Fig. 51.1
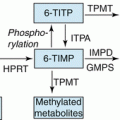
Pharmacology of cyclophosphamide. Cyclophosphamide is metabolised by the hepatic cytochrome P450 enzymes including CYP3A4 and CYP2B6 to form the active metabolite 4-hydroxy-cyclophosphamide. As the active hydroxylated cyclophosphamide is hydrophilic, it does not diffuse readily across the lipid bilayer into cells, and membrane transporters are suggested to aid cell entry in vivo. Once intracellular, it spontaneously degrades into the cytotoxic DNA alkylator phosphoramide mustard, producing a toxic by-product, acrolein. Acrolein is directly responsible for haemorrhagic cystitis and hepatic aminotransferase derangement in patients administered cyclophosphamide. In cells with high concentrations of aldehyde dehydrogenase (such as intestinal mucosa and hepatocytes), carboxy-cyclophosphamide is produced from 4-hydroxy-cyclophosphamide (through the tautomer aldophosphamide), with no phosphoramide mustard or acrolein being produced, hence sparing these tissues from the majority of the toxic effects from cyclophosphamide
51.3 The Use of Cyclophosphamide in AIBD
51.3.1 Pemphigus Vulgaris/Pemphigus Foliaceus
Cyclophosphamide has been examined in a number of randomised control trials for the treatment of pemphigus vulgaris (PV) and pemphigus foliaceus (PF). A study of 28 patients was conducted comparing pulsed IV dexamethasone-cyclophosphamide plus daily oral cyclophosphamide versus pulsed IV cyclophosphamide and daily oral prednisolone [3]. This study demonstrated earlier remission in the pulsed IV cyclophosphamide/oral prednisolone group (mean 7.3 weeks vs 3.2 weeks p = 0.02); however no difference was demonstrated in overall rates of remission or relapse [3].
When comparing cyclophosphamide against other immunosuppressive medications, no significant difference was seen in any clinical outcomes when comparing cyclophosphamide and mycophenolate [4]. No difference in time to disease control or relapse was found when cyclophosphamide was compared with cyclosporine in a small study of 18 participants [5].
With regard to the steroid-sparing role of cyclophosphamide, Chams-Davatchi conducted a landmark multiarm RCT into the steroid-sparing role of adjuvants, comparing prednisolone alone to three adjuvant agents: mycophenolate, azathioprine and pulsed cyclophosphamide [6]. The study demonstrated a significant decrease in the total dose of glucocorticoids when prednisolone was combined with a cytotoxic agent (p = 0.047) using analysis of variance. The average cumulative prednisolone doses over 12 months were prednisolone alone 11,631 mg, azathioprine 7,712 mg, mycophenolate 9,798 mg and cyclophosphamide 8,286 mg. No differences were detected in clinical endpoints, including time to partial response, complete response and treatment failure [6].
A recent meta-analysis conducted by Martin et al. found a superior steroid-sparing role for cyclophosphamide compared to azathioprine (−564.00 mg prednisone 95 % CI −1,048.54, −79.46, p = 0.023) [7]. However the statistical analysis conducted by the authors, which accounted for comparison of multiple groups, found no significant difference (p = 0.971). There was weak evidence of a benefit for azathioprine over cyclophosphamide in disease control RR (1.80, 95 % CI 0.89, 3.64, p = 0.10) [7]; however this is based on a study with small patient numbers [5]. The evidence comparing azathioprine to cyclophosphamide is inconclusive [7].
Martin’s systematic review identified three studies which included comparisons of cyclophosphamide against glucocorticoids, azathioprine, cyclosporine and mycophenolate [7]. There was a significant steroid-sparing effect seen when cyclophosphamide was combined with steroids (MWD −3,355 mg; 95 % CI −6,144 to −566, p = 0.018) [8]. The steroid-sparing effect of cyclophosphamide was found to be greater than that of mycophenolate mofetil and lesser than that of azathioprine [8]. It must be noted however that reduction in ‘cumulative steroid dose’ is a surrogate outcome.
51.3.2 Bullous Pemphigoid
Published experience with cyclophosphamide in the treatment of bullous pemphigoid (BP) is very limited. Individual case reports have noted the benefits of oral or IV cyclophosphamide combined with pulsed dexamethasone in cases resistant to other forms of therapy [9]. However, 100 mg of daily cyclophosphamide has also been noted, in a small series of ten patients, to give no steroid-sparing effect and an unacceptably high drug-related mortality and morbidity [9]. Most recent guidelines [9] do not recommend the use of cyclophosphamide in BP due to these safety concerns, and it should be considered only if other treatments have failed or are contraindicated [9].
51.3.3 Mucous Membrane Pemphigoid
Cyclophosphamide is commonly used in mucous membrane pemphigoid (MMP) as a steroid-sparing agent in individuals with severe or rapidly progressing disease [10, 11]. Dosages range from 1 to 2.5 mg/kg/day in oral form or can be given in monthly IV pulses of 0.5–1 g/m2 of body surface area when rapid control is desired [11]. A recent Cochrane review commented that if appropriately used and monitored, cyclophosphamide may be safer than long-term corticosteroid treatment in MMP [10]. Recent evidence suggests that cyclophosphamide may be more effective than dapsone in MMP although this is yet to be confirmed in large randomised trials [10, 11].
51.3.4 Pemphigoid Gestationis
Whilst cyclophosphamide is a known teratogenic drug, cyclophosphamide is used in the postpartum period for pemphigoid gestationis (PG). A recent review by Intong and Murrell notes a case report of an individual with sever persistent postpartum PG along with anti-phospholipid syndrome who had inappropriate response to oral corticosteroids [12]. IV cyclophosphamide 0.75 mg/m2 was commenced at 9 months postpartum, with two doses 4 weeks apart, and one additional dose 5 months later resulting in complete resolution 18 months post partum [12].
51.3.5 Paraneoplastic Pemphigus
Cyclophosphamide is an effective treatment modality in paraneoplastic pemphigus (PNP) when used in combination with systemic steroids. Treatment modalities include both pulsed IV and oral administration with either pulsed or daily corticosteroids [13–15]. Becker reports a case of PNP in the setting of Waldenstrom’s macroglobulinaemia treated with dexamethasone (100 mg IV at three weekly intervals) and cyclophosphamide (500 mg IV day 1–3 every 3 weeks) with complete response of desquamation and erosions as well as oral and genital lesions between 4 and 6 weeks [14]. Herzberg presents two cases of non-Hodgkin’s lymphoma (one follicular NHL and one large cell NHL) who developed PNP with buccal ulceration, haemorrhagic crusted lips, conjunctivitis and papulosquamous cutaneous eruptions with sparse bullae [13]. Both were successfully treated with 100 mg prednisone and 150 mg oral cyclophosphamide daily [13]. Nousari et al. have also reported the effectiveness and safety of ablative intravenous cyclophosphamide (200 mg/kg daily over 4 days) in a patient with chronic lymphocytic leukaemia-associated PNP [15]. A summary table of the level of evidence for the use of cyclophosphamide in AIBD is presented in Table 51.1.
Table 51.1
Evidence base for cyclophosphamide use in AIBD
Cyclophosphamide | PV/PF | BP | MMP/EBA | PG | PNP |
|---|---|---|---|---|---|
Monotherapy/adjuvant | Adjuvant | Adjuvant | Monotherapy/adjuvant | Adjuvant | Adjuvant |
Oral or IV dosing
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|