Cutaneous Bacterial Infections
Francisco G. Bravo
The skin is the immediate interface between humans and their environment and serves as an effective barrier to a host of microorganisms that are present ambiently throughout the world. Some are normal cutaneous flora, whereas others are potential pathogens. This chapter summarizes the cutaneous bacterial infections that children may develop with emphasis on a synthesis of the clinical and histologic features.
IMPETIGO
Definition and Epidemiology
Impetigo is one of the most common bacterial infections in dermatology and general medicine. It is caused by Group A beta-hemolytic Streptococcus pyogenes, Staphylococcus aureus, or a combination of both.
Impetigo is the most common infection between the ages of 2 and 5 years, but can be seen at any age. It is highly transmissible.
Etiology
Impetigo has two clinical forms, represented by “honey-crusted,” nonbullous impetigo, caused by S. pyogenes and S. aureus, and a bullous form, caused exclusively by the specific phage types of S. aureus.
Group A streptococci are highly pathogenic in comparison with other groups of streptococci. They can be isolated from the skin up to 10 days before the disease manifests clinically, and they can be isolated from the oropharynx 14 to 20 days after appearing on the skin.1,2
Bullous impetigo is mostly produced by S. aureus group II, phage type 71, and also phage types 3A, 3C, and 55. Two toxins, with desmoglein 1 as their target, have been described. The exfoliative toxins produced by Staphylococcus can produce impetigo and staphylococcal scalded skin syndrome by cleaving a peptide bound in desmoglein 1, imitating the mechanisms seen in pemphigus foliaceus.3
Clinical Presentation
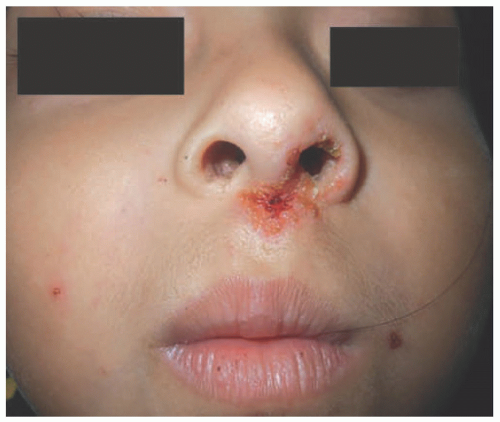
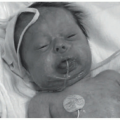
Nonbullous impetigo (crusted impetigo, impetigo contagiosa) is the most common form seen in children over 2 years of age, as well as in adults. It represents 70% of all impetigo cases. S. aureus, either alone or in combination with Group A beta-hemolytic streptococci, is responsible for 80% of cases.1 The infection may arise in normal skin or in already damaged skin, including atopic dermatitis, insect bites, scabies, or pediculosis. The lesions are round, with a very fragile central vesicle that breaks easily and becomes a honey crust (Figure 17-1). They measure 2 to 4 cm, grow centrifugally, and develop satellite lesions. They are preferably located on exposed areas, such as the face and extremities. The patient may develop regional lymphadenopathy and fever. The skin lesions can self-resolve in 2 to 3 weeks without therapy.
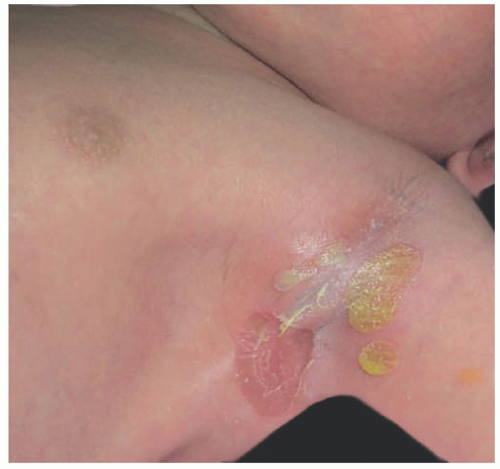
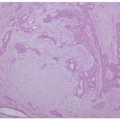
Bullous impetigo starts with smaller blisters that are initially tense, with clear contents. These later become flaccid and contain pus (Figure 17-2). The lesions are usually multiple, with satellites; they can reach up to 2 cm in diameter, and when the blister breaks, the center looks erythematous and the roof remnants give the lesion an
annular morphology. They may occasionally be polycyclic (Figure 17-3). The lesions are mostly located on intertriginous areas such as diaper zone, axillae and neck, but they can also be acral in location. Regional lymphadenopathy is rare.
annular morphology. They may occasionally be polycyclic (Figure 17-3). The lesions are mostly located on intertriginous areas such as diaper zone, axillae and neck, but they can also be acral in location. Regional lymphadenopathy is rare.
Histologic Findings
Nonbullous impetigo (which is only rarely biopsied) may initially show a few neutrophils at the subcorneal level and in the underlying epidermis. Crusted impetigo is represented by a histologic crust, containing a mixture of serum with parakeratotic and neutrophilic debris.
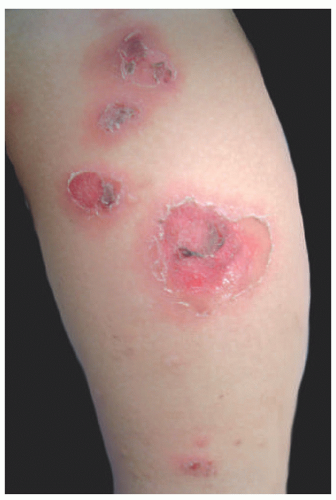 FIGURE 17-3. Bullous impetigo: remnants of the roof of the blisters are seen at the lesional edges, giving the lesions an annular appearance. Courtesy of Dr Rosa Ines Castro. |
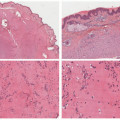
Classical bullous impetigo shows a subcorneal epidermal blister containing neutrophils (Figure 17-4).4 Spongiosis and focal acantholysis may also be seen (Figure 17-5).5 The underlying dermis contains variable inflammation, commonly featuring neutrophils, which may also invade the epidermis. Tissue Gram stains may show cocciform bacteria, representing either S. aureus or S. pyogenes.
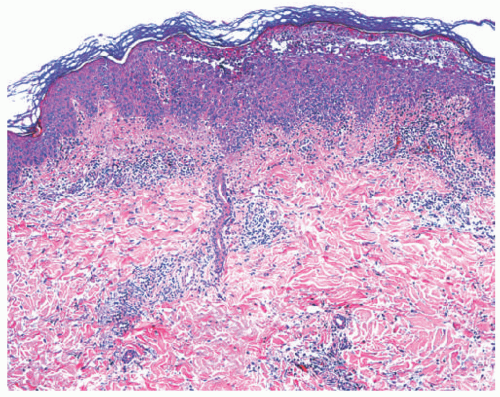 FIGURE 17-4. Bullous impetigo: subcorneal blister with acantholysis. Hematoxylin and eosin (H&E) stain. ×200. Courtesy of Dr Luis Requena. |
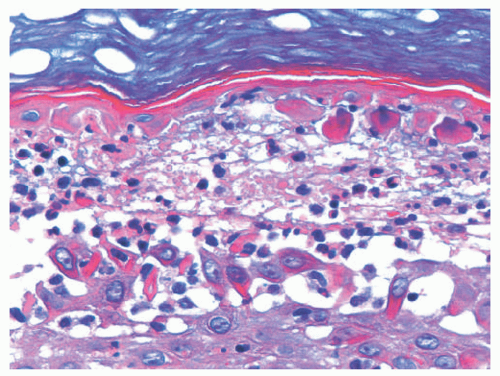 FIGURE 17-5. Bullous impetigo: high power of the case in Figure 17-3. H/E ×400. Courtesy of Dr Luis Requena. |
Differential Diagnosis
Commonly, in the first hours of its evolution, tinea corporis may be associated with a honey crust, and it can be confused with crusted nonbullous impetigo. In Herpes simplex infection, cutaneous vesicles are smaller and have a more grayish color than those of impetigo. Histologically, herpetic lesions show nuclear molding and multinucleated giant cells. Subcorneal blisters with neutrophils can be seen in acute generalized eruptive pustulosis, early psoriasis, candidiasis, tinea, and acropustulosis of infancy. The clinical context and periodic acid-Schiff (PAS) staining will help in establishing the correct diagnosis.
CAPSULE SUMMARY
IMPETIGO
Impetigo is caused by beta-hemolytic S. pyogenes and S. aureus. Nonbullous impetigo is most commonly associated with S. aureus and S. pyogenes. It is usually located on exposed areas such as face and extremities, and honey-crusted lesions predominate. Bullous impetigo is most commonly associated with S. aureus group II, phage type 71. On intertriginous areas, lesions are flaccid and vesicular. They later break, leaving annular lesions with a collarette. Histologic findings include either a subcorneal blister containing neutrophils or a crust over the epidermis with spongiosis.
Infections With Methicillin-Resistant S. Aureus
The first strain of S. aureus that was resistant to methicillin was described in 1961. In the general population, one in three people carries S. aureus in the nose, without any manifestations, whereas 2 out of 100 people harbor methicillin-resistant S. aureus (MRSA). MRSA was initially seen in hospitalized patients who required invasive interventions, such as placement of intravenous lines, bladder catheters, and mechanical ventilation. By the end of the 1990s, another variant of MRSA was described in the mid-western United States, which was associated with skin and soft tissue infection in persons in the general populace. It was named “community-acquired” MRSA (CA-MRSA).
MRSA bacteria are different from staphylococci that produce exfoliative toxins and those associated with bullous impetigo. They carry the gene that allows the production of the Panton-Valentine Leucocidin exotocin.1 In the community, MRSA is considered to be a rare cause of impetigo. CA-MRSA infections are more likely to present with the formation of furuncles and abscesses than with impetigo. MRSA is now the most common cause of purulent skin and soft tissue infections in many regions of the United States.6
ECTHYMA
Definition and Epidemiology
Ecthyma is a bacterial infection that is deeply seated in the skin and has a tendency to affect the lower dermis, producing ulceration with crusting. Ecthyma gangrenosum (EG) is the term used for a similar, ulcerated lesion caused by a hematogenous spread of infections with Pseudomonas aeruginosa or other causes of bacterial septicemia.
Ecthyma is commonly seen on the legs in children and elderly persons, and it is a frequent complication of papular urticaria. EG is more commonly encountered in immunocompromised patients with Gram-negative sepsis, most often but not exclusively caused by P. aeruginosa.
Etiology
Most ecthymas are produced by Group A streptococci, either as a primary infection or as a complication of preexisting conditions such as an insect bite or excoriation. Coagulase-positive staphylococci have also been isolated as coinfectants from the lesions of streptococcal ecthyma. EG is mostly seen in immunocompromised patients, particularly those with underlying malignancies. Once considered a clinical entity that was pathognomonic of P. aeruginosa infection, it is now known to be potentially associated with additional microorganisms, including other Pseudomonas species, Escherichia coli, Citrobacter, Klebsiella, Morganella, Candida, and Fusarium.7
Clinical Presentation
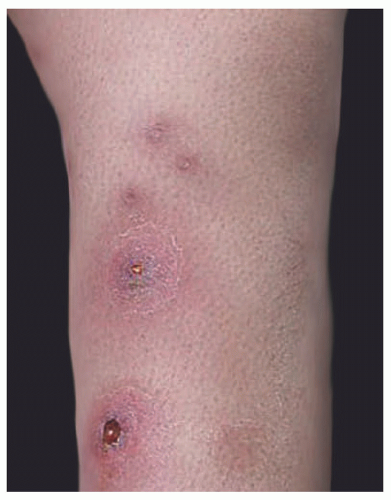
Classical ecthyma begins as a vesicle or a pustule, surrounded by a zone of erythema. As it enlarges, it will develop a central yellow-green crust, with a “punched-out” appearance (Figure 17-6).8 If the overlying scab is removed,
a saucer-shaped ulcer will be revealed with elevated borders and a grainy base. Typical locations are the shins and the dorsa of the feet. Lesions can be multiple. In addition to children, ecthyma also occurs in intravenous drug users, patients infected with HIV, and individuals with poor hygiene, malnutrition, or previous trauma.9
a saucer-shaped ulcer will be revealed with elevated borders and a grainy base. Typical locations are the shins and the dorsa of the feet. Lesions can be multiple. In addition to children, ecthyma also occurs in intravenous drug users, patients infected with HIV, and individuals with poor hygiene, malnutrition, or previous trauma.9
EG starts as a hemorrhagic pustule that evolves into a necrotic ulcer; rather than a crust, it is covered by a black scab and surrounded by a red halo. EG is predominantly located on the gluteal and perineal skin, but up to 30% of cases may be seen on the extremities or the face.8 Lesions are larger than those of classical ecthyma. EG has also been reported in other circumstances besides malignancy, and even in immunocompetent persons.8 Healthy infants may develop EG in the diaper area after receiving antibiotics, in conjunction with maceration of the skin.
Histologic Findings
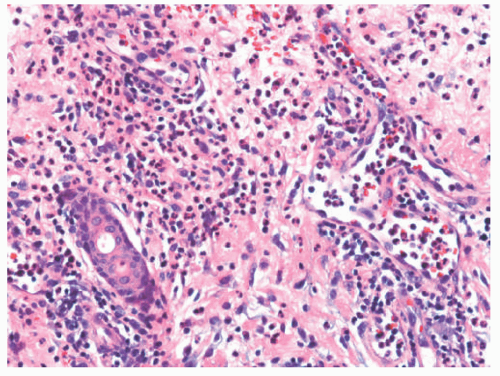
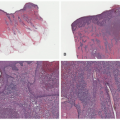
The histologic appearance of classical ecthyma shows ulceration, with superficial crusting and many neutrophils in the underlying dermis (Figures 17-7 and 17-8). Accumulations of cocci can be seen inside the crust.
In EG, necrosis is present in the epidermis and dermis, along with hemorrhage and a mixed inflammatory cell infiltrate around an area of infarction, caused by vascular thrombosis. Vasculitis can be seen at the edges of the ulcer. Sometimes, the inflammatory infiltrate is sparse. Gram-negative bacteria can be detected in the vessel walls.10
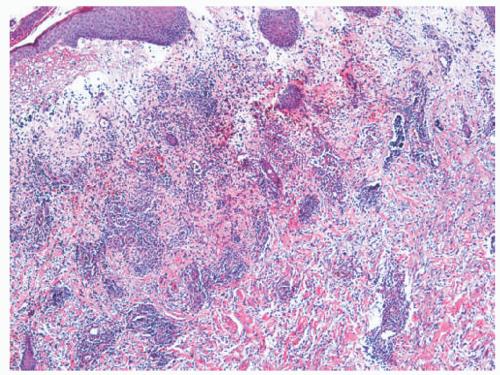 FIGURE 17-7. Ecthyma, histologic findings include edema in the papillary dermis and neutrophils in the mid-dermis. H/E ×100. Courtesy of Dr Luis Requena. |
Differential Diagnosis
Ecthyma can be confused clinically with excoriated papules of prurigo nodularis. Well-developed lesions can be mistaken for leishmaniasis. The histologic findings in EG can also be seen in cutaneous infections with Vibrio vulnificus.
CAPSULE SUMMARY
ECTHYMA
Classical ecthyma is streptococcal in nature, located on the pretibial areas with crusted and punched-out lesions. Affected patients are usually children. Biopsies show ulceration and neutrophilia, and bacteria can be seen in the crust.
EG is associated with sick, immunocompromised patients. P. aeruginosa is the predominant, but not the only, etiologic agent. The lesions are more necrotic than crusted. Histologically, necrosis is evident, and the inflammatory infiltrate is mixed and sometimes sparse. There may be evidence of vasculitis, with bacteria in the vessel walls.
BACTERIAL FOLLICULITIS
Definition and Epidemiology
Bacterial folliculitis is the infection of hair follicles and perifollicular areas by bacteria, presenting with multiple dome-shaped pustules that are centered on the follicular ostia. The most common causative organism is S. aureus.
Folliculitis in children most commonly affects the scalp, whereas in adults it has a predilection for the bearded portion of the face, the axillae, the gluteal area, and the extremities. Excessive sweating, a humid environment, poor hygiene, the use of occlusive preparations and dressings, and maceration of the skin are predisposing factors, as is continuity with draining abscesses. Repeated bathing in poorly chlorinated swimming pools, hot tubs, and whirlpools is associated with Pseudomonas folliculitis. Gram-negative bacterial folliculitis may be seen in patients who have had a prolonged treatment of acne with oral antibiotics.
Etiology
The most common causative agent for bacterial folliculitis is S. aureus, but occasionally coagulase-negative Staphylococcus species can cause the condition. “Hot tub folliculitis” is classically associated with infections by P. aeruginosa. Bacteria seen in posttreatment folliculitis in acne patients are usually Enterobacteriaceae and Proteus species.
Clinical Presentation
Bacterial folliculitis can be divided into superficial and deep forms. The superficial variant, known as impetigo of Bockhart, comprises tiny, fragile pustules, centered on the follicular ostia. In children, it usually affects the scalp. Hair shafts can be seen piercing the pustules. Preexisting conditions such as living in hot humid climates, obesity, the use of occlusive dressings, and diabetes mellitus all favor the appearance of folliculitis. Deep forms of folliculitis include furuncles—most commonly caused by S. aureus infection— and folliculitis barbae or sycosis barbae.8 Patients with hot tub folliculitis caused by P. aeruginosa develop lesions on the buttocks, hips, and axillae.11 Otitis and mastitis are possible, as well as systemic symptoms such as fever and malaise. Children and adolescents may have a singular presentation of folliculitis, with the development of red papules and nodules on the hands and feet.12,13
Histologic Findings
In the superficial form of bacterial folliculitis, a subcorneal neutrophilic pustule is seen that is centered on the follicular infundibulum and the surrounding dermis. Lymphocytes and macrophages may be present as well. The inflammatory infiltrate may involve the entirety of the follicle.
In the deeper variants of folliculitis such as the furuncle, one sees dermal abscesses that are centered on the mid or lower portions of the follicle; the follicular epithelium may be destroyed, but a remnant of hair shafts can be seen. The epidermis is only secondarily involved and is replaced by a crust, and the process may extend into the subcutis.
Pseudomonas folliculitis presents histologically with a combination of superficial and deep folliculitis. There is a predominance of neutrophils in the infiltrate with the destruction of follicular walls. Gram-negative folliculitis in acne patients is also superficial and deep.
Differential Diagnosis
Tinea on the face may have a follicular component that remains when pediatric patients are treated only with topical antimycotic therapy. The associated lesions may histologically show either suppurative or lymphocytic inflammation, sometimes with giant cells. An important clue is the presence of spores or hyphae in the follicular lumina. Pityrosporum folliculitis can cause a rupture of the follicles, and yeast forms are seen inside of them, sometimes spilling into the surrounding dermis. Eosinophilic pustular folliculitis of infancy occurs during the first 14 months of life, presenting as crops of papules and pustules on the scalp with possible extension to other sites. Many eosinophils are seen microscopically, and the infiltrate may be interfollicular rather than follicle centered.14
CAPSULE SUMMARY
FOLLICULITIS
Superficial folliculitis is most commonly associated with infection by S. aureus (impetigo of Bockhart). Clinically, it comprises tiny pustules on the scalp with hair shafts in the center of them. Histologically, one sees a neutrophilic infundibulitis and peri-infundibulitis. Chronic folliculitis is a deep process, either a follicle-centered abscess (furuncles caused by Staphylococcus) or a superficial and deep suppurative infection as in P. folliculitis.
ERYSIPELAS
Definition and Epidemiology
Etiology
The vast majority of erysipelas cases are caused by infection with Group A beta-hemolytic S. pyogenes. Group B S. pyogenes may also produce disease in newborns. Other potentially causative microbes include S. pyogenes of Group G, C, and D, as well as S. aureus, Streptococcus pneumoniae, Klebsiella pneumoniae, and Yersinia enterocolitica.
Clinical Presentation
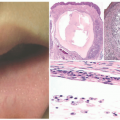
Currently, the most common location for erysipelas is the legs (Figure 17-9), with the face being next in frequency. This infection is commonly associated with a break in the skin, such as a fissure, an insect bite, or superficial dermatophytosis. In infants, the disease may disseminate from the umbilical stump to the skin of the abdomen or from a circumcision site to the genitoperineum. Predisposing conditions include venous or lymphatic obstruction, diabetes mellitus, and nephrotic syndrome. Erysipelas commonly debuts with the sudden onset of fever, chills, and malaise.
Clinical findings include a rapidly developing red plaque that is warm, tender, shiny, elevated, sharply demarcated, and confluent, occasionally with a “peau d’orange” appearance. Vesicles, bullae, and ecchymotic lesions may develop as the infection progresses. When erysipelas subsides, mild hyperpigmentation and fine scaling may be the residua. Complications include bacteremia, local abscess formation, thrombophlebitis, gangrene, and glomerulonephritis.
Histologic Findings
Histologically, erysipelas is characterized by marked subepidermal edema, and a diffuse, variably dense interstitial infiltrate of neutrophils in the underlying dermis, without abscess formation.10 The infiltrate is accentuated around blood vessels, and nuclear dust may be present. Capillary and lymphatic vessels may be dilated, and granulation tissue may be located beneath the zone of papillary edema as the disease subsides.
Differential Diagnosis
The principal histologic differential diagnoses for erysipelas are Sweet syndrome, which usually has a denser mid-dermal infiltrate of neutrophils, and leukocytoclastic vasculitis. Rheumatoid dermatitis-panniculitis and erythema marginatum may also be neutrophilic, but they have different clinical manifestations.
CAPSULE SUMMARY
ERYSIPELAS
Clinically, presentation is an acute onset of fever, chills, and malaise and the subsequent appearance of a red, shiny, warm, tender plaque on a leg or the face. Histologically, one sees edema of the papillary dermis and a diffuse interstitial dermal infiltrate of neutrophils.
ERYSIPELOID
Definition and Epidemiology
This uncommon cutaneous infection, also known as “erysipeloid of Rosenbach,” is caused by Erysipelothrix rhusiopathiae, a Gram-positive pleomorphic bacillus. The distribution of the causative microorganism is worldwide. Domestic swine, rodents, and birds are the most common reservoirs. Fish flesh is usually not infected, but the bacteria commonly contaminates the slimy coating over fish scales. Erysipeloid is typically occupationally acquired, and it is seen in butchers, fishermen, fish handlers, slaughterhouse workers, veterinarians, and other persons having contact with the animal vectors. The infection is rare in children.
Etiology
E. rhusiopathiae is a linear or slightly curved thin bacillus, measuring 0.8 to 2.5 in length. Sometimes, the organisms are arranged in single, short chains, or they may occur in groups with a “v” configuration. They are Gram-positive bacteria, but may look Gram-negative because the organisms decolorize easily. Erysipelothrix is not acid-fast. Isolation from the skin requires taking a sample from the edges of cutaneous lesions, keeping in mind that the bacteria typically are most numerous in the deep tissues. Isolation
in culture is difficult. Biochemically, the bacteria produce neuraminidase and hyaluronidase.15
in culture is difficult. Biochemically, the bacteria produce neuraminidase and hyaluronidase.15
Clinical Presentation
Erysipeloid is a form of bacterial cellulitis. The lesions are commonly located on the hands. Edema, erythema, and throbbing pain are part of the classical presentation. A history of a cut or a puncture while handling contaminated material can often be obtained. Systemic symptoms are uncommon. The cutaneous lesions are well-demarcated violaceous plaques, with a clear center and a diamond shape. Suppuration is rare, and lesional pain is often severe. The latter two findings separate erysipeloid from erysipelas and other types of bacterial cellulitis.
Pediatric cases are uncommon and are usually seen in immunocompromised patients. Their lesions may also be located on the feet, evolving from hemorrhagic blisters to eschars, and a discrete history of environmental exposure to the organism may be absent.16
Histologic Findings
The epidermis in erysipeloid is spongiotic or necrotic, and subepidermal bulla formation has also been described. Massive edema of the papillary dermis is common, and the subjacent corium contains neutrophils, eosinophils, lymphocytes, plasma cells, and histiocytes. Blood vessels may be dilated, with enlarged endothelial cells.17 The classical description of erysipeloid states that bacteria cannot be seen histologically in the lesions. However, a recently reported case in a child did feature visible bacteria in a tissue Gram stain.16
Differential Diagnosis
The main differential diagnoses for erysipeloid are other infections that can involve the hands and feet, such as erysipelas, other forms of bacterial cellulitis, cutaneous anthrax, and blistering distal dactylitis (BDD). Viral infections, including herpetic whitlow and orf, may be considered, as well as “fish tank granuloma,” caused by Mycobacterium marinum infection. The dense neutrophilic infiltrate is an important clue to the presence of a bacterial infection.
CAPSULE SUMMARY
ERYSIPELOID
Erysipeloid of Rosenbach is a rare cutaneous infection of the hands and feet that is caused by E. rhusiopathia. Butchers, fish handlers, veterinarians, and other persons who have contact with animal vectors are predisposed to this disease, but a clear environmental exposure history may be lacking in children with erysipeloid.
BLISTERING DISTAL DACTYLITIS
Definition and Epidemiology
Blistering distal dactylitis (BDD) is a bacterial infection that affects the volar images of the distal fingers or toes. It is caused in the majority of cases by Group A S. pyogenes.
The disease was initially described as one that was mainly seen in children between the ages of 2 and 16 years. Recently, adult cases have also been reported in immunocompromised and diabetic patients18,19 as well as in infants who are less than 9 months of age.20 So far, no examples have been described in elderly persons.
Etiology
The microorganism considered as the most common cause of BDD is Group A, beta-hemolytic S. pyogenes. In a minority of cases, S. aureus and Group B streptococci have been implicated as well.
Clinical Presentation
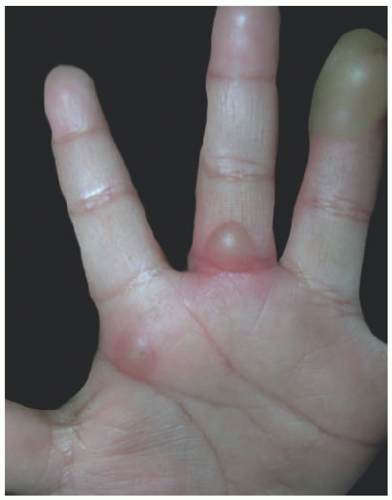
Blisters develop on the distal volar fat pad of a finger or occasionally on a toe. The proximal phalanges and palms are more rarely involved. The lesions may evolve into erosion, which may be variably painful or asymptomatic. More than one digit can be affected, and more than 1 lesion can be seen (Figure 17-10); cutaneous bullae are oval, measuring 1 to 3 cm. Many patients have associated oropharyngitis, conjunctivitis, and perianal cellulitis, but systemic symptoms are usually absent.
Findings
The only reference regarding histologic findings in BD is a description of blistering due to massive subepidermal edema.21
Differential Diagnosis
The most common differential diagnosis is that of herpetic whitlow; indeed, cases of combined streptococcal dactylitis and herpetic infection have been described. Trauma, burns, dyshidrosis, contact eczema, psoriasis, and insect bites can also be considered as clinical possibilities.
CAPSULE SUMMARY
BLISTERING DISTAL DACTYLITIS
This presents as a distal blister on a finger pad of a child or teenager that is caused by Group A Streptococcus. Massive subepidermal edema may be present histologically.
BACTERIAL CELLULITIS
Definition and Epidemiology
Cellulitis is an infection of the dermis and subcutaneous tissue by Staphylococcus or Streptococcus sp. or Gram-negative bacteria. The main difference between this condition and erysipelas is that the infection is more deeply seated.
Cellulitis can occur at any age, especially when there is a break in the skin. Special types worth mentioning include newborn cellulitis caused by Group B streptococci and Haemophilus influenzae type b cellulitis. The latter is still prevalent in developing countries where Haemophilus vaccines are not routinely used.
Etiology
Common pathogens that cause cellulitis in children include S. pyogenes and S. aureus, followed by S. pneumoniae; Groups B, G, or C streptococci, E. coli, and H. influenzae type b.
Clinical Presentation
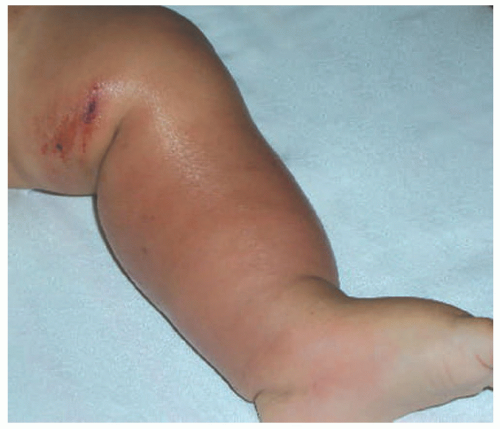
Redness, swelling, tenderness, increased local temperature, and poor demarcation of the cutaneous lesions are classically seen in the presentation of cellulitis. Most patients have fever, chills, and malaise, and some may develop sepsis. Periorbital cellulitis can be associated with MRSA (Figure. 17-11).
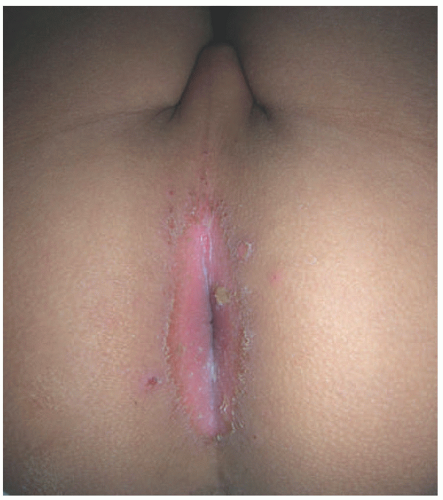
Perianal cellulitis affects young children. It manifests with striking erythema of the anus (Figure 17-12), painful defecation, and bloody stools. The etiology is almost always S. pyogenes,22 and affected individuals may also have simultaneous streptococcal pharyngitis. Haemophilus cellulitis has special characteristics in pediatric patients. It is usually facial, with either a perioral or periorbital plaque, and a reddish-blue to purple color. This condition occurs in children who are less than 2 years old, following a respiratory or sinus infection, and it has the potential to involve the central nervous system.
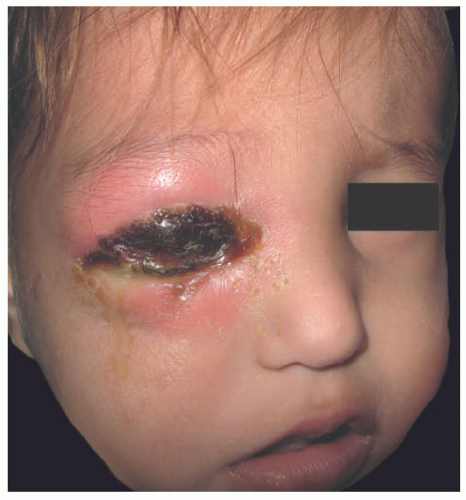 FIGURE 17-11. Cellulitis of the periorbital area. The patient subsequently developed osteitis. Courtesy of Dra. Rosa Ines Castro. |
Histologic Findings
Bacterial cellulitis is characterized by a deep dermal and subcutaneous infiltrate of neutrophils. When suppurative ulceration is seen clinically, necrotizing vasculitis and thrombosis are common histologically. In necrotizing forms of cellulitis, bacteria are usually visible microscopically, but they may be absent otherwise.
Differential Diagnosis
The main clinical differential diagnosis includes various infections with organisms that are capable of producing cellulitis, including fungi, Vibrio vulnificus, and other bacteria.23 As true of erysipelas, Sweet syndrome and rheumatoid dermatosis can also be considered.
CAPSULE SUMMARY
BACTERIAL CELLULITIS
Bacterial cellulitis is a deeply seated bacterial infection. Diffuse neutrophilic infiltrates are present in the deep dermis and subcutaneous tissue.
NECROTIZING FASCIITIS
Definition and Epidemiology
Necrotizing fasciitis (NF) is a rare but aggressive infection of the skin and soft tissue that involves subcutaneous tissue and fascia. It has the potential for widespread tissue destruction and carries a high mortality rate.
The estimated prevalence of NF in pediatric patients is 0.08 per 100 000 children per year,24 and overall mortality is around 15%. The incidence of this condition in children is bimodal, with 2 peaks—less than 2 years and around 10 years of age. Mortality in adults can be as high as 30%, even with appropriate therapy.25
Etiology
Group A streptococci and S. aureus are the main isolates in reported pediatric cases of NF. Superantigens of streptococci, such as SpeG and the streptococcal mitogen exotoxin Z (SmeZ), bind to CD4+ T cells, B cells, monocytes, and dendritic cells, overstimulating the local immune response and causing tissue necrosis.24
Many instances of staphylococcal NF are caused by community-acquired MRSA. P. aeruginosa is the third most common bacterial cause and is always associated with some sort of immunodeficiency.26 Polymicrobial infections are less common in children with NF when compared with adults.
Clinical Presentation
Although preceding trauma is a common inciting factor for NF, children may develop the condition without it. In adults, symptoms and signs that raise the suspicion of NF include pain out of proportion to the clinical lesions, rapidly enlarging, bullae, ecchymoses, anesthesia, crepitation on palpation, and accompanying sepsis. In children, tenderness may be more common than pain. Fever is common in neonates, and also in teenagers. The anatomic location for the lesions varies depending on patient age; under 1 year, truncal disease predominates, whereas the extremities or head and neck are the most common locations in older children. Fournier gangrene can occur in children under 3 years of age; it is related to unsanitary diaper use (Figure 17-13).
Histologic Findings
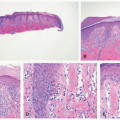
Microscopic findings include dense neutrophilic infiltrates in the deep dermis, subcutaneous tissue, and underlying fascia (Figures 17-14 and 17-15). Tissue necrosis and vascular thrombosis are commonly seen, and Weedon defines NF as a form of septic vasculitis.21 Bacteria can be present in sheets, and they may be seen with Hematoxylin and eosin (H&E) stain.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree