Targeted anticancer agent
Cutaneous adverse effects
Treatment options
EGFR inhibitors
– Papulopustular eruption
– Xerosis
– Nail changes (paronychia, pyogenic granulomas)
– Hair changes ( trichomegaly, hirsutism)
– TCS, oral TCNs
– Emollients
– TCS or topical antibiotic, silver nitrate
– Regular trimming, laser hair reduction
BRAF and MEK inhibitors
– Rash
– Photosensitivity
– Xerosis
– Squamoproliferative lesions
– Melanocytic nevi and melanoma
– TCS, antihistamines
– Photoprotection
– Sensitive skin care, emollients
– Routine skin examinations, surgical excision, oral retinoids
– Routine skin examinations, photoprotection
mTOR inhibitors
– Edema
– Stomatitis
– Delayed wound healing
– Compression garments
– TCS, topical analgesics
– Dressing
Tyrosine kinase inhibitors
– Hypo > hyperpigmentation
– Rash
– Self-resolution upon discontinuation
– TCS, antihistamines
Antiangiogenesis agents
– Mucosal bleeding
– Stomatitis
– Thromboembolism
– Delayed wound healing
– Ulcerations in striae
– TCS, topical analgesics
– Dressing
Special Implications in Children
Key Points
Few targeted anticancer agents have been approved in children.
Side effects from these agents in children appear to mirror their adult counterparts.
Malignant cells often hijack key pathways that also control normal development; thus targeted anticancer therapies may have unique adverse effect profiles and long-term implications in developing children.
While more than 60 agents have been approved in adults, very few have been approved for use in children. The mTOR inhibitors (e.g., everolimus) used in tuberous sclerosis complex for subependymal giant cell astrocytoma (SEGA) and c-KIT inhibitors (e.g., imatinib) used for Ph+ chronic myeloid leukemia (CML)/acute lymphoblastic leukemia (ALL) have been recently approved for pediatric use [2]. Currently, the most frequently studied targeted molecules in pediatric oncology trials are inhibitors of EGFR, BRAF, MEK, mTOR, BCR-ABL/KIT, and multikinase and antiangiogenesis agents. Multiple clinical trials with other combinations of these targeted agents are also ongoing in pediatric patients.
While data on dAEs in children is limited, their ubiquitous use in adults is helpful in predicting and developing management strategies in children. In fact, a thorough review of the literature and available clinical trial data on dAEs from targeted therapies in pediatric patients found that to date the side effects in children appear to mirror their adult counterparts [3]. The authors further found that rash was the most common AE encountered during treatment (19%), usually appears in a dose-dependent fashion, tends to vary by tumor type, and may be dose limiting [3]. Like in adults, patients on EGFR inhibitors (EGFRi) appeared to be at highest risk. Interestingly, the incidence and severity of the typical acneiform rash seen with this family of medication have been correlated with a better progression-free survival and overall survival in adults [4]. This has yet to be studied in children.
Importantly, malignant cells often hijack key pathways and the same molecular networks that control normal development. Thus, targeted anticancer therapeutics may have unique adverse effect profiles and long-term implications in developing children [2]. For example, an unanticipated observation in children treated with tyrosine kinase inhibitors (TKIs) for BCR-ABL1+ leukemias is that of growth suppression or growth failure. This is hypothesized to be due to concomitant inhibition of platelet-derived growth factor receptor (PDGFR) in chondrocytes, thus impairing proliferation and linear bone growth [5–7]. Spermatogenesis is also dependent upon PDGFR and c-KIT, and may be affected by exposure to this family of drugs; the long-term effects on fertility remain to be seen [8, 9]. The developing immune system and the potential for immunosuppressive effects of targeted agents should also be considered. Everolimus is associated with an increased risk of infection, and although the degree of immunosuppression is generally manageable, the extent to which immunosuppression persists and the potential for secondary malignancy may ultimately limit its chronic use [10].
EGFR Inhibitors
Key Points
EGFRi are associated with specific and predictable cutaneous toxicity in 50–90% of cases.
The most common dAE consists of a papulopustular eruption in a seborrheic distribution. Xerosis, hair changes, mucositis, and paronychia are other commonly reported cutaneous side effects.
Management is dependent upon severity of involvement, and usually enables continuation of therapy.
EGFRi were among the first targeted therapies and are used in the treatment of several malignancies in adults, including non-small-cell lung, colorectal, head and neck, and breast cancers [11]. EGFRi include monoclonal antibodies that target the extracellular ligand-binding domain of EGFR (cetuximab, panitumumab), small-molecule TKIs that target EGFR intracellularly (erlotinib and gefitinib), dual-kinase inhibitors of EGFR and human EGFR-2 (HER 2) (lapatinib), inhibitors of erbB (canertinib, afatinib), and other less specific multikinase inhibitors such as vandetanib [12]. Given the distribution of EGFR in the epidermis and pilosebaceous unit, it is not surprising that modulation of this receptor evokes skin and hair adverse effects. Interestingly, EGFR has also been shown to play a putative role in restraining interleukin-1 (IL-1)-dependent inflammatory reactions at the hair follicle level, shedding light on the follicular and papulopustular eruptions seen in conjunction with EGFR blockade [13].
Cutaneous AEs from EGFRi range in incidence from 50 to 90%, and include a spectrum of predictable and specific toxicities. Among these, papulopustular eruptions, xerosis, hair changes, mucositis, and paronychia are the most commonly reported. Data from pediatric patients treated with EGFR inhibitors (erlotinib, vandetanib) were reviewed by Belum et al. and rash was noted in 42/57 (73.7%) patients [3]. More specifically, an “acneiform rash ” was noted with vandetanib (3/15) and erlotinib (3/13) [14, 15].
The most common eruption reported in the adult literature (over 80% of patients on cetuximab) consists of follicular papules that evolve into pustules (Fig. 8.1) and that may coalesce into lakes of pus in a seborrheic distribution (scalp, face, and upper torso). This papulopustular eruption typically presents within 2 weeks of initiation, is dose dependent, may improve in some patients with continuation of therapy, and typically resolves after therapy is discontinued [16, 17]. While commonly described as acneiform, the eruption lacks comedones and cystic nodules and is inflammatory in nature. Significant pruritus is also reported in association with this dAE. Sun exposure may exacerbate this specific skin toxicity without necessarily inducing classic sunburn erythema [18]. Grade of severity is based on the body surface area involved and degree of limitation in performing activities of daily living [19].
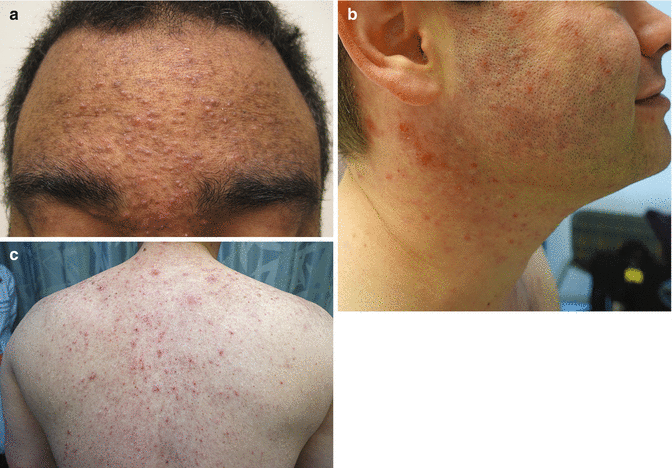

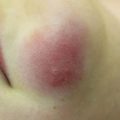
Fig. 8.1
(a–c) Papulopustular eruption secondary to EGFR inhibition
In general, management of toxicities is dependent upon severity of involvement. However, in adults treated with panitumumab, the use of a preventative regimen reduced the incidence of grade 2 or greater toxicity and resulted in less quality-of-life (QOL) impairment than reactive treatment alone [20]. This regimen consisted of an oral tetracycline (TCN) antibiotic , topical corticosteroids (TCS) , sun protection , and emollients. It has not been studied in children, where TCNs require additional considerations. When taking a reactive approach to grade 1 eruptions, low-potency TCS or topical antibiotics may be sufficient [21]. Given the inflammatory nature of this eruption, TCS are generally indicated. Grade 2 and 3 eruptions are best managed with systemic TCNs, which are considered first-line agents given their anti-inflammatory properties, in addition to TCS [22]. All patients should be counseled on sun protection and emollients. Calcineurin inhibitors, gel-based topical antibiotics, and benzoyl peroxide are avoided due to irritant potential and inability of most patients to tolerate them, supporting the finding that this is not simply drug-induced acne vulgaris.
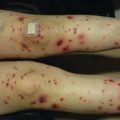
Xerosis is also reported in 1/3 of patients receiving EGFRi , has a significant impact on QOL, and commonly leads to asteatotic dermatitis (Fig. 8.2) [23]. It is characterized by dry and scaly skin reminiscent of atopic dermatitis, and is thought to be due to loss of the water-retaining function of the epidermis, developing as a result of disturbed keratinization and sebaceous gland function [24]. In addition, effects on the innate immune system lead to reduced defense and overgrowth of Staphylococcus aureus [25]. As in atopic dermatitis, gentle skin care, emollients, dilute bleach baths, and antihistamines may be of benefit [26].
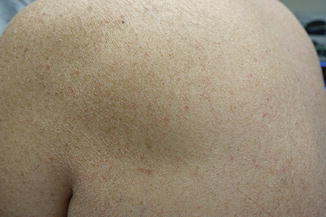

Fig. 8.2
Severe xerosis with asteatotic changes on EGFRi
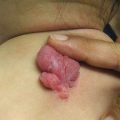
Nail and/or nail fold toxicity occurs in approximately 17% of patients, with the first digit being most commonly affected [27]. Nail changes usually appear after 2 months of treatment and can manifest as onycholysis, nail fragility with brittleness, and paronychia, or pyogenic granuloma-like lesions which may first occur after many months of therapy (Fig. 8.3). Culture is recommended in cases of inflammatory paronychia to rule out secondary infection, given disrupted epidermal defense. In addition to antimicrobial soaks (including dilute bleach or dilute vinegar, with explicit reminders to never combine these) and topical antimicrobials depending on the suspected or cultured organism, warm compresses, TCS, and systemic TCNs can be used for management [26]. Silver nitrate-based chemical cautery is helpful when granulation tissue is evident.
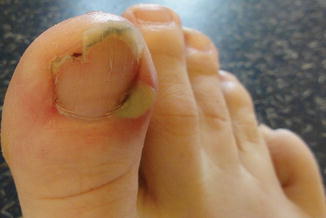

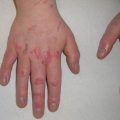
Fig. 8.3
Nail changes associated with EGFR inhibition include paronychia
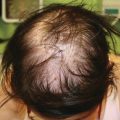
Changes in hair texture and growth pattern can be seen after 2–3 months of treatment and can be quite significant, particularly if patients have chemotherapy-induced alopecia immediately prior to or at the time of initiating EGFRi . Hair typically grows more slowly, and becomes more fine, brittle, and kinky [28]. Trichomegaly of the eyelashes is also characteristic (Fig. 8.4), and has been reported in 17% of pediatric patients [3, 29]. Management of this dAE is critical, as lashes may curl inward and lead to corneal scarring if they are not trimmed. Mild diffuse alopecia, in a pattern similar to androgenic hair loss, and poliosis have also been reported [30].
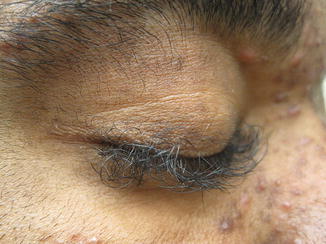

Fig. 8.4
Trichomegaly associated with EGFR inhibition
Mucositis may develop, manifesting with aphthae, xerostomia, or geographic tongue [31]. Genital involvement is less common. Other reactions to EGFRi include anaphylaxis (1.2–3.5% of patients taking cetuximab), enhancement of radiation dermatitis, ocular complications from dryness or trichomegaly as above, vasculitis, necrolytic migratory erythema, and transient acantholytic dermatosis [26, 32–34].
