The concept of vaginitis is widely accepted. Most physicians assume that vaginitis represents an infection, with nearly all vaginal complaints diagnosed as Candidiasis, bacterial vaginosis, or trichomonas. However, like the mouth, the vagina is covered with squamous epithelium, and therefore affected by various dermatoses. Some dermatoses prominently affect mucous membranes, such as lichen planus, pemphigus vulgaris, cicatricial pemphigoid, and blistering forms of erythema multiforme. In addition, some dermatoses affect only the vagina, including desquamative inflammatory vaginitis and atrophic vaginitis. The diagnosis and management of these diseases are hampered by the difficulty of visualizing the vagina, and the lack of medications other than antimicrobials available for use in the vagina.
Vaginitis is a common symptom, generally characterized by introital itching or burning and vaginal discharge. Acute vaginitis most often is produced by infection, especially yeast. Candida albicans produces variable redness and, when severe, vaginal secretions containing white blood cells. Trichomonas vaginitis usually produces vaginal and cervical redness and increased white blood cells. Bacterial vaginitis is an uncommon condition with these same clinical findings. Infections are addressed elsewhere, and are not discussed in this article. However, inflammatory vaginitis is not always infectious in origin. In fact, chronic vaginitis usually is not infectious in origin, but rather produced by epithelial abnormalities. Many of these are normally managed by dermatologists when occurring on other skin surfaces. Unfortunately, no specialty claims noninfectious vaginal dermatoses, and the management of most of these are not included in training programs or discussed in most venues.
Isolated vaginal disease
Although rarely addressed, mucous membrane dermatoses, such as lichen planus, pemphigus vulgaris, and cicatricial pemphigoid, that affect the vagina as part of more generalized involvement are well known. Much less well understood is isolated vaginal inflammation.
Atrophic Vagina/Vaginitis
A common cause of introital burning, dryness and dyspareunia is estrogen deficiency. For years, this was an uncommon condition, because systemic estrogen replacement was supplied regularly to postmenopausal women without specific contraindications. When the results of the Women’s Health Initiative were released, systemic estrogen lost favor and the use of systemic estrogen declined sharply. Clinicians often forget to address topical estrogen replacement, so that in addition to hot flashes and other menopausal symptoms, women experience thinning of the vaginal epithelium and dryness, a condition called an atrophic vagina. Occasionally, inflammation occurs when these thin, fragile vaginal walls develop erosions, producing atrophic vaginitis.
Many women with an atrophic vagina experience no symptoms of estrogen deficiency, particularly if they are not sexually active. However, the prevalence of atrophic vagina in postmenopausal women is approximately 50%, making this a very significant and common problem. Patients report a sensation of uncomfortable dryness, and introital irritation occurs in some. Those who are sexually active often describe dyspareunia.
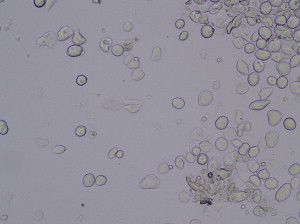
The clinical appearance of the postmenopausal vulva and vagina ranges from nearly normal to marked changes of atrophy to include diminution of the labia minora, with pallor, dryness, inelasticity, and mild narrowing of the introitus. The vagina is dry, pale, and lacking in the normal rugae, and the cervix flattens against the vaginal wall. A cotton-tipped applicator inserted to collect vaginal secretions seems completely dry in severely affected women, and a wet mount in these patients is nearly acellular. Most epithelial cells are parabasal cells, which are squamous epithelial cells that are immature and round, with a large nucleus compared with the cytoplasmic volume, unlike the large, flat, cuboidal, folded epithelial cells shed from a mature epithelium. Lactobacilli are absent ( Fig. 1 ). Vaginal candidiasis is very uncommon in estrogen-deficient women. Less affected patients exhibit more modest findings, less dryness, and more desquamated mature squamous cells.
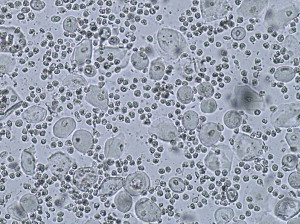
When the vaginal walls become irritated from the friction of sexual activity or a cystocele or rectocele protruding into the vagina, erosions sometimes develop, resulting in frank atrophic vaginitis, rather than simply an atrophic vagina. These women often experience burning and discomfort even without a trigger such as sexual activity or a gynecologic examination. Although women with an atrophic vagina report dryness rather than vaginal discharge, women with atrophic vaginitis often describe a yellow vaginal discharge. On physical examination, variable degrees of vaginal erythema and a purulent vaginal discharge are present. A wet mount shows an increase in white blood cells, so that fully developed atrophic vaginitis exhibits parabasal cells, sheets of neutrophils, no lactobacilli, and sometimes an increase in background bacteria ( Fig. 2 ).
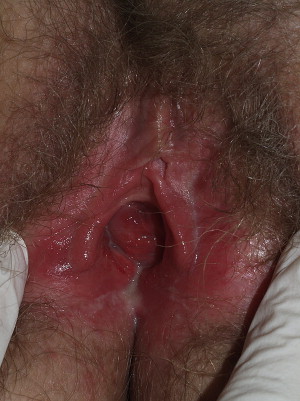
The underlying primary cause of an atrophic vagina and atrophic vaginitis is estrogen deficiency, which can occur after menopause, after delivery of a baby, with breastfeeding, and, to a variable degree, as a result of oral contraception. Obese women experience modest protection from peripheral conversion of estrogen in fat, and some clinicians believe that women who are regularly sexually active are less prone to experience an atrophic vagina.
The treatment of an atrophic vagina and atrophic vaginitis is usually quick and rewarding. Generally, a topical estrogen effects dramatic improvement within a week. Options for estrogen replacement include estradiol cream (Estrace) or equine conjugated estrogen (Premarin) cream, 1 g inserted into the vagina 3 nights a week. Alternatively, an estradiol tablet (Vagifem), 25 or 10 mcg, is inserted into the vagina 3 nights a week. Very popular and effective is an estrogen-impregnated ring (EstRing) that is inserted into the vagina and gradually releases estradiol over 3 months. The creams are messy and, sometimes irritating, with conjugated equine estrogen sometimes more irritating than estradiol. Insertion of the rather large estrogen ring into a dry, inelastic vagina can be uncomfortable, and is more comfortable after several weeks of estrogen cream or tablets.
An estrogen-deficient vagina is somewhat protected from Candidiasis, because yeast usually only produces infection in the presence of lactobacilli. However, the addition of a topical estrogen increases the risk of yeast, particularly during the first month. Patients can be advised to call if they experience any sudden itching, or can be prescribed a 150- to 200-mg fluconazole tablet orally once a week the first month to prevent this occasional occurrence.
Occasionally patients cannot tolerate any of the topical estrogen preparations because of burning or irritation. These women can be treated initially with systemic estrogen, either in a patch or an oral formulation. Often after a few weeks, the now well-estrogenized vagina tolerates the topical preparations. In addition, estrogen can be compounded into an ointment base, which is less irritating.
Many women, because of the results of the Women’s Health Initiative, are afraid of estrogen, even when prescribed topically. Other women have a personal or family history of breast cancer. Generally, these women can be reassured of the safety of topical estrogen, because the systemic absorption is low. The cream preparations have been associated with endometrial hyperplasia and breast tenderness in some patients, although lower-dose preparations are less likely to produce these side effects. Most oncologists permit topical estrogen in women with breast cancer. However, neutralization of the estrogen antagonists such as tamoxifen has been reported.
The occasional woman with an atrophic vagina who cannot tolerate or is not permitted to use topical estrogen sometimes experiences some improvement with a vaginal moisturizer. A common choice is polycarbophil-based vaginal moisturizer Replens. Although unpopular with many gynecologists, petroleum jelly is comforting to many women, and can be inserted with a finger and applied sparingly to the introitus. Women with the inflammation of atrophic vaginitis may benefit from the addition of a hydrocortisone acetate, 25 mg, rectal suppository inserted into the vagina, or a moderately potent corticosteroid ointment such as desonide ointment, 0.05%. Most recently, hyaluronic acid sodium salt, 5 mg, has been shown to be effective compared with topical estrogen.
The duration of therapy is variable. Usually, postmenopausal, sexually active women benefit from ongoing estrogen replacementat at a dosing of once or twice a week. Women who are not sexually active and who experienced the sudden onset of symptoms are sometimes able to discontinue their topical estrogen replacement and remain comfortable. Patients should be advised, if their estrogen is discontinued, that recurrence of symptoms is common. Sometimes, women who are atrophic as a result of hormonal contraception, especially injectable medroxyprogesterone (DepoProvera), benefit long-term from a change of contraception.
Desquamative Inflammatory Vaginitis
First described in 1968, desquamative inflammatory vaginitis (DIV) is probably a common condition. DIV is defined by its clinical and wet mount characteristics, and is a diagnosis of exclusion. Although once thought to represent lichen planus of the vagina, differences in the presentation and course of DIV indicate that it is a distinct entity.
Occurring in both premenopausal and postmenopausal women, DIV typically presents with introital irritation, burning, and dyspareunia, accompanied by a clinically and microscopically purulent vaginal discharge. Women with DIV are more likely than matched controls to have had diagnoses of Candida vaginitis, bacterial vaginosis, and pelvic inflammatory disease, which could be relevant or a manifestation of previous misdiagnoses by clinicians unfamiliar with this condition. Hormone replacement use is also more common in these women.
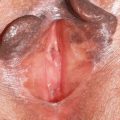
An examination of the vulva shows introital redness of variable degrees; some women experience extension of erythema to the labia minora, where puffiness is often seen ( Fig. 3 ). No specific vulvar lesions are present, nor is resorption of vulvar architecture seen, as occurs with lichen planus, lichen sclerosus, and the immunobullous diseases of cicatricial pemphigoid and pemphigus vulgaris. Clinicians who neglect the vaginal examination and wet mount can mistake the nonspecific vulvar findings for vestibulodynia (vulvar vestibulitis syndrome) or generalized vulvodynia, both painful syndromes sometimes associated with nonspecific erythema.
The vaginal walls are variably red. Because mucous membranes are normally somewhat red, especially in light-complexioned people, the degree of vaginal erythema can be difficult to judge. In more marked disease, the vagina can be covered with small red macules, reminiscent of the “strawberry cervix” classic for trichomonas. Erosions and vaginal synechiae are absent. An abnormal vaginal discharge is present, usually abundant and yellowish in color, with a pH greater than 5. Green and gray discoloration of secretions also has been described.
A wet mount shows an increase in white blood cells and parabasal cells (immature squamous epithelial cells shed from a vaginal epithelium that is rapidly proliferating because of inflammation). Lactobacilli are absent, as expected with a pH greater than 5. The wet mount is identical to that for atrophic vaginitis, and basically indicates only marked inflammation of vaginal walls (see Fig. 2 ). A culture should be performed, and this will either show no pathogens, or clinical findings will persist after correction of any incidental infections. Fairly often, a culture yields group B streptococcus ( Streptococcus agalactiae ), but symptoms do not improve with appropriate antibiotic therapy. Some clinicians believe that this organism plays a role in the pathogenesis of DIV, and cite an occasional woman with this finding on culture whose vaginitis resolves with an antibiotic. Others, however, contend that an inflammatory vaginitis with S agalactiae on culture, and signs and symptoms that resolve while on antibiotics, should be termed bacterial vaginitis . Most patients with inflammatory vaginitis whose culture yields group B streptococcus experience no change with antibiotic therapy, indicating the colonizing nature of this organism.
The diagnosis of DIV is made by the presence of a clinically purulent vaginal discharge; variable redness of the vaginal mucosa; a pH greater than 5; a wet mount showing an increase in leukocytes and parabasal cells and an absence of elongated rods presumed to represent lactobacilli; absence of estrogen deficiency; and cultures showing no causative organism. Exactly how many leukocytes or parabasal cells are required is not defined. Whether every one of these criteria must be present also is unknown.
The cause of DIV is unknown. Because many patients respond to topical clindamycin cream and because cultures often yield S agalactiae or Escherichia coli , some investigators have postulated infection as a factor. However, because elimination of these organisms with alternative active antibiotics does not result in improvement in most patients, because corticosteroids also are often beneficial, and because clindamycin has nonspecific anti-inflammatory as well as antimicrobial effects, many clinicians believe that DIV is a hypersensitivity or autoimmune phenomenon. Reports of DIV occurring in women with malabsorption and vitamin D deficiency have led to a theory of vitamin D deficiency as a pathogenic factor. This author measured vitamin D levels in 20 women with DIV; although several were slightly vitamin D deficient as defined by newer guidelines, none experienced improvement in DIV with vitamin D replacement and normalization of levels (Libby Edwards, MD, unpublished data, 2008).
Biopsies have been reported only in one small series, showing either a lichenoid or a nonspecific mixed inflammatory infiltrate. The inflammation was often mild compared that expected based on the degree of clinical redness and inflammation noted on wet mount. Direct immunofluorescence was nonspecific.
The variability of clinical findings, biopsy results, and response to therapy suggests that DIV may not be one disease, but rather may represent several, possibly related inflammatory processes. Some patients exhibit white blood cells on wet mount, but no parabasal cells, or no lactobacilli, or only a slight increase in white blood cells. Currently, no specific terminology exists to describe vaginal inflammation that does not meet all of the criteria of DIV. Vaginal inflammation in the absence of infection and specific lesions is an unexplored but common and important occurrence, and a fertile area for observational studies. In the meantime, management of all of these women is as reported for DIV.
The treatment of DIV consists of either clindamycin cream or corticosteroids. Clindamycin cream, 2%, should be inserted into the vaginal nightly, presumably for its anti-inflammatory effects rather than antimicrobial actions. Patients are treated for 2 to 4 weeks, and then reevaluated. Some women can discontinue their medication and remain clear for extended periods, whereas others require intermittent dosing or adjustment of dosing to the least frequent insertion that is effective.
Alternatively, topical corticosteroids can be used. Hydrocortisone acetate, 25 mg, rectal suppositories are inserted per vagina at bedtime. If this does not effect improvement, options include the compounding of 500-mg hydrocortisone suppositories, insertion of an ultrapotent topical corticosteroid such as clobetasol, or the use of combination clindamycin and a topical corticosteroid. Anecdotally, this author has found neither systemic corticosteroids nor oral antibiotics of any kind, including clindamycin, to be useful for DIV.
Many clinicians prescribe weekly fluconazole to patients receiving vaginal antibiotics or corticosteroids to prevent secondary Candidiasis. Those patients who cannot take fluconazole because of medication interactions can insert an azole suppository once or twice a week to minimize the risk of yeast.
Occasionally DIV does not respond to these therapies. These patients should be reevaluated for estrogen deficiency; cervicitis; infection to include trichomonas; any causes of erosion, including bullous or erosive mucous membrane dermatoses, complications of surgical sling placement with secondary erosion; or granulation tissue from previous surgeries. Clearly, further studies are needed to elucidate the cause and alternative therapies.
Vestibulodynia/Vulvar Vestibulitis
Vestibulodynia (previously known as vulvar vestibulitis) is a pain syndrome localized to the introitus and often mistaken for vaginitis. Currently believed to result primarily from pelvic floor dysfunction, neuropathy, and resulting anxiety and depression that worsen symptoms, vestibulodynia is not characterized by a vaginal discharge or any objective abnormalities, including inflammation, of the vagina.
The vagina as one site of multimucosal erosive dermatoses
Erosive Lichen Planus
Erosive vulvovaginal lichen planus is a common disease that usually occurs in a setting of erosions of other mucous membranes. The oral mucosa, vulva, and vagina are classically affected. Vaginal lichen planus can occur as an isolated condition, but this is extraordinarily rare. Most women with vaginal lichen planus exhibit vulvar disease and oral disease, but extragenital lichen planus of keratinizing epithelium is very uncommon in these women. Perianal and esophageal involvement are also common.
These women are usually postmenopausal, and describe pruritus, burning, soreness, and dyspareunia. The vagina exhibits patchy erosions, often within diffuse redness. Vaginal secretions are often grossly and microscopically purulent, with a wet mount showing, in addition to white blood cells, parabasal cells shed from the base of erosions and a paucity of lactobacilli. More-advanced disease is characterized by narrowing of the vagina and synechiae of the anterior and posterior walls that eventuate in complete obliteration of the vaginal space so that intercourse and the insertion of a speculum are impossible ( Fig. 4 ).