The Prevalence of Self-Reported Sensitive Skin in Large Cohorts Identified by Surveys
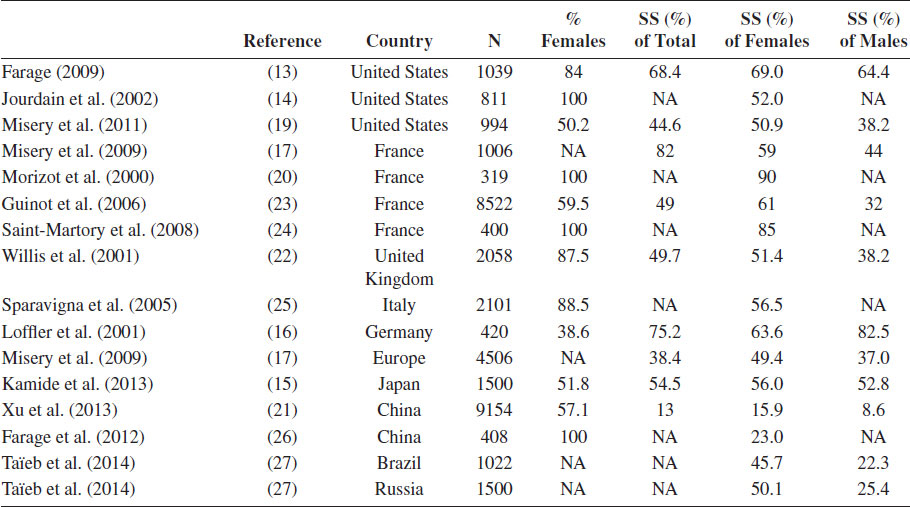
Note: Percentages are either extracted or calculated from the result sections of the original articles. NA, not applicable; SS, subjects with sensitive skin.
Clinical Manifestation
There is no consensus on the symptom profile of sensitive skin. People assessing their skin as sensitive experience unpleasant sensations of the skin, particularly characterized by stinging, burning, or itching sensations (20,24,28). Remarkably, visible signs of skin irritation, as erythema or skin dryness, are absent in many individuals (20,22,24,29,30). Since the symptoms reported by subjects are rather nonspecific, the differentiation from or relationship with dermatological disorders such as irritant contact dermatitis, allergic contact dermatitis, rosacea, physical urticaria, and dermographism, xerosis, (atopic) dermatitis, and photodermatoses may be challenging. Deeper understanding of sensitive skin will enable placing this condition in the context of skin pathology.
Factors impeding the detection and definition of sensitive skin are the frequent absence of clinical signs and subjective character of symptoms (20,31,32) and the heterogeneity thereof (20).
Eliciting Factors
Sensitive skin perceptions are elicited by exogenous and endogenous factors that usually have a considerably low impact on individuals and frequently do not cause skin irritation (13,18,22,23,25).
In order to define the symptom profile and to address specific exogenous and endogenous factors which may elicit skin reactions, nonuniform comprehensive questionnaires were conducted in different populations (18,22,23), showing various risk factors and triggers. Among these factors, the important triggers are cosmetics and soaps (20,21,24–26), environmental climate [temperature changes (14,15,20,21), heat (14,15,20,24,25), cold (14,15,20,25), and dampness (21)], sun exposure (14,21,24), stress or emotions (20,24,25), wind (14,15,25), and friction from clothes (24).
Having a dry or greasy skin (17,21), a history of childhood atopic dermatitis (AD) (17,21,22), being female (21), and having a fair skin type (21) are important host factors increasing the risk of having sensitive skin (21).
Subject Selection in Clinical Studies
To enable conduction of clinical studies in a population with sensitive skin and with nonsensitive skin, assumptions on the definition and inclusion criteria have to be made. Erroneous selection of subjects may possibly lead to formulating wrong conclusions on pathomechanisms. Moreover, since symptoms might be subclinical in situations in which the skin is not challenged, differences in physiology may be minute and difficult to detect, and therefore careful selection of subjects with sensitive skin is of high importance.
A performed systematic literature review on objective measurements on sensitive skin identified studies including subjects by means of perception, for example, burning, stinging, itching, and general skin discomfort (33). This self-reported skin sensitivity was determined (i) by sensory skin reactions following application of specific stimuli or (ii) by questionnaire. Subsequently, the relation of the group characteristics of the selected populations and the selected study population with objective measurements were studied, both at baseline and following specific stimulations.
Subject Selection and Provocation by Stimuli
Many research groups select sensitive subjects and nonsensitive subjects as a control group by evaluation of sensory discomfort after application of a chemical agent on the skin. Lactic acid is one of the most frequently used agents (34–43), and the lactic acid stinging test (LAST) was previously even proposed as the best diagnostic test available for sensitive skin (44). LAST identifies “stingers”—subjects perceiving sensations of stinging after application of lactic acid on the nasolabial fold—which are assumed to correlate with sensitive skin. A detailed evaluation of studies including subjects by self-perceived sensitive skin and using LAST as a provocative method to enhance skin reactions shows that in subjects with sensitive skin, higher stinging scores are observed (45), time to onset and peaking of stinging response are shorter (45), and the overall stinging scores are higher (46,47). However, the test is subjective in nature and lacks sensitivity, 59.9–80% (27,45,48) of subjects with sensitive skin report a positive reaction to LAST, and specificity, 66.7% of nonsensitive subjects experience this reaction (48) for sensitive skin. Thus, lactic acid mainly elicits some, but not all key symptoms in the clinical profile of sensitive skin, predominantly skin stinging and slightly burning. This might be the reason for the insufficient sensitivity. In addition to this, studies have shown that a positive response to one substance does not predict a reaction to another substance (40,45,49–52), questioning the validity of using a single substance to test skin sensitivity. Next to using lactic acid as a challenge, studies of sensitive skin reactions and sensations have been performed using a wide range of chemical agents. They include but are not limited to sodium lauryl sulfate (SLS), capsaicin, menthol, benzoic acid, trans-cinnamic acid, octane, cumene, methyl nicotinate and acetyl-β-methylcholine chloride (vasodilators), ethanol, allergens, occlusion, cocamidopropyl betaine and benzalkonium chloride (surfactants), and balsam of Peru. Furthermore, material interactions (53) and electrical provocations (54) followed by noninvasive measurements of the biophysical parameters of the skin have been appraised in people reporting perceptions of sensitive skin to quantify sensory irritation and to reveal the underlying mechanisms of sensitive skin. Many provocations used in experiments resulted in sensory skin reactions, but, again, these might not be specifically addressing the sensitive skin.
Subject Selection by Questionnaire
An increasing number of studies select subjects by means of self-perceived sensitive skin inventoried by questionnaires, using different definitions of sensitive skin, as explained in the “Epidemiology” section. Some research groups included solely the question, “Do you have a sensitive skin, yes or no?” In one study, skin sensitivity was scored on a labeled magnitude scale, permitting both semantic descriptors and a continuum of intensity rankings to compare individuals (55). In contrast, other research groups conducted extensive nonuniform questionnaires addressing sensory, objective symptoms and additionally encompassing numerous potentially eliciting factors by means of inclusion of subjects with sensitive skin and with nonsensitive skin.
A ten-item questionnaire was developed by Misery et al. (56). With this questionnaire, researchers made an attempt to establish standardized patient questionnaires, which might allow scoring in a reproducible manner on a “sensitive scale” and might be suited for monitoring the evolution of the skin condition (56). However, only a selected population with sensitive skin diagnosed in a private clinic was included in this study, limiting the application of the developed scale as a diagnostic tool for selecting sensitive and nonsensitive subjects for research purposes.
Pathomechanisms and Objective Readouts
Kligman et al. (57) used a novel nomenclature for sensitive skin by proposing different subtypes of sensitive skin defined by clinical presentation or possible underlying etiology as follows: (i) subjective irritation refers to an irritant response without visible clinical signs; (ii) neurosensory irritation signifies neutrally mediated responses such as itching, stinging, burning, and tightness; (iii) chemosensory relates to sensory responses induced by chemicals in contrast to physical, mechanical, and environmental factors; and (iv) psychophysical irritation implies a psychological component. Willis et al. (22) described several clinical forms depending on the intensity of clinical symptoms: (i) Very sensitive skin is dry or fatty and bitterly reacting to both exogenous factors, that is, cosmetic products and environmental factors, and endogenous features. The clinical symptoms are acute and permanent, and both factors trigger determining psychological reactions. (ii) Environmentally sensitive skin is skin that is often clear, dry, and thin and essentially reactive to environmental factors, that is, heat and rapid temperature changes, with frequent bouts of flushing. (iii) Cosmetically sensitive skin is essentially reactive to cosmetics. This intolerance is lighter and often limited to some identifiable cosmetic products. Farage and Maibach also proposed a heterogeneous phenomenon with multiple etiologic aspects (58); Richters et al. (33) critically appraised the evidence of objective measurements at baseline and following skin provocations. This research group chose an approach by including studies which had selected sensitive subjects based on perception. The translation from skin perceptions to objective measurements was made, and it was shown that the strongest evidence exists for the role of an impaired skin barrier in sensitive skin. This impaired barrier might cause sensory perceptions, and vascular reactivity might develop subsequently.
However, strong evidence and consistency on pathways underlying sensitive skin is still lacking at this moment.
Skin Barrier Impairment
Many research groups focused on the skin barrier function and measured transepidermal water loss (TEWL) as a parameter of skin barrier function. In some studies, a higher TEWL was observed in the unchallenged skin of subjects with sensitive skin compared to that in subjects with nonsensitive skin (16,29,37,59). In stingers, a higher TEWL was also measured compared to that in nonstingers (34,37,38,41,52). Additionally, a higher TEWL at baseline seems to correlate with stronger TEWL increase following provocation (34): plastic occlusion resulted in significantly longer evaporation halflife time in subjects with sensitive skin (59), and a trend to increased recovery time of TEWL following SLS stimulation was also observed. A low hydration of the stratum corneum is also associated with an impaired skin barrier. A significantly lower stratum corneum hydration in facial areas in subjects with sensitive skin (47) and in stingers (34) and a nonsignificantly lower stratum corneum hydration in facial areas in subjects with sensitive skin were found (37). Challenging the skin chemically resulted in a significantly lower stratum corneum hydration in stingers compared to that in nonstingers (41). Clinically, a dryer skin is also observed (25). In contrast to these findings, other studies found no difference in stratum corneum hydration between subjects with sensitive skin and subjects with nonsensitive skin (16,29) or between stingers and nonstingers (34,37,41) either challenged or unchallenged. The skin of stingers with self-reported sensitive skin appears to be rougher since Fast Fourier transform evaluation of tapes after tape-stripping the stratum corneum in 243 stingers reveals lower contents of cells (42). This implies a more irregular, rougher, and possibly less hydrated skin as well as impaired intercorneocyte adhesion. Objectively measured skin elasticity and distensibility show no correlation with hypersensitivity (43).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree