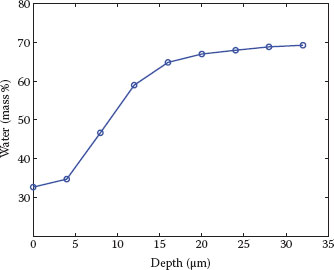
FIGURE 10.1 Principle of measurement of CRS. (a) Monochromatic laser light is focused in the SC in a volume of about 5 μm length (axial resolution) and 1 μm width (dimension of the laser spot). Photons interact with the molecules, releasing some of their energy. Of the photons that exit the skin, only the ones coming from the focus region are detected thanks to the presence of a confocal pinhole. (b) The photons which underwent frequency shifts due to the release of energy to molecules during interaction are used to obtain Raman spectra. The position and the intensity of each peak are representative of the different molecules and their amounts, respectively. Raman spectra can be obtained in a low-(fingerprint) as well as high-energetic region; each region contains different information about the molecular composition of the SC. (c) By focusing the laser light at different depths, concentration profiles of molecules are obtained. The minimum measurement time for acquiring a spectrum ranges from 1 to 3 s according to the energetic region. (Modified from Falcone et al. (12).)
Technical Implementations
The first commercially available confocal Raman instrumentation for in vivo measurements on human subjects was introduced on the market in 2004 (model 3510 skin composition analyzer, RiverD International B.V., the Netherlands) (13). The device consists of an inverted microscope coupled to a Raman microspectrometer assembled on a top-table configuration (10). Laser light is transmitted from the source to the sample through a flat fused silica window. The skin of the volunteer has to be placed in contact with the window, and laser light is focused at different depths by means of a precision translation table. While this top-table implementation allows obtaining an excellent refractive index matching, leading to maintenance of a good depth resolution and of a high signal-to-noise ratio, it limits the body locations that can be measured (14).
An alternative technical implementation overcoming this inconvenience is based on coupling the Raman microspectrometer with fiber optic probes for sample irradiation and scattered light collection (15). While this design allows easier handling of the probe with access to more body locations, technical issues linked to the fiber optic design arise, such as lower depth-resolving power in the SC and strong background signal in the fingerprint region given by the fused silica used in the fiber construction (14).
In vivo confocal Raman instruments may typically employ one or two laser sources in the visible or near infrared range (10,11); the choice of the wavelength depends on the application, since shorter wavelengths allow faster collection of Raman spectra but generate higher tissue autofluorescence in the fingerprint region, while longer wavelengths generate less autofluorescence but require longer exposure times (13).
Characterization of SC Molecular Composition and Structure
Water
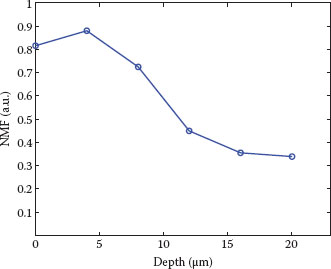
Water content measured with CRS is usually expressed as the ratio of the Raman signal intensity of water band (due to OH-stretching vibrations) integrated from 3350 to 3550 cm−1 to that of protein (due to CH3 symmetric stretching vibrations) integrated from 2910 to 2965 cm−1 (10). Normalization by the protein band serves to compensate for signal losses taking place at increasing depths. A typical water concentration profile obtained with CRS is shown in Figure 10.2. The validity of this method of water measurement was demonstrated by comparison with the Karl Fisher titration method in an ex vivo study (16). Decreased capacitance values, as well as subjective assessment of dry skin, have been reported for sensitive skin in experimental and epidemiological studies, respectively (4,17). As CRS is validated and gives water concentration as a function of depth in the skin, we recommend benchmarking of subjective assessments as well as of traditional biophysical measurements with the direct measurement of the water content in the SC by means of CRS. Besides measuring water concentration at baseline, the water holding properties of the SC could be evaluated dynamically following, for example, exogenous water application (18,19). Changes in water holding properties could be due to an interplay of factors linked to barrier function structure and composition (such as corneocyte maturity, lipid amount and organization, surface path length (19)), water binding to hygroscopic substances (natural moisturizing factor [NMF]) (20), and water diffusion in SC due to different mobilities of water molecules. Many of these factors can be measured by CRS, as described in the following paragraphs. For what concerns water diffusion in SC, a study showed that CRS can distinguish the contributions of three different water-binding states (unbound, partially bound, and totally bound), determined by the strength of hydrogen bonds between the water molecules and other SC components (21).
FIGURE 10.2 Water concentration profile measured on the volar forearm of a healthy volunteer. Water concentration shows a steep gradient across the SC and reaches a plateau in the upper epidermis. The determination of water concentration is quantitative (i.e., expressed in grams of water per 100 g of wet tissue or mass percentage) because of the addition of a calibration factor based on a solution of protein in water (see data in Caspers et al. (10)).
As a last remark, one study reported that two distinct groups of subjects could be identified based on the difference in water profiles measured with CRS before and after repetitive tape stripping: one group with almost no changes in the water profile and a group in which considerable changes were present (22). The difference in water influx from the viable epidermis following tape stripping could reflect differences in SC composition and be possibly linked to sensitive skin.
Natural Moisturizing Factor
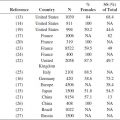
NMF is a hygroscopic mixture of several substances, including amino acids and their derivatives, mainly derived from the degradation of the epidermal protein filaggrin (FLG), as well as components such as urea, lactate, sodium, and potassium, derived from eccrine sweat (23). NMF is an efficient humectant, helping to bind water within the cells and to maintain skin hydration and flexibility (24). It could therefore be another parameter of interest in the evaluation of water-handling properties in the SC of subjects with sensitive skin. The relation between the amount of (bound) water and the NMF content across the SC could also be explored (25). NMF information in Raman spectra can be extracted from the fingerprint region, and a semiquantitative method based on least square fitting with good agreement with in vitro results has been described (10). A typical NMF concentration profile obtained with CRS is shown in Figure 10.3.
Less clear is the role of NMF content on the impairment of skin barrier. Previous studies have shown that patients with atopic dermatitis (AD) carrying mutations in the FLG gene have significantly lower NMF content with respect to wild-type AD patients; at the same time, no significant differences could be found in the impairment of the skin barrier evaluated with TEWL (26,27). Similarly, sensitive skin could be characterized by a decrease in NMF, explaining the frequent association with dryness symptoms, and yet no detectable difference at the level of skin barrier impairment measured by TEWL.
FIGURE 10.3 NMF concentration profile measured on the volar forearm of a healthy volunteer. Increase of NMF from the bottom of the SC is in correspondence with the degradation process of FLG. The characteristic depletion near the skin surface has been associated with washing-out processes, for example, due to daily cleansing (24). NMF is expressed here as the weighted sum of the dominant constituents pyrrolidone carboxylic acid, ornithine, serine, proline, glycine, histidine, and alanine. The determination of NMF concentration is semiquantitative (i.e., expressed in arbitrary units, relative to the concentration of keratin) (see data in Caspers et al. (10)).
Intercellular Lipids
The lipid matrix surrounding the corneocytes in the SC plays an essential role in the skin barrier function by preventing loss of water and other electrolytes and by blocking the entry of exogenous compounds (28). The lipid matrix is composed of a mixture of ceramides, cholesterol, and fatty acids arranged in parallel layers (lamellae) between the corneocytes; within the lamellae, lipids are present in three different lateral organizations, ranging from a very dense to a disordered liquid phase (29). An imbalance of the intercellular lipids has been suggested among the mechanisms leading to an impaired barrier function in sensitive skin (3). In addition, a study demonstrated decreased ceramide levels in the face of subjects with sensitive skin (30). Several spectral features providing direct information on the lateral organization and the conformational order of the lipid chains have been identified in both the fingerprint and high wave number regions (31). In addition, the same semiquantitative method developed for NMF can be used to differentiate cholesterol from ceramides/fatty acid contribution (10). Finally, lipid content can be expressed as the ratio of the Raman signal of lipids (due to CH2 asymmetric stretching) integrated from 2866 to 2900 cm−1 to that of protein (due to CH3 symmetric stretching vibrations) integrated from 2910 to 2965 cm−1 (32).
Besides the diagnosis of sensitive skin, lipid measurement with CRS could be beneficial for studies of the adverse effects of psychological stress on sensitive skin and on other skin disorders. In fact, stress resulted as one of the main triggering stimuli of sensitive skin in an epidemiological study (33) and an association with the exacerbation of several skin disorders is recognized (34). Epidermal lipid synthesis was shown to decrease following acute psychological stress, possibly due to increased glucocorticoid levels; this, in turn, would decrease the production and secretion of lamellar bodies, necessary for the recovery of barrier function following perturbation (34). Such mechanism would explain the delayed recovery of barrier function following tape stripping in the presence of various types of stress (35,36).
Stratum Corneum Thickness
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree