurrent episodes are characterized by grouped vesicles that quickly develop into well-demarcated, round, and arcuate erosions. On dry, keratinized skin such as the shaft of the penis or the hair-bearing labia majora, round crusts may be the predominant lesion. Linear fissures can appear as a manifestation of HSV infection, occurring most often in skin folds, such as the interlabial sulci of the vulva or within normal skin wrinkling of the penile shaft.
Because the primary episode of HSV infection for many patients is subclinical, these people are unaware of their infection. Thus, their first clinical episode of infection exhibits the localized grouping of vesicles and erosions and milder clinical course of recurrent HSV infection. Because the appearance of this nonprimary but first clinical episode is delayed, the time and circumstances of transmission cannot be determined; on occasion, this delay is years long.
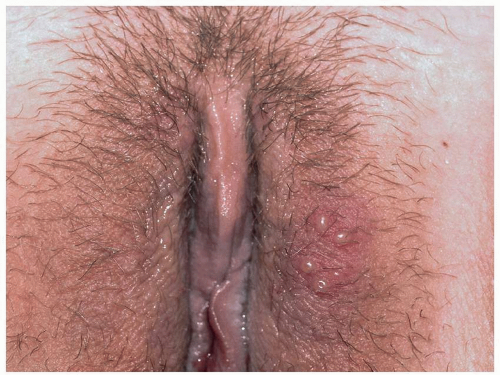 FIG. 10.1. Clustered vesicles are classic for a recurrent HSV infection; the inferior vesicle has the classic central dell of a herpes vesicle. |
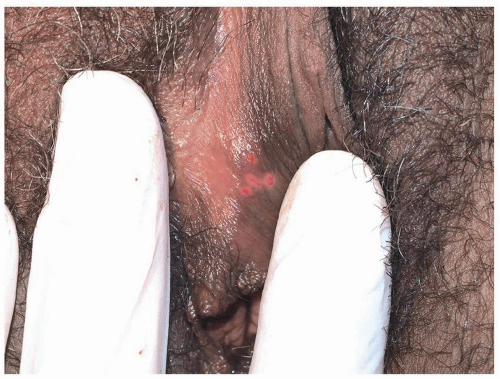 FIG. 10.6. Tiny, grouped, clustered erosions are the most common manifestation of HSV on a mucous membrane or modified mucous membrane. |
HSV infections often are associated with marked morbidity in immunosuppressed patients (see Chapter 16). Patients with altered cellular immunity, as occurs in those with HIV disease and patients receiving immunosuppressive agents, are at increased risk of ulcerative and chronic HSV infection (Figs. 10.10 and 10.11). In addition, there is a synergistic effect of HSV and HIV, with an association of HSV infection with more rapid progression of HIV disease (3).
Also, HSV has been shown to be a risk factor for bacterial vaginosis in women (4). In the past, there was concern that HSV might act as a cocarcinogen with some HPV types in the production of cervical cancer. This has not been borne out (5,6). However, the association with depression, loss of self-esteem, and shame is well recognized (7).
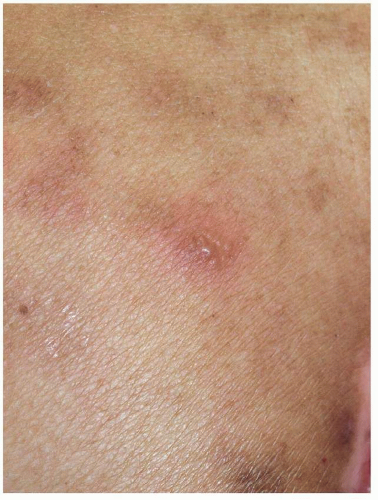 FIG. 10.8. The recurrent episodes of sacral HSV have left patches of poorly demarcated postinflammatory hyperpigmentation. |
Diagnosis
Although the diagnosis usually can be made on the appearance of skin lesions and history, laboratory confirmation of a genital HSV infection is desirable because of the psychological impact of this disease on the patient and his or her partner. A viral culture from a swab of the base
of a fresh erosion is a common but by no means the most sensitive method of detecting HSV. False-negative results are a common occurrence in many laboratories, especially when the sample is obtained late in the outbreak or delivery of the sample to the laboratory is delayed. Therefore, a negative culture should not convince a suspicious clinician to eliminate the diagnosis. The Western blot and polymerase chain reaction techniques are now widely available and reasonably priced, so these are tests of choice. Although more uncomfortable, a shave skin biopsy from the edge of an erosion or of an intact vesicle is another very sensitive test to confirm the presence of a herpesvirus infection, but a biopsy does not differentiate HSV from varicella-zoster virus (VZV). The Tzanck preparation, although very quick, is less reliable, even in experienced hands. Direct fluorescent antibodies are available for HSV1 and 2 for more rapid diagnosis, although an adequate sample is of utmost importance (8). Scraping the base of the ulcer with a number 15 blade usually suffices for an adequate cell sample. Serology is not an adequate means of diagnosing an episode of HSV. Positive IgG for HSV varies very widely according to the country, occurring in up to 80% of adults; this denotes exposure, but not current, active disease. Negative serology indicates no past infection, but does not rule out a primary episode, since about 6 weeks is required for conversion.
of a fresh erosion is a common but by no means the most sensitive method of detecting HSV. False-negative results are a common occurrence in many laboratories, especially when the sample is obtained late in the outbreak or delivery of the sample to the laboratory is delayed. Therefore, a negative culture should not convince a suspicious clinician to eliminate the diagnosis. The Western blot and polymerase chain reaction techniques are now widely available and reasonably priced, so these are tests of choice. Although more uncomfortable, a shave skin biopsy from the edge of an erosion or of an intact vesicle is another very sensitive test to confirm the presence of a herpesvirus infection, but a biopsy does not differentiate HSV from varicella-zoster virus (VZV). The Tzanck preparation, although very quick, is less reliable, even in experienced hands. Direct fluorescent antibodies are available for HSV1 and 2 for more rapid diagnosis, although an adequate sample is of utmost importance (8). Scraping the base of the ulcer with a number 15 blade usually suffices for an adequate cell sample. Serology is not an adequate means of diagnosing an episode of HSV. Positive IgG for HSV varies very widely according to the country, occurring in up to 80% of adults; this denotes exposure, but not current, active disease. Negative serology indicates no past infection, but does not rule out a primary episode, since about 6 weeks is required for conversion.
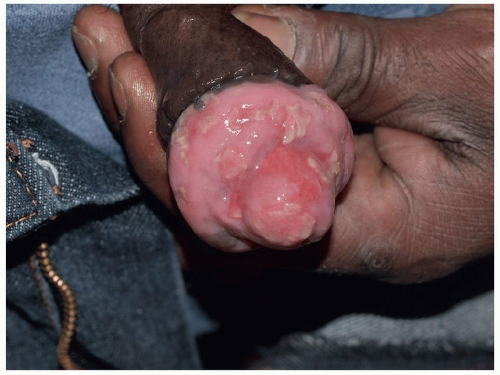 FIG. 10.10. HSV in an immunosuppressed patient is not self-limited, but rather enlarges relentlessly losing all semblance of its original blistering nature. |
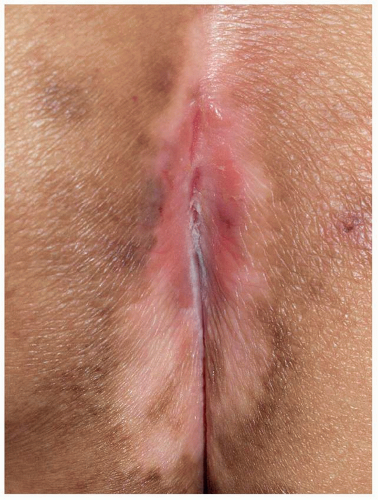 FIG. 10.11. Occasionally, HSV appears as a fissure, generally within skin lines. Often, a careful examination shows a larger, vaguely oval erosion suggesting an unroofed vesicle. |
The histologic appearance of HSV includes an intraepidermal vesicular dermatitis formed by acantholysis. Individual keratinocytes show intracellular edema (ballooning) and reticular degeneration (intracellular edema causing cell walls to burst). Eosinophilic intranuclear inclusion bodies may be present. Multinucleated keratinocytes are pathognomonic for herpesvirus infections but do not distinguish between HSV and VZV infections.
HSV must be differentiated from several other vesicular and pustular diseases. Occasionally, the differentiation of genital HSV infection from VZV infection can be difficult, because both exhibit grouped vesicles or erosions. However, genital VZV infection usually occurs in the older patient and covers a dermatomal pattern, also affecting the medial thigh or buttock unilaterally. VZV infection occurs only once in the immunocompetent patient, rather than recurrently, as is usual with HSV infection. Another likely disease to confuse with genital HSV infection is candidiasis of the modified mucous membranes of the vulva or the uncircumcised glans penis, in which yeast can cause coalescing erosions when superficial pustules rupture. A fungal smear is positive in that case. Folliculitis, both irritant and staphylococcal, produces discrete red papules and pustules that can mimic HSV. Erythema multiforme and Stevens-Johnson syndrome both mimic and sometimes follow HSV infection. The most common cause of recurrent erythema multiforme is recurrent HSV infection in a patient with an enhanced immunologic response to this virus. These patients usually exhibit both intraoral and genital erosions that are less well demarcated, whereas HSV does not occur inside the mouth, except for the initial primary outbreak. In addition, the occurrence of red, nonscaling, flat-topped papules on the palms, soles, and sometimes other keratinized surfaces is common. Finally, a fixed drug eruption can mimic HSV infection by producing same-site recurrent blisters or erosions, but the lesions are generally larger rather than showing small coalescing vesicles or erosions.
HERPES SIMPLEX VIRUS INFECTION
Diagnosis
Morphology:
Primary, first-episode HSV with scattered and grouped vesicles and round erosions on genital and perianal skin
Recurrent with grouped vesicles or erosions
Confirmation by biopsy, polymerase chain reaction technique, or direct fluorescent antibody
Pathophysiology
HSV types 1 and 2 are double-stranded DNA viruses (Herpesvirus hominis) capable of producing vesicular and erosive mucocutaneous disease. Oral mucocutaneous and ocular disease is produced by HSV1 in most cases, whereas genital HSV infection is caused by HSV2 in the majority of affected patients. The proportion of HSV1 occurring on the genital skin is increasing, presumably due to the increase in oral-genital sexual activity. In fact, in one area of Canada, a recent report found genital HSV
associated with HSV1 by culture in more individuals than HSV2 (9). Fortunately, genital disease produced by HSV1 is less severe and less frequently recurrent than is that produced by HSV2, and neonatal HSV is less problematic in women with HSV1.
associated with HSV1 by culture in more individuals than HSV2 (9). Fortunately, genital disease produced by HSV1 is less severe and less frequently recurrent than is that produced by HSV2, and neonatal HSV is less problematic in women with HSV1.
Management
The management of HSV infection begins with the sensitive and nonjudgmental education of the patient. The patient should be counseled regarding the infectious nature of the disease, its recurrent nature, and the importance of avoidance of sexual intercourse when open lesions are present. Circumcision and condoms have been shown to confer modest protection from infection. Patients should be aware that intermittent shedding of the virus occurs when there is no evidence of active HSV infection, and the infection can be transmitted even in the absence of blisters and erosions. In fact, most HSV infection is contracted during asymptomatic episodes. This shedding has also been documented in patients receiving long-term suppressive antiviral medication. Therefore, the use of a condom, even when there are no obvious lesions, is wise and confers significant protection (12). The possibility of transmission of HSV to a newborn during delivery should be discussed to ensure the protection of future infants from maternal infection, although most neonatal HSV infections occur due to maternal primary infection.
Both primary and recurrent primary HSV infections are shortened by prompt treatment with specific oral antiviral agents. Three current choices for the treatment of recurrent HSV are as follows: 5 days of acyclovir 200 mg five times a day (or, for primary HSV infection, for 10 days), famciclovir 125 mg twice daily, and valacyclovir 500 mg twice daily (1 g twice daily for 10 days for primary HSV infection). These therapies exhibit equivalent efficacy, with choices made on the basis of cost and convenience of dosing. Side effects are minimal with each. Topical acyclovir is not beneficial, and the advantage of topical penciclovir over placebo for shortening the course of HSV infection is minimal (13).
Intravenous acyclovir is occasionally needed if disease is severe, especially if the patient is immunosuppressed or if gastrointestinal absorption is a major problem, as can occur in the HIV-positive patient. Resistance of HSV to currently available oral medications occurs only occasionally and always in immunosuppressed patients (see Chapter 16). Foscarnet is an alternative treatment, although it is available only for intravenous use. Several other agents have been reported as beneficial in acyclovirresistant HSV, including intravenous and topical cidofovir, topical trifluorothymidine, topical interferon-α in dimethyl sulfoxide, and imiquimod.
For patients experiencing frequent or severe episodes of HSV infection, chronic prophylactic acyclovir at a dose of 400 mg twice daily, famciclovir 250 mg twice daily, or valacyclovir 500 to 1,000 mg/day is a safe, practical, and effective means of preventing most recurrences. Suppressive oral antiviral therapy also decreases asymptomatic shedding and, therefore, probably infectivity, but asymptomatic shedding of virus is not eliminated. Transmission of HSV infection still occurs in patients taking these medications and experiencing no clinical outbreaks.
Therapeutic vaccines for patients with problematic HSV infections are being investigated and show some promise (14,15).
Crucial to the management of anogenital HSV infection is counseling and support. These patients exhibit marked anxiety, depression, fear, and self-esteem issues. There is even evidence that stress and depression increase shedding and the frequency of outbreaks (16,17).
Some patients erroneously believe that HSV infection can be followed by postherpetic neuralgia. Fortunately, that is not a sequel to this infection; this only follows herpes zoster infection. Primary HSV infection can produce malaise and fever, and sometimes outbreaks can create radicular pain. Very rarely, HSV can cause HSV encephalitis.
HERPES SIMPLEX VIRUS INFECTION
Management
Patient education
Antiviral therapy:
Acycovir 400 mg t.i.d. or 200 mg 5× per day for 5 days (recurrent HSV) or 7 to 10 days (primary),
Valacyclovir 500 mg b.i.d. for 5 days (recurrent HSV) or 1 g b.i.d. for 7 to 10 days (primary)
Famciclovir 125 mg b.i.d. for 5 days (recurrent HSV) or 250 b.i.d. for 7 to 10 days (primary)
Suppression with chronic:
Acyclovir 400 mg b.i.d.
Valacyclovir 500 mg—1 g q d
Famciclovir 250 mg b.i.d.
Herpes Zoster Infection (Shingles)
VZV causes two diseases: herpes zoster, or shingles, and chickenpox. Both affect the genitalia but present very differently. The first episode of VZV (varicella, chickenpox) presents with generalized distribution of discrete red papules, vesicles, and crusts, but there is a predilection for the genital skin and vagina. Shingles, the second episode of
VZV is localized, typically very painful, and with minimal constitutional symptoms. Hopefully, with the availability of the varicella and shingles vaccines, these conditions will become uncommon.
VZV is localized, typically very painful, and with minimal constitutional symptoms. Hopefully, with the availability of the varicella and shingles vaccines, these conditions will become uncommon.
Clinical Presentation
Herpes zoster occurs primarily in patients with immune deficiency by virtue of age, disease, or medications but occasionally occurs in young and healthy individuals. The prevalence of this miserable condition should decrease as more older people receive the recently available vaccine.
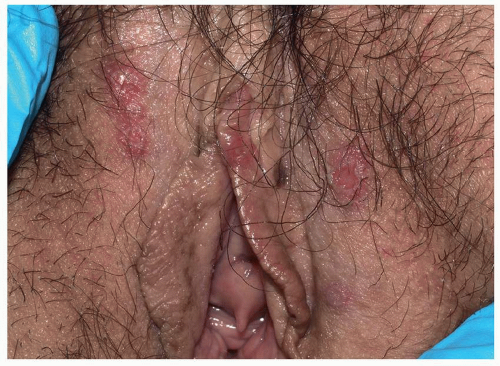
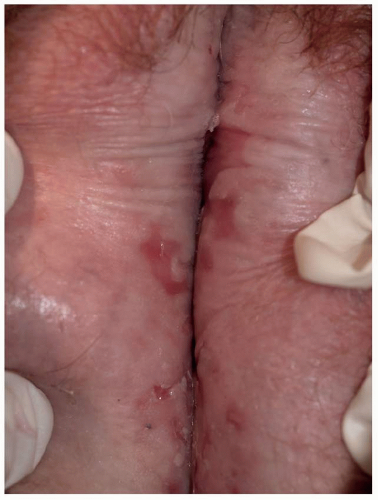
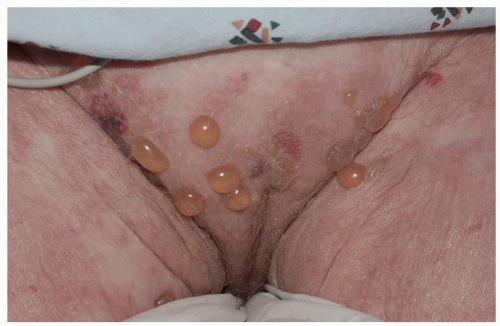
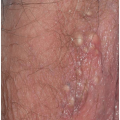
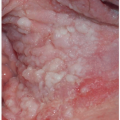
Many patients experience a localized prodrome of pain, itching, burning, or aching, followed in one or a few days by pink, nonscaling papules and plaques in a unilateral dermatomal distribution (Figs. 10.12 and 10.13) in the symptomatic area. Grouped vesicles overlying these red plaques quickly follow and then coalesce into larger blisters. Often, the blisters become purple or gray due to hemorrhage into the blister. Over the next 3 weeks, the blisters crust and heal. Blisters occurring on mucous membranes and modified mucous membranes of the genitalia are quickly unroofed due to friction on this moist and very thin skin, so that erosions usually predominate. Likewise, the friction from skin folds and clothing often tears blister roofs even on the more keratinized genital skin. Herpes zoster can become disseminated, ulcerative, chronic, or hyperkeratotic in immunosuppressed patients (see Chapter 16).
Sometimes, the eruption is accompanied by mild headache, malaise, and a low-grade fever. However, the primary complication of herpes zoster infection is the development of postherpetic neuralgia, in which chronic pain persists after the eruption resolves. This is more likely in individuals over 60 years of age and in immunosuppressed people. Some clinicians theorize that this may be responsible for some genital pain syndromes such as vulvodynia and scrotodynia, but preceding VZV infection affecting the genitalia is almost never identified in patients with genital pain syndromes. Unlike HSV, VZV does not recur in immunocompetent patients.
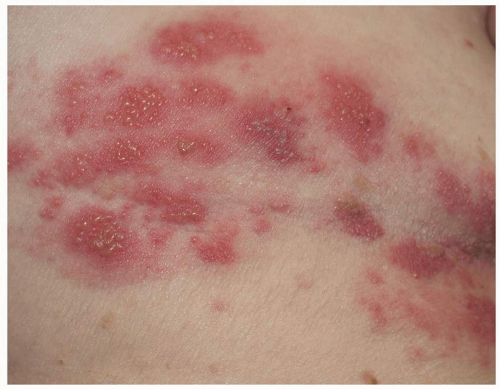 FIG. 10.12. Clustered and coalescing vesicles on a red base, distributed along a dermatome, are characteristic of herpes zoster. |
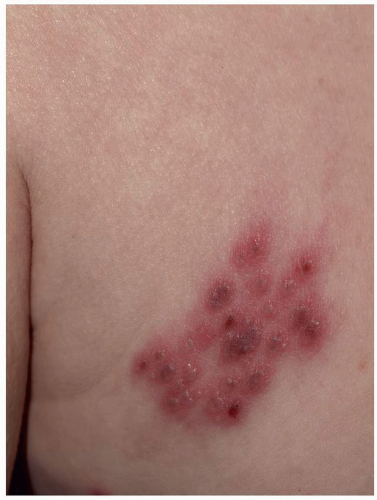 FIG. 10.13. The vesicles of herpes zoster arise from a deeper red base than do those of HSV, and bleeding into the vesicles of herpes zoster sometimes gives them a gray or purple appearance. |
Varicella (chickenpox) is now rare due to availability of the varicella vaccine but presents with fever and constitutional symptoms and then a generalized eruption of vesicles. These begin as small red papules and then evolve into vesicles with a central depression and then a central crust. Mucous membranes are often involved (Fig. 10.14).
Diagnosis
The definitive diagnosis of herpes zoster is usually made on the basis of the presentation and morphology. When necessary, this can be confirmed by a positive culture or the identification of viral DNA with the PCR technique. The virus is rather fastidious, so cultures are sometimes falsely negative, but the PCR technique is very sensitive and now easily available. The Tzanck preparation from epithelial cells scraped from the base of a blister or erosion shows giant cells when examined microscopically, but the validity of this test is extremely dependent on the examiner, and the results are subjective. A biopsy is extremely sensitive but does not differentiate between HSV infection and herpes zoster, a distinction that can usually be made on clinical grounds. The histologic appearance of herpes zoster infection is identical to that of HSV infection. Keratinocytes swell and burst, and some epithelial cells form giant cells. Occasionally, leukocytoclastic vasculitis underlies these epithelial changes, but this does not indicate a primary or possibly systemic problem of vasculitis.
The differentiation of genital herpes zoster infection from HSV infection may be difficult. HSV is a recurrent disease, whereas herpes zoster is a one-time event, except in significantly immunosuppressed patients. Herpes zoster is unilateral, but vesicles may erode early in both diseases. Bullous impetigo and bullous pemphigoid can sometimes mimic herpes zoster, but these are usually bilateral and are not particularly painful. Bullous pemphigoid generally exhibits distant lesions that exclude the diagnosis of herpes zoster, and impetigo can be differentiated by culture.
HERPES ZOSTER VIRUS INFECTION
Diagnosis
Morphology of plaques of clustered vesicles in a dermatomal distribution
Confirmation by PCR if needed
Pathophysiology
Herpes zoster infection is a blistering disease that results from reactivation of VZV that has been latent in nerve ganglia since a remote episode of varicella. This condition occurs most often in older individuals whose immune system is less efficient or in immunocompromised patients.
Management
For most patients with herpes zoster, pain control is the most important aspect of care. Narcotic pain analgesia is usually needed for the first few weeks. The early diagnosis and treatment (within 72, but especially 48 hours) with oral antiviral therapy somewhat reduces the duration of an acute infection. This improvement is not striking, so therapy is optional in younger, healthy patients. Treatment after this time does not affect the short-term or long-term course of the disease. However, immunosuppressed or very elderly people should be treated. Some studies have shown an arguably significant reduction of postherpetic neuralgia in patients treated early.
Choices include acyclovir 800 mg five times a day, famciclovir 500 mg three times daily, and valacyclovir 1 g every 8 hours. The advantage of the more expensive valacyclovir and famciclovir is the less frequent dosing schedule.
Those patients who experience postherpetic neuralgia deserve medication for neuropathic pain, including amitriptyline, gabapentin, pregabalin, venlafaxine, or duloxetine (see Chapter 5). Sometimes, referral to a pain clinic is necessary.
Otherwise, the best current management is prevention. A vaccine for the prevention of herpes zoster is now available, approved by the Food and Drug Administration for people over 60 years of age.
HERPES ZOSTER VIRUS INFECTION
Management
Patient education
Antiviral therapy:
Acyclovir 800 mg 5× per day for 7 to 10 days
Valacyclovir 1 g q 8 h for 7 to 10 days
Famciclovir 500 mg t.i.d. for 7 to 10 days
Pain control
Impetigo
Impetigo is a bacterial infection of the superficial epidermis that can produce blisters and erosions. Some phage types of Staphylococcus aureus produce separation of the stratum corneum, and rapid loss of this fragile blister roof ensue. Well-demarcated, round erosions with collarettes are typical (Fig. 10.15). When produced by α-hemolytic Streptococcus infection, erosion and crusting commonly occur instead of vesicles. Impetigo is often associated with bacterial folliculitis, infection that has extended into nearby hair follicles, producing discrete red papules, pustules, and small erosions and crusts. The diagnosis is suspected by the morphology and is confirmed by a bacterial culture and response to therapy. Treatment consists of an antibiotic effective against both S. aureus and Streptococcus species, such as cephalexin, erythromycin, dicloxacillin, clindamycin, and trimethoprim-sulfamethoxazole. Unfortunately, resistant S. aureus has become common so that empiric therapy is often ineffective, and culture with sensitivities has become important. Despite prompt response to therapy, lesions sometimes recur after therapy. This occurs most often in patients who are nasal carriers of S. aureus, and these patients benefit from the administration of intranasal mupirocin cream or ointment four times a day, for 1 week each month for several months to minimize this carrier state.
Noninfectious Blistering Eruptions
Noninfectious blistering diseases primarily consist of autoimmune or hypersensitivity reactions, or chemical or thermal trauma. They cause blistering and erosions of skin and mucosal surfaces including the genitalia.
In autoimmune blistering diseases, autoantibodies are directed against target antigens that are involved in helping epithelial cells adhere to each other or to the basement membrane. Such antibodies fix to the target antigen and trigger complement activation through the classical pathway. As with other blistering processes, lesions of mucous membranes and modified mucous membranes are so fragile that blisters are so transient as to be unnoticed, and the usual presentation is that of erosion. However, most patients also exhibit blisters on keratinized skin surfaces, announcing the bullous nature of the skin disease.
Histology is essential for a correct diagnosis. Biopsies can be obtained from cutaneous or mucosal sites from the edge of a blister or erosion for routine histology and from adjacent normal epithelium for direct immunofluorescent studies.
Pemphigus
Clinical Presentation
Pemphigus affects the skin and mucous membranes in people of middle and old age (mean age is the sixth decade). Childhood cases are rare.
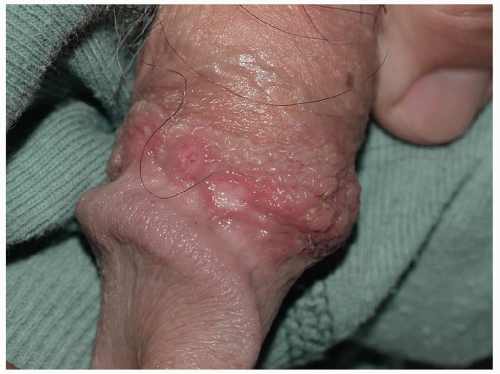
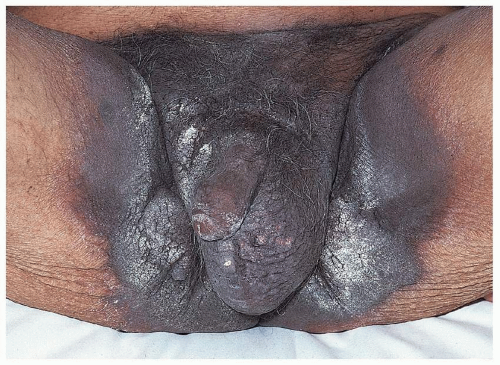
Pemphigus vulgaris (common pemphigus) accounts for 80% of all patients with pemphigus. Of these, 60% present with oral lesions that may produce extensive erosion, and months may elapse before keratinized skin lesions form. More than 90% of patients with pemphigus vulgaris suffer mucosal involvement at some time during the course of the disease; genital involvement is common, occurring in about 41% patients, and 35% of women exhibit Pap smears with acantholysis and inflammation consistent with pemphigus, although these findings alone do not denote this diagnosis (18). Mucosal sites reported include nose, pharynx, esophagus, conjunctiva, cervix, vagina, and rectum, and modified mucous membranes affected include the vulva. Blisters of pemphigus occurring on these areas quickly erode, so that the first recognized lesions usually are well-demarcated, bland, nonspecific, uniform erosions (Figs. 10.16, 10.17 and 10.18). Although pemphigus is generally
a nonscarring disease, the severely affected vulva exhibits resorption of the labia majora and the clitoris is covered by scar, and the uncircumcised penis may develop phimosis (Fig. 10.19).
a nonscarring disease, the severely affected vulva exhibits resorption of the labia majora and the clitoris is covered by scar, and the uncircumcised penis may develop phimosis (Fig. 10.19).
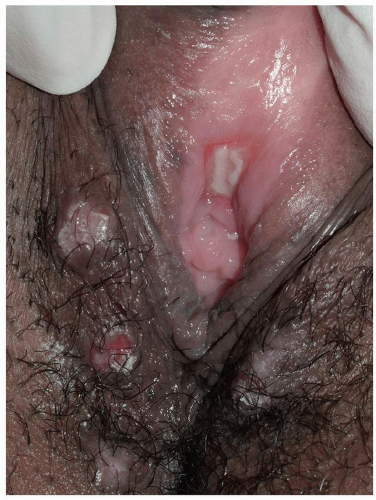 FIG. 10.17. A larger erosion of pemphigus vulgaris shows a yellowish fibrin base, and the lesions that extend to dry, keratinized skin are covered by a white necrotic blister roof. |
Pemphigus vulgaris of the penis is less common than is pemphigus of the vulva, and when present, it occurs most often on the glans, but the distal shaft and corona also are sites that are affected. The oral mucosa is usually affected in both men and women (Fig. 10.20).
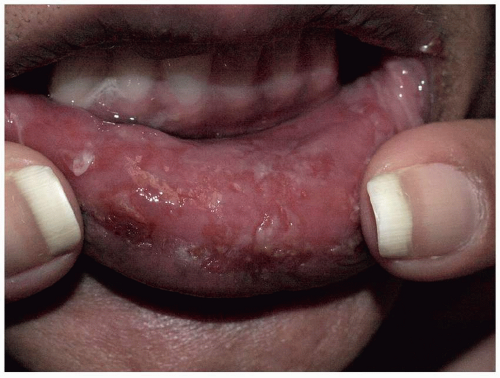 FIG. 10.20. As found in many blistering diseases, genital pemphigus vulgaris is generally accompanied by oral disease, manifested here by erosions of the lip. |
Blisters on non-mucous membrane skin are superficial and flaccid and arise from noninflamed skin. With gentle traction on a blister, the tenuous adherence of the upper epidermis to underlying tissue allows the blister to extend (Nikolsky sign). Even on keratinized skin, these very superficial blisters rupture easily to leave large eroded areas, but they normally heal without scarring. However, the erosions of pemphigus vulgaris on the vulva produce resorption of the labia minora and clitoral hood, and pemphigus of the uncircumcised glans penis can cause phimosis. Comorbidities of hypothyroidism and diabetes have been reported with pemphigus (19).
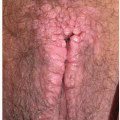
Pemphigus vegetans is a variant characterized by erosions with peripheral pustules in the early phases of the disease. Later, large, vegetating, and almost verrucous plaques occur (Figs. 10.21 and 10.22).
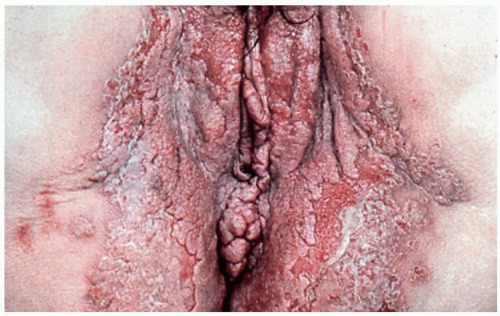 FIG. 10.21. Pemphigus vegetans produces thickened, hyperkeratotic plaques in areas of previous erosion and blistering. |
Pemphigus foliaceus presents in older patients as superficial crusting plaques, often on the central trunk and in skin folds. The blistering nature can be identified by a careful observer by the well-demarcated and arcuate borders and by the realization that crusting is often a sign of blistering. This form of pemphigus generally spares mucous membranes, so the dry, keratinized skin of the genitalia is more likely than is moist mucous membrane skin to be affected.
Diagnosis
The diagnosis is made from characteristic histologic changes on biopsies for routine microscopy and direct immunofluorescence. A skin biopsy shows an intraepidermal blister. Pemphigus vulgaris and early pemphigus vegetans exhibit a blister located just above the basal cell layer. The basal cells adhere to the basement membrane, but not to each other or to overlying cells, thus producing a picture similar to that of a row of tombstones. In addition, within the blister cavity are detached epidermal cells that, with loss of cohesion to surrounding cells, appear round and are called acantholytic cells. Direct immunofluorescence biopsies obtained from normal-appearing skin near a blister or erosion show immunoglobulin G (IgG) deposition in the epidermal intercellular substance. In addition, indirect immunofluorescence of patients’ serum showing antibodies that bind to the epidermal cell surface is characteristic of this disease. The patients’ serum contains IgG autoantibodies directed against the glycoproteins desmogleins and desmocollins in the desmosomes of stratified squamous epithelium.
Biopsies of pemphigus vegetans often show, in addition, neutrophilic inflammation producing intraepithelial abscesses. In its latter, hyperkeratotic form, pemphigus vegetans reveal squamous hyperplasia and pseudoepitheliomatous hyperplasia on biopsy, in addition to the characteristic suprabasilar blister. Pemphigus foliaceus shows a very superficial blister formed by acantholysis in the uppermost epidermis.
Pemphigus can be confused with most other blistering diseases. Pemphigus vulgaris often begins with mucosal erosions indistinguishable from erosive lichen planus. Routine biopsies usually differentiate the two diseases, and pemphigus vulgaris progresses to produce blisters and erosions on keratinized skin, unlike in lichen planus. Pemphigus vulgaris can mimic mucous membrane pemphigoid, with both producing nonspecific mucosal erosions. The milder and intermittent occurrence of a fixed drug eruption and the explosive onset of blistering forms of erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis (TEN) usually distinguish these diseases from pemphigus.
PEMPHIGUS VULGARIS
Diagnosis
Morphology of mucosal erosions, flaccid blisters of dry, keratinized skin
Confirmation by routine and direct immunofluorescent biopsies
Pathophysiology
Pemphigus is a group of autoimmune intraepidermal bullous diseases produced by autoantibodies to the surface of epidermal cells. This ultimately produces a loss of adhesion of these cells that leads to a superficial blister within the epidermis. The most common and dangerous form of pemphigus, pemphigus vulgaris, is produced by loss of adhesion of the basal cells from the upper epidermis. Like pemphigus vulgaris, pemphigus vegetans is produced by a split in the epidermis just above the basal cell layer, but it is characterized by the later development of thickened, hyperkeratotic skin. Pemphigus foliaceus occurs when cells in the upper epidermis lose cohesion and form such a superficial blister that the blistering nature is often misdiagnosed clinically as scaling or crusting.
Both drug-induced pemphigus and paraneoplastic pemphigus are reported. Mediations most often implicated are penicillamine and angiotensin-converting enzyme inhibitors, captopril and enalapril (20,21). Injury has also precipitated pemphigus at times (22,23). One form of pemphigus, benign familial pemphigus (also called Hailey-Hailey disease), is of autosomal dominant rather than autoimmune origin, and it is discussed later in this chapter.
Management
Genital pemphigus generally is not controlled with topical therapy. This usually widespread disease is treated primarily systemically. The mainstay therapy of pemphigus is systemic corticosteroids, using prednisone or its equivalent beginning at 60 to 80 mg/day.
Because of the danger of long-term systemic corticosteroids, adjuvant, steroid-sparing therapy has a major role in pemphigus treatment, at least in reducing the risk of relapse. Current medications used include azathioprine, mycophenolate mofetil, cyclophosphamide, cyclosporine, intravenous immunoglobulin, plasma exchange, and infliximab (24).
Most recent, most exciting, and by far the most expensive are intravenous rituximab, intravenous immunoglobulin, and a combination of the two, which have produced long-standing remission in a number of patients (25,26,27).
For the genital area, local area is important for comfort and to prevent scarring. Topical corticosteroid therapy in addition to systemic therapy may be useful. Intralesional rituximab has been reported as useful (28). Measures to prevent secondary infection, both bacterial and candidal, are also important. Unlike many skin diseases in men, pemphigus is not cleared with circumcision and requires medical therapy. Women with vaginal involvement should insert dilators regularly to prevent vaginal adhesions, and uncircumcised men should retract the foreskin daily and apply petroleum jelly to prevent phimosis.
The prognosis of pemphigus vulgaris has improved markedly since the use of corticosteroids. In the past, the disease itself or fulminant secondary infection led inexorably to death in the majority of cases. Current treatment has decreased mortality to 10%, and morbidity most often now results from adverse reactions to medication. Generally, after pemphigus is controlled, medications can be tapered to lower doses. Occasionally, medications can even be discontinued. However, recurrence is common and may occur after several years in remission.
PEMPHIGUS VULGARIS
Management
Prednisone 40 to 60 mg/day initially
Steroid-sparing adjuvant medication
Cyclophosphamide, azathioprine, methotrexate, mycophenolate mofetil
Infliximab
Rituximab
IVIG
Local care to prevent scarring
Bullous Pemphigoid
Clinical Presentation
Bullous pemphigoid is the most common autoimmune blistering disease. It occurs at any age but most commonly presents in adults older than 60 years. Men and women are affected equally. Genital involvement is relatively uncommon, occurring in about 10% of patients, with the keratinized epithelium more likely to be affected than the mucous membranes.
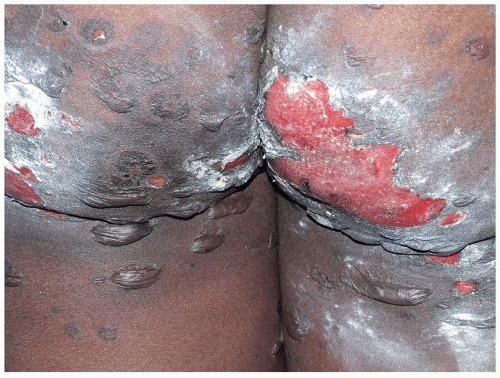 FIG. 10.23. Bullous pemphigoid exhibits tense blisters that can coalesce into larger lesions that tear, leaving denuded areas. |
Pruritus is intense and may precede the blisters for months. In addition, before clinically obvious blisters occur, many patients exhibit pink plaques that mimic urticaria. Subsequently, small vesicles and then bullae arise from the inflamed plaques. Clinically, skin involvement may be minor or very extensive. The areas most likely to be involved are the hair-bearing areas of inner thighs, inguinal crease, and perineum. Blisters are tense and filled with straw-colored and, occasionally, hemorrhagic fluid (Figs. 10.23, 10.24 and 10.25). Scarring does not occur in the absence of a complicating event such as secondary infection, since bullous pemphigoid does not affect mucous membranes.
An association of bullous pemphigoid with neurologic disease, especially dementia and Parkinson disease, has
been described (29). There is a controversial association of bullous pemphigoid with underlying malignancy (30). A recent survey reports that hematologic malignancies in patients with bullous pemphigoid are increased compared to matched controls (p < 0.0001) (31). Other reports find no association with malignancies (32).
been described (29). There is a controversial association of bullous pemphigoid with underlying malignancy (30). A recent survey reports that hematologic malignancies in patients with bullous pemphigoid are increased compared to matched controls (p < 0.0001) (31). Other reports find no association with malignancies (32).
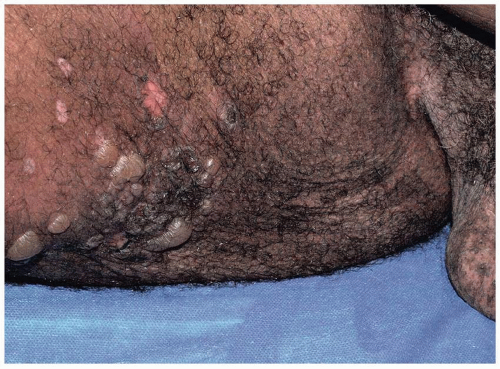 FIG. 10.24. Bullous pemphigoid affects the keratinized skin, generally sparing the mucous membranes and modified mucous membranes of the genitalia. |
Diagnosis
A provisional diagnosis of bullous pemphigoid can be made on the basis of the morphology of the lesions and characteristic onset in elderly patients. However, confirmation by biopsy for routine histology and direct immunofluorescence is mandatory. Biopsy of an intact new blister shows characteristic histology; subepidermal blistering with a variable dermal inflammatory infiltrate including eosinophils is usual. Direct immunofluorescent biopsy of perilesional skin shows in vivo deposition of IgG and complement components along the basement membrane zone (BMZ). A direct immunofluorescent examination of salt-split skin shows that the binding is epidermal to the roof of the blister. This differentiates bullous pemphigoid from epidermolysis bullosa acquisita, in which the binding is to the base or dermal side of the blister. Indirect immunofluorescent studies of the sera of patients with bullous pemphigoid show the presence of IgG directed toward the pemphigoid target antigens in the lamina lucida of the BMZ.
Most other blistering diseases can be considered in the differential diagnosis of bullous pemphigoid. Stevens-Johnson syndrome and TEN are sometimes difficult to distinguish, but these essentially always exhibit mucosal lesions, and the onset is generally more abrupt. Except for the absence of scarring, bullous pemphigoid is often indistinguishable from EBA, both clinically and histologically. A direct immunofluorescent study on salt-split skin is required for absolute differentiation of these two diseases. Pemphigus vulgaris, unlike bullous pemphigoid, generally affects mucous membranes prominently and is characterized by erosions and flaccid bullae rather than by tense blisters. Less often, bullous impetigo can mimic bullous pemphigoid. However, blisters are usually few in number and flaccid and of sudden and recent onset. Mucous membranes are spared.
Pathophysiology
This autoimmune blistering disease occurs when autoantibodies are directed at a portion of the (BMZ), ultimately causing detachment of the epidermis from the dermis. Although usually occurring spontaneously and without an identifiable precipitating factor, bullous pemphigoid is sometimes associated with specific drugs, such as furosemide, penicillins, psoralens, ibuprofen, and some angiotensin-converting enzyme inhibitors. In addition, some systemic diseases appear to be linked, such as diabetes mellitus (perhaps by increased glycosylation of BMZ components), multiple sclerosis, and rheumatoid arthritis. Studies to establish such links are difficult because bullous pemphigoid is rare and affects the elderly in whom concomitant disease is more likely. Although an association of bullous pemphigoid with internal malignancy was reported in the past, more recent studies showed no or very little increase of malignancy compared with age- and sex-matched controls.
Management
For localized and prebullous disease, treatment with potent topical steroids sometimes is sufficient, especially in the elderly and those for whom systemic corticosteroids are especially dangerous (33). For generalized bullous pemphigoid, systemic steroids are generally the treatment of choice, however, required in doses varying from 40 to 60 mg/day, tapered according to response. Steroidsparing immunosuppressive agents, whose effect is more delayed, are usually added; these include azathioprine, cyclophosphamide, or minocycline with nicotinamide. Methotrexate has also been used as a steroid-sparing agent. More recently, the biologic medications, including omalizumab have been used (34). As for pemphigus vulgaris, IVIG and rituximab are used, and the combination appears clearly superior (35). The astronomical cost is limiting for the use of this non-FDA approved, and therefore often uncovered, therapy.
In addition to this specific treatment for bullous pemphigoid, local care of the affected genitalia is important. Infection control and topical corticosteroids can minimize discomfort. Because mucous membranes are rarely affected, vaginal synechia does not occur.
The lesions in bullous pemphigoid are nonscarring, and the course of bullous pemphigoid is self-limiting, with remission in treated patients occurring in 2 to 6 years. Partly because bullous pemphigoid occurs primarily in the elderly, and because of the toxicity of therapy, the overall mortality of bullous pemphigoid in the first year
or two after diagnosis is 15% to 20% (36). Although treatment does not significantly improve mortality, it improves the quality of life, which can be ruined by the intractable and miserable pruritus. The condition may recur, but recurrences are usually milder than the initial bout.
or two after diagnosis is 15% to 20% (36). Although treatment does not significantly improve mortality, it improves the quality of life, which can be ruined by the intractable and miserable pruritus. The condition may recur, but recurrences are usually milder than the initial bout.
Mucous Membrane Pemphigoid (Cicatricial Pemphigoid)
Clinical Presentation
Mucous membrane pemphigoid is much less common than bullous pemphigoid, but this form of pemphigoid is much more likely to affect the mucous membrane of the genitalia and often more difficult to diagnose as many patients exhibit no intact blisters on extramucosal skin. The age of onset of mucous membrane pemphigoid is middle to old age and is more common in women in a ratio of 1.5:1.
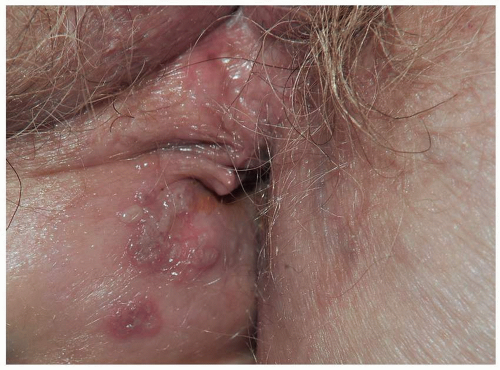
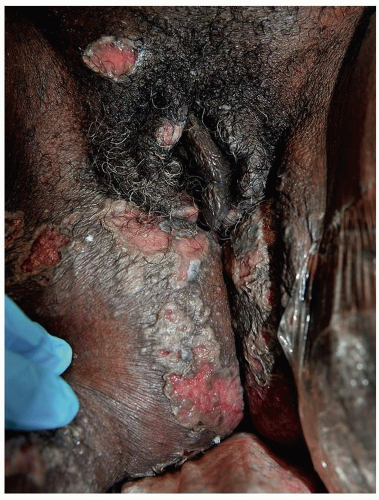
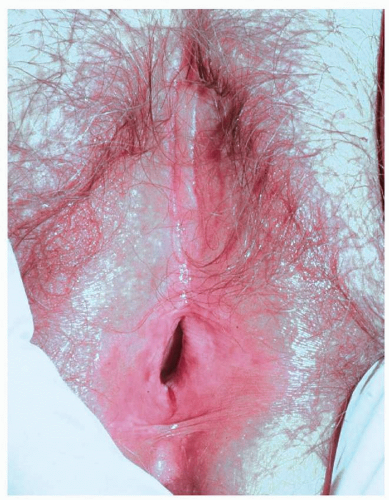
Mucous membrane pemphigoid primarily affects the mucous membranes with resulting scarring. It affects keratinized skin only in about 30% of cases. Because the diagnosis is difficult in the absence of intact blisters on hair-bearing skin, it may be delayed or sometimes missed. Mucous membrane pemphigoid usually starts with irritation, mild blisters, and erosions of the mouth, the eye, and the genitalia. Genital involvement is seen in 50% of cases of mucous membrane pemphigoid. Men report penile lesions, dysuria, and difficulty retracting the foreskin. Women report pain, pruritus, and dysuria. Morphologically, genital mucous membrane pemphigoid is characterized by painful erosions and scarring (Figs. 10.26, 10.27, 10.28 and 10.29). The preceding blisters are often short-lived, and the underlying blistering nature of the disease is missed. These mucous membrane erosions may progress rapidly to produce scarring with considerable morbidity. The initial, nonspecific, enlarging erosions produce scarring in more developed disease. Men may develop meatal stenosis and phimosis, and women may experience urethral stenosis, fusion of the labia, clitoral burial, and introitus stenosis.
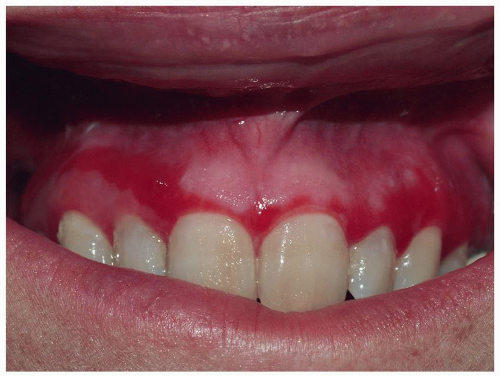 FIG. 10.27. Desquamative gingivitis indistinguishable from that of pemphigus vulgaris and, some cases, of erosive lichen planus is usually seen in patients with mucous membrane pemphigoid. |
Painful erosion in the mouth is usual. Erythema and erosions of the gingiva may lead to scarring and retraction with resulting secondary dental disease. Nasal mucosa, larynx, and pharynx may also be affected, and
stridor or dysphagia may result. The eye, despite feeling dry and gritty, may appear normal initially, but an ophthalmologic examination detects early abnormalities: reduced lacrimation or adhesions of the bulbar to the palpebral conjunctivae. Later, more extensive scarring may cause severe adhesions, entropion, and corneal scarring with blindness.
stridor or dysphagia may result. The eye, despite feeling dry and gritty, may appear normal initially, but an ophthalmologic examination detects early abnormalities: reduced lacrimation or adhesions of the bulbar to the palpebral conjunctivae. Later, more extensive scarring may cause severe adhesions, entropion, and corneal scarring with blindness.
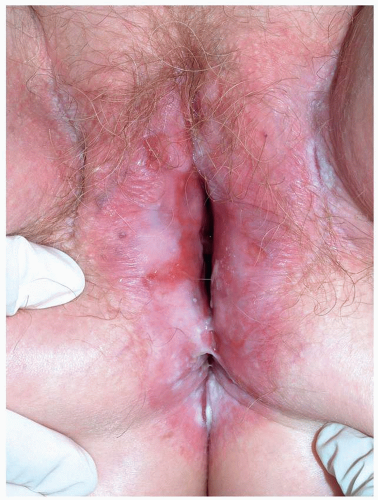 FIG. 10.28. Remarkable scarring with loss of vulvar architecture in a setting of erosions is characteristic of mucous membrane pemphigoid as well as lichen planus and pemphigus vulgaris,
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|