BRAF and MEK Inhibitors
Key Points
Activating BRAF mutations are found in over 50% of melanomas, but also in other cancers such as hairy cell leukemia and Langerhans cell histiocytosis.
Dermatologic adverse effects secondary to BRAF inhibitors affect up to 95% of patients, manifesting as nonspecific/morbilliform rash , evolving over time to become more folliculocentric. Photosensitivity, alopecia, pruritus, superficially desquamative hand-foot syndrome, and proliferative lesions of the epidermis have also been reported.
Dermatologic adverse effects secondary to MEK inhibitors are similar to EGFRi and include papulopustular eruptions favoring the scalp, face, chest, and back as well as xerosis, pruritus, and photosensitivity.
The combination of BRAF and MEK inhibitors improves survival in metastatic melanoma and induces fewer cutaneous side effects.
The MAP kinase, or RAS-RAF-MEK-ERK, pathway is a sequential enzyme cascade that leads to a multitude of effects on cellular proliferation, differentiation, migration, and apoptosis [35]. BRAF is a serine-threonine protein kinase in this signaling cascade. Activating BRAF mutations are found in more than 50% of malignant melanomas and the majority of these mutations affect a specific amino acid residue (V600) in BRAF. These mutations also occur at lower frequencies in other types of proliferative lesions, including benign nevi, and a subset of head and neck squamous cell, colon, lung, and thyroid cancers as well as hairy cell leukemia and malignant histiocytoses [36]. Vemurafenib and dabrafenib are selective inhibitors that target these kinases and are FDA approved for advanced melanoma (2011, 2013, respectively). They have demonstrated favorable clinical responses in melanoma patients carrying the mutation BRAF V600E and to date are most commonly used in combination with MEK inhibitors (MEKi) in an attempt to overcome BRAF resistance. BRAF inhibitors (BRAFi) present unique and predictable dAEs that are due, at least in part, to molecular events linked to the mechanism of action of these drugs, paradoxical activation of MAPK. The dAEs appear proportional to dose and duration of drug exposure [37].
As with EGFRi , dAEs are the most common adverse effects in patients treated with BRAFi, affecting up to 95% of patients; a nonspecific rash occurs in up to 75% of patients [38]. Patients often present with a mild, transient morbilliform eruption within the first few weeks of therapy that is asymptomatic or mildly pruritic and resolves with time or TCS. Failure to respond should raise suspicion for a delayed-type hypersensitivity reaction. The persistent “rashes” associated with BRAFi are better classified as folliculocentric or papulopustular, sometimes with a keratosis pilaris-like appearance, and occur primarily on the face (Fig. 8.5) and upper torso and arms (Fig. 8.6).
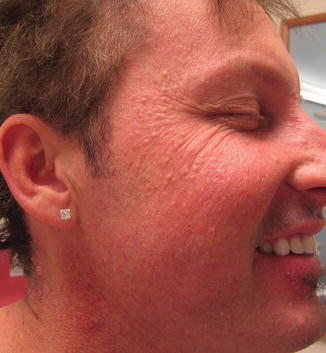
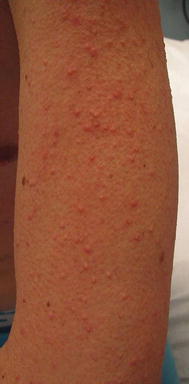

Fig. 8.5
Milia-like folliculocentric papules on the face secondary to BRAF inhibition

Fig. 8.6
Keratosis pilaris-like papules on the arm secondary to BRAF inhibition
Photosensitivity is common affecting 30–57% of patients (Fig. 8.7); this is UVA induced and patients are advised to wear sunscreen daily to avoid toxicity from incidental indoor exposure through windows [39]. Alopecia, pruritus, superficially desquamative hand-foot syndrome, and proliferative lesions of the epidermis have also been reported. Proliferative lesions classically develop within 8–12 weeks of treatment. These lesions represent a continuum and encompass benign papillomas, verrucae, verrucous keratoses, seborrheic keratoses, warty dyskeratomas, palmar/plantar hyperkeratosis, actinic keratoses, and keratinizing skin tumors such as keratoacanthomas and cutaneous squamous cell carcinomas (SCC) [40]. These are treated with skin-directed therapies, reserving surgery for invasive SCC. An additional and occasionally challenging effect of BRAFi is the occurrence of melanocytic changes. Darkening of existing nevi, regression of nevi, eruptive nevi (Fig. 8.8), and development of atypical melanocytic proliferations have all been described [40]. Second primary melanomas have also been reported [41]. Paradoxical MAP kinase pathway activation by wild-type BRAF or RAS mutant cells provides a plausible mechanism for SCC and, potentially, melanocytic tumor development as well [42].
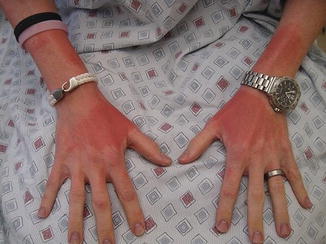
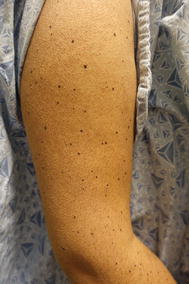

Fig. 8.7
Severe sunburn after minimal sun exposure while on BRAF inhibitor

Fig. 8.8
Eruptive dark nevi with a background of keratosis pilaris-like papules secondary to BRAF inhibition
Understanding this paradoxical MEK/MAPK activation in normal cells and tissues contributed to the hypothesis that MEKi may reduce cutaneous tumor formation induced by BRAFi [43]. MEK inhibitors, such as trametinib, target the MAPK pathway further downstream, and in studies combining a MEKi with a BRAFi in patients with metastatic melanoma, there was not only an extension of progression-free survival but also a reduced incidence of treatment-associated SCCs from 19 to 7% [44]. While patients treated with MEKi monotherapy suffer from an array of dAEs, patients treated with BRAF/MEK combination therapy have few skin side effects.
While often described as similar to EGFRi toxicities, dAEs seen with MEKi have some morphologic distinctions. Eruptions include an inflammatory papulopustular morphology favoring the scalp, face, chest, and back (52–93% of patients) as well as xerosis, and pruritus [45]. In many patients, the papulopustular eruption differs from that induced by EGFRi in that patients present with tiny, monomorphic follicular papules (Fig. 8.9) and eroded pustules that may extend to the lower trunk and extremities. Additional common AEs from MEKi that can confound the presentation include edema (with resultant erythema and dermatitis), fevers, and photosensitivity.
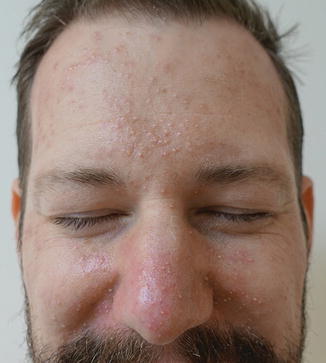

Fig. 8.9
Tiny monomorphic papules and pustules secondary to MEK inhibition
mTOR Inhibitors
Key Points
mTOR inhibitors are used increasingly in children, and currently approved for patients with SEGA in the setting of tuberous sclerosis complex.
Most common adverse effects involve the oral mucosa, manifesting as painful stomatitis and oral ulcers.
“Rash” is less commonly seen and described as nonspecific in nature.
The phosphatidylinositol 3-kinase (PI3K)/AKT signaling pathway is critical to cell growth and survival and has been shown to govern normal vascular development and angiogenesis [46]. Sirolimus, a mammalian target of rapamycin (mTOR) inhibitor, integrates signals from the PI3K/AKT pathway to coordinate proper cell growth and proliferation by regulating ribosomal biogenesis and protein synthesis [47]. Everolimus is similar to sirolimus in that it is an effective antiproliferative and immunosuppressive agent, developed to improve upon the pharmacokinetics of sirolimus [48]. Tuberous sclerosis and lymphangio leiomyomatosis are caused by inactivating mutations in the tuberous sclerosis complex tumor-suppressor proteins TSC1 and TSC2, leading to increased activation of mTOR [49]. Disorders that lead to inappropriate activation of the PI3K/AKT/mTOR pathway have also been shown to result in tissue overgrowth in association with vascular anomalies. Sirolimus is now approved in children with tuberous sclerosis complex, and increasingly studied in patients with vascular anomalies such as kaposiform hemangioendothelioma with Kasabach-Merritt phenomenon and lymphatic malformations [50]. As mTOR inhibitors (mTORi) have significant antitumor activity, they are increasingly being studied in pediatric cancers, including rhabdomyosarcoma, osteosarcoma, medulloblastoma, and neuroblastoma, alone and in combination with chemotherapy.
Dermatologic AEs seen in pediatric patients treated with mTORi usually consist of a macular or papular exanthema, or papulopustular eruption thought to be due to the inhibition of PI3K-AKT-mTOR signaling, which is one of the downstream effector pathways of the EGFR [51]. However, the most common AEs involve the oral mucosa, with painful stomatitis causing significant QOL impairment in adults and children. A phase 3 trial of everolimus in children with SEGA in the setting of tuberous sclerosis found that most AEs were grade 1 and 2, with mouth ulcers (32%) and stomatitis (31%) being the most frequently reported [52]. Rash was reported in 12% of patients, and one patient developed zoster during treatment. A trial studying the safety of everolimus in patients under 3 years of age found a similar AE profile, with the most common adverse effect being stomatitis (66.7%) [53]. Temsirolimus is a potent and highly specific inhibitor of mTOR, the ester form of sirolimus, and was the first mTORi approved by the US Food and Drug Administration for use in oncology in advanced renal cell carcinoma in the adult population. A study in pediatric patients with recurrent and refractory solid tumors showed similar AEs, including rash (32%) and mucositis (32%) [54]. Cutaneous AEs from PI3K isoform inhibitors are poorly described to date, but have been observed to include eczematous, psoriasiform, and pityriasiform eruptions.
Tyrosine Kinase Inhibitors
Key Points
TKIs that target BCR-ABL and c-kit such as imatinib have been approved in children with leukemias ( CML/ ALL).
Edema, mainly periorbital, is a distinct and specific side effect of imatinib.
A dose-dependent papular rash can also occur, and is generally follicular or finely keratotic in nature. Reversible pigmentary changes have also been reported in 33–41% of patients.
The development of BCR-ABL/KIT TKIs has greatly improved the outcome of patients with CML and gastrointestinal stromal tumors. Imatinib was the first molecule developed to inhibit the tyrosine kinase BCR-ABL; it also inhibits c-kit and PDGFRs. Because it acts on multiple targets, this class of drug is used and studied in other malignancies [55, 56]. Cutaneous AEs have been reported to occur in 7–88.9% of patients taking imatinib [56, 57]. Edema , mainly periorbital, is a distinct and specific side effect. It can also occur in the extremities and occasionally as central fluid retention. It has been speculated that inhibition of PDGFR leads to an increase in dermal interstitial fluid, as it has a central role in regulating interstitial fluid homeostasis [56]. A dose-dependent papular rash can also occur that is generally follicular or finely keratotic in nature. Reversible pigmentary changes manifesting as localized, patchy, or diffuse hypopigmentation and depigmentation have been reported in 33–41% of patients [58, 59]. These changes are caused by the inhibition of c-kit, which regulates melanocyte development, migration, and survival. Interestingly, hyperpigmentation has been reported in 3.6% of patients, usually occurring in hair, nails, and oral mucosa [57]. Dyspigmentation has been less commonly noted in pediatric patients (13%) [3].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree