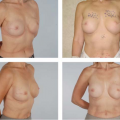
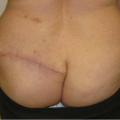
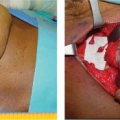
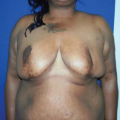
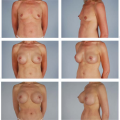
Considerations of Previous Augmentation in Subsequent Breast Reconstruction
Scott L. Spear
Mark W. Clemens
Michael A. Howard
Introduction
Breast cancer is the most common cancer in women and the second-leading cause of cancer deaths in the United States (1). In 2008 alone, 180,000 new cases were diagnosed and 40,00 deaths were attributed to breast cancer. At the same time, two million American women have breast implants, a population that is rapidly growing (2). The reality is that a significant number of these women, as they age, will develop breast cancer. This has led to several studies on this unique patient population. No long-term studies have demonstrated delayed detection of breast cancer or a poorer prognosis in previously augmented women. It is important to note that numerous large studies have come to the definitive conclusion that breast implants do not cause breast cancer (3,4,5). This chapter reviews our experience with reconstructing the previously augmented breast.
Oncologic Considerations in Augmentation Patients
Cancer Epidemiology
Historically, some have been concerned that breast augmentation might increase a woman’s risk of developing cancer. This concern resulted in part from lab studies showing sarcoma formation in rats when exposed to a silicone implant (6). However, several large population studies failed to demonstrate a higher risk of breast cancer in augmented women (3,5,7,8,9,10,11,12). These populations even trended toward lower cancer rates than the nonaugmented patients.
Of interest, some research has even suggested that breast implants may reduce the risk of breast cancer in humans (3,13). Mechanisms postulated for this possibility include immune system upregulation due to the presence of a foreign body (14) and implant-related tissue compression resulting in decreased blood flow to the breast and reduction in breast gland temperature, which leads to reduced metabolic rate. This protective effect remains theoretical and we do not advocate breast augmentation as a strategy for breast cancer risk reduction.
Cancer Stage At Diagnosis
Due to the presence of the implant, concern also exists that breast cancer screening will be less effective, thereby leading to patients presenting with more-advanced breast cancer stage at diagnosis. Several studies have been published looking at this topic (Tables 73.1 to 73.4) and demonstrate that patients present with similar tumor size, lymph node status, and overall stage, as well as have a similar prognosis to nonaugmented patients (5,15). Our data for pathologic staging of 28.1% stage 0, 28.1% stage I, 37.5% stage II, 6.3% stage III, and no stage IV is similar to the postaugmentation Calgary experience of 12.2% stage 0, 36.6% stage I, 36.6% stage II, and 4.9% stage III tumors. Furthermore, several studies indicate that the breast augmentation population does not experience a delay in diagnosis or a higher rate of cancer recurrence and death (16,17,18).
In summary, today’s literature supports the view that (a) implants do not cause cancer and (b) cancers that do develop in augmented women are similar in presentation to those in nonaugmented women.
Breast Cancer Screening in Patients with Implants
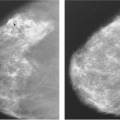
Physical examination and mammography are the mainstays of initial breast cancer screening and detection (19) (Table 73.1). With traditional mammographic techniques, portions of the breast are potentially obscured by a large implant. With traditional mammography techniques, subglandular implants have been reported to obscure at least 44% of breast tissue, while submuscular implants, in comparison, may obscure up to 25% (20). Breast imaging was improved by Eklund, who advocated taking additional mammographic views with the breast implant displaced toward the axilla (21,22). Capsular contracture, if present, may make this technique more difficult, and the sensitivity of mammography is further reduced by the presence of Baker grade III or IV capsular contracture (23). With minimal capsular contracture, visualization was reduced by 30%, compared to 50% loss in moderate to severe capsular contracture. It follows that implants could elevate the false-negative rate and decrease the sensitivity of mammography, in theory delaying cancer detection (24,25,26,27,28).
Over recent years, we have noted an increased rate of initial cancer detection in augmented females from 38% to 59% between 2001 and 2007. This may be attributable to improvements in conventional compression mammography and the increased use of subpectoral implant placement. Now that displacement views are standard, current large studies indicate that women with implants, when compared to age-matched controls presenting with cancers of similar stage, size, estrogen receptor status, and nodal status, actually tend to be of lower grade (29). The theory is that the implant may actually provide a platform against which to palpate tumors, improving breast exam efficacy (30). The literature also does not suggest that implants increase the false-positive rate for self-exam or mammography.
Magnetic resonance imaging (MRI) is emerging as a valuable tool for breast imaging. A large meta-analysis of 4,271
patients found 144 breast cancers detected by MRI, for an overall cancer yield of 3% (31). The sensitivity of MRI ranged from 71% to 100% across the studies. MRI may have a role in screening high-risk patients, but no clear standard has emerged (32).
patients found 144 breast cancers detected by MRI, for an overall cancer yield of 3% (31). The sensitivity of MRI ranged from 71% to 100% across the studies. MRI may have a role in screening high-risk patients, but no clear standard has emerged (32).
Table 73.1 Method of Cancer Detection Among Women With Mammary Augmentation | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Recent literature has raised a concern that some approaches to augmentation may affect the ability to perform a sentinel lymph node biopsy (33,34,35). The theory is that transaxillary and possible periareolar incisions can lead to scarring, which may impair lymphatic mapping and accurate sentinel lymph node identification. In a patient who has received a transaxillary augmentation, Shons advocates a full axillary dissection over a sentinel lymph node biopsy (36). However, the American Society of Clinical Oncology guidelines advocate a sentinel lymph node biopsy if the surgery was performed more than 6 months earlier, stating that the lymphatic drainage from the upper portions of the breast should be intact (37). Previous studies have shown a 100% success rate in identifying sentinel lymph nodes after augmentation (27).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree