Keywords
Psoriasis, Combination, Acitretin, Cyclosporine, Methotrexate, Biologics, Etanercept, Adalimumab
Key points
- •
Combination therapies of biologics and systemic agents for psoriasis treatment can potentially enhance efficacy of treatment, increase onset of remission, and decrease side effects by allowing for dose reductions.
- •
Potential therapeutic combinations examined include methotrexate paired with biologics; cyclosporine paired with biologics; acitretin paired with biologics; or phototherapy paired with biologics.
- •
Randomized clinical trials, case series, and case reports chronicling these therapeutic combinations have demonstrated good safety and efficacy data, indicating an important future role for combination therapy in the treatment of psoriasis.
Introduction
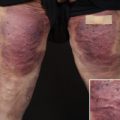
Psoriasis is a chronic inflammatory disease that negatively affects approximately 2% of the US population with significant impact on quality of life. Approximately 20% of these patients have moderate-to-severe disease. Its systemic effects include cardiovascular, metabolic, rheumatologic, and psychiatric comorbidities with older studies showing 25% to 38% of patients with psoriasis dissatisfied with their traditional treatments. Of those affected with psoriasis, 17% require treatment beyond traditional topical corticosteroids, such as phototherapy or systemic agents. More traditional systemic treatments include methotrexate, cyclosporine, and acitretin with newer biologic treatments, such as ixekizumab, secukinumab, ustekinumab, adalimumab, etanercept, and infliximab, quickly becoming important players in the therapeutic arena. A new oral agent, apremilast, has also been in use since 2014, with a few case reports of its use in combination with other therapies.
As treatment methods progress for this chronic disease, clinicians have been forced to expand therapeutic regimens and explore new combination therapies for patients with recalcitrant disease or dose-related cumulative toxicity from traditional treatments. Combination treatment of biologics with systemic agents, such as methotrexate, cyclosporine, and acitretin, can potentially enhance efficacy, hasten onset of remission, and decrease potential side effects by allowing for dose reductions ( Table 15.1 ). When combining immunosuppressive agents, the possibility of additive immunosuppression should be considered, and the dosage and period of overlap of more than one immunosuppressive medication should be minimized. This chapter investigates different combination therapies with biologics and traditional oral treatments.
| Combination Therapy | Source | Dosing | Comparisons | Study Type |
|---|---|---|---|---|
| Methotrexate + Ustekinumab | Puig, 2012 | Ustekinumab 90 mg (transient increase in injection frequency) + methotrexate 10 mg/wk | None | Case report |
| Methotrexate + Adalimumab | Philipp et al, 2012 | Standard dosing adalimumab + methotrexate 12.4 ± 4.5 mg/wk | None | Retrospective chart review |
| Methotrexate + Infliximab | Dalaker & Bonesronning, 2009 | 3 mg/kg infliximab at week 0, 2, 6, then q 8 wk + methotrexate 7.5–15 mg/wk | None | Retrospective chart review |
| Wee et al, 2012 | 3 mg/kg or 5 mg/kg infliximab at week 0, 2, 6, then q 8 wk + methotrexate 5–20 mg/wk | None | Retrospective chart review | |
| Methotrexate + Etanercept | Gottlieb et al, 2012 | Etanercept (50 mg twice weekly × 12 wk then 50 mg/wk × 12 wk) + methotrexate (7.5–15 mg/wk) | Etanercept monotherapy (50 mg twice weekly × 12 wk then 50 mg/wk × 12 wk) | Randomized, controlled trial |
| Zachariae et al, 2008 | Etanercept (50 mg twice weekly × 12 wk then 25 mg twice weekly × 12 wk) + methotrexate (stable dosing × 3 mo) | Etanercept (50 mg twice weekly × 12 wk then 25 mg twice weekly × 12 wk) + methotrexate (stable dosing × 3 mo then 4 wk taper) | Randomized, controlled trial, open label | |
| Gelfand et al, 2008 | Etanercept (50 mg twice weekly × 12 wk, then continuous or interrupted etanercept 50 mg/wk × 12 wk) + stable dose of methotrexate | Etanercept monotherapy (50 mg twice weekly × 12 wk, then continuous or interrupted etanercept 50 mg/wk for 12 wk) | Randomized, controlled trial, open label | |
| Driessen et al, 2008 | Etanercept (50 mg twice weekly × 12 wk, then etanercept 25 mg/wk twice weekly × 12 wk) + methotrexate (2.5–35 mg/wk) | None | Case series | |
| Cyclosporine + Etanercept | Yamauchi & Lowe, 2008 | Cyclosporine (200 mg BID until PASI-50), then etanercept (50 mg weekly while cyclosporine tapered) | None | Case series |
| Lee et al, 2010 | Cyclosporine (200 mg daily) + etanercept (50 mg weekly) | None | Case series | |
| Cyclosporine + Adalimumab | Karanikolas et al, 2011 | Cyclosporine (2.5–3.75 mg/kg/d) + Adalimumab (40 mg weekly) | Cyclosporine (2.5–3.75 mg/kg/d) monotherapy or adalimumab (40 mg weekly) | Nonrandomized, open-label clinical trial |
| Acitretin + Etanercept | Gisondi et al, 2008 | Etanercept (25 mg weekly) + oral acitretin (0.4 mg/kg daily) | Etanercept monotherapy (25 mg weekly) or oral acitretin (0.4 m/kg daily) | Randomized, controlled trial |
Methotrexate
Methotrexate is the most frequently used oral treatment for psoriasis and has been found to be significantly effective in randomized, controlled trials, open-label studies, and retrospective studies. Combination therapy of methotrexate with biologics is an emerging field of treatment with many considerations. Importantly, combining biologic therapies with methotrexate has not been shown to increase the bone marrow toxicity or hepatotoxicity of methotrexate. Although there is no published literature about the combination of methotrexate with the newer anti-interleukin-17 (IL-17) drugs, secukinumab or ixekizumab, one would expect the combination to be more effective than monotherapy with either agent.
Methotrexate and Ustekinumab
Ustekinumab is a human monoclonal immunoglobulin G antibody that binds to the p40 subunit of IL-12 and IL-23. Dosing is weight based with a 45-mg or 90-mg dose every 12 weeks for a patient weighing 100 kg or less or greater than 100 kg, respectively. For patients who are not adequately treated with ustekinumab alone, combination therapy has been seen to be useful particularly in those suffering from psoriatic arthritis, as a bridging therapy, for palmoplantar disease control or for recalcitrant disease.
The success of the combination of methotrexate and ustekinumab was highlighted in a case report of a patient who had failed therapy with psoralen and UV-A (PUVA), cyclosporine, infliximab, etanercept, and various combination therapies, including etanercept/methotrexate, etanercept/methotrexate/acitretin, adalimumab/efalizumab/cyclosporine, cyclosporine/efalizumab/cyclosporine/methotrexate, and cyclosporine/adalimumab. Responses to all these regimens and combinations had slowly lost their efficacy, usually after just a few months. Treatment with ustekinumab showed improvement within 1 month, although response was eventually lost as with the patient’s other therapies. By adding methotrexate 10 mg to the regimen along with a slight increase in injection frequency, the patient regained response with persistent control.
Methotrexate and Adalimumab
Adalimumab is a fully human monoclonal antibody approved for use with plaque psoriasis at a dose of 80 mg at week 0, 40 mg at week 1, and then 40 mg every other week. A retrospective chart review examined responses to combination therapy with standard dosing of adalimumab, without loading doses for some patients, and methotrexate dosing at 12.4 ± 4.5 mg per week. In 27 of 32 patients, a very good to moderate response, defined as a Physician’s Global Assessment (PGA) score of 0, 1, 2, or 3 was seen. In fact, a reduction in methotrexate dosage was possible in 5 patients. Combination therapy showed a good safety profile with AEs limited to mild infections, abdominal complaints, and diarrhea. Adalimumab was stopped in 1 patient due to recurrent infection; gastrointestinal effects were ameliorated with methotrexate dosage adjustments. de Groot and colleagues evaluated inflammatory markers in psoriatic lesional skin with decreased markers seen in combination therapy compared with methotrexate or adalimumab alone.
Data from clinical trials for rheumatoid arthritis show increased levels of adalimumab in patients treated with combination therapy as compared with adalimumab alone. Thus, levels of adalimumab may be increased by an unknown mechanism when using it in combination with methotrexate. In fact, tumor necrosis factor-α (TNF-α) antagonists used with methotrexate have been approved for the use of rheumatologic diseases. Studies have also shown a decrease in antiadalimumab antibody formation in patients on combination therapy.
Overall, larger trials are lacking, but positive results have been seen in multiple case series with psoriasis or psoriatic arthritis as well as randomized trials for those with rheumatoid arthritis. Certainly, more data are needed to fully elucidate risks and benefits of this combination therapy.
Methotrexate and Infliximab
Infliximab, a TNF-α antagonist, is a chimeric monoclonal antibody shown to be effective at a dosage of 5 mg/kg for the treatment of psoriasis. Approved for combination usage of infliximab 3 mg/kg with methotrexate or infliximab 5 mg/kg with azathioprine in the treatment of rheumatoid arthritis and Crohn disease, respectively, such combination therapy for psoriasis is less common.
A subanalysis in the IMPACT 2 (Infliximab Multinational Psoriatic Arthritis Controlled Trial 2) studied the efficacy of combination methotrexate (25 mg/wk or less with mean dosage 16.2 mg/wk) and infliximab. At week 54 of the trial, Psoriasis Area Severity Index (PASI)-75 was achieved in 53% of patients on combination therapy compared with 48% of patients on infliximab monotherapy. Safety profiles were similar between the 2 groups.
A retrospective chart review was conducted on 23 patients with psoriasis treated with 3 mg/kg infliximab combined with intramuscular methotrexate (7.5–15 mg every week). All patients had previously failed treatment with methotrexate, cyclosporine, or PUVA. Infliximab infusions were given at weeks 0, 2, 6, and then every 8 weeks. If patients maintained response after 6 months of treatment, infusion intervals were lengthened to 9 weeks up to 14 weeks. Most patients were given infusions every 8 to 10 weeks. Twenty-one patients reached 50% improvement by week 14 of treatment. The remaining 3 patients continued with treatment with sustained improvement. Treatments effects included headache, dizziness, and thromboembolism in a patient with multiple other risk factors. Mild infections were also reported and treated successfully with standard therapy. No hepatotoxicity, which is traditionally seen with methotrexate therapy, was reported. To address loss of response, which has previously been reported with infliximab monotherapy after 1 year of treatment, treatment intervals were shortened or methotrexate dose was increased.
Generally, it is advantageous that infliximab be dosed with concomitant methotrexate to inhibit the formation of antidrug antibodies. It is also thought that the concurrent dosing of methotrexate during the study reduces such immunogenicity. Another study by Wee and colleagues showed the incidence of infusion reactions was much lower in those receiving infliximab with methotrexate than with those receiving only infliximab. Studies in both psoriatic arthritis and rheumatoid arthritis have established better patient outcomes using combination therapies with methotrexate. Lower doses of TNF-α antagonists are also more cost-effective for the patient. Infliximab has been associated with hepatosplenic T-cell lymphoma in inflammatory bowel disease patients treated concurrently with azathioprine. Thus, caution should be taken in using combination treatment and should not be first line because larger clinical trials are needed.
Methotrexate and Etanercept
Etanercept is a TNF-α antagonist used for moderate-to-severe psoriasis at doses of 50 mg twice weekly for up to 3 months and then 50 mg once weekly. Current recommendations for patients with psoriatic arthritis include the approved use of etanercept with methotrexate for those who have not responded to methotrexate alone. In fact, long-term safety of this combination therapy has been established in patients with rheumatoid arthritis.
Randomized, controlled trials have shown superior efficacy of etanercept and methotrexate therapy compared with etanercept monotherapy in psoriasis. In the largest randomized, controlled trial (n = 478) lasting 24 weeks, Gottlieb and colleagues dosed etanercept at 50 mg twice weekly for the first 12 weeks and then 50 mg/wk for 12 weeks with 1 group also receiving methotrexate dosed at 50 mg/wk and 7.5 to 15 mg/wk, respectively. Results showed 77.4% of patients in the combination group obtaining 75% disease improvement compared with 60.3% in the monotherapy group. There were a higher number of adverse events (AEs) seen in the combination therapy group, 75%, as compared with the monotherapy group, 60%, with reported events being similar in the 2 groups.
In a randomized, open-label, 24-week study, Zachariae and colleagues chronicled adding etanercept to the treatment regimen of patients who had failed or had little effect from methotrexate for 3 months. Patients were randomized to either etanercept with methotrexate continued or etanercept with methotrexate eventually tapered and discontinued over a 4-week period. Etanercept was dosed at 50 mg twice weekly for 12 weeks and then 25 mg twice weekly for 12 weeks. Of the patients who had been on etanercept/methotrexate combination as compared with etanercept/methotrexate taper, 66.7% versus 37.0% were judged as “clear” or “almost clear.” PASI-75 scores were also seen to be improved at both weeks 12 and 24 in the combination group. AEs were increased with statistically significant infectious AEs in the combination therapy group compared with the etanercept monotherapy group. Most AEs in either group were mild or moderate. Mild-to-moderate hepatic enzyme elevation was seen in 17.9% and 12.9% of the etanercept/methotrexate taper and combination group, respectively. Thus, etanercept and methotrexate combination therapy is a good treatment option with safety profiles generally being similar between the groups.
Finally, the EASE study (Etanercept Assessment of Safety and Effectiveness), a multicenter, randomized, open-label trial, compared patients with psoriasis receiving continuous etanercept 50 mg twice weekly for 12 weeks, followed by continuous or interrupted etanercept 50 mg every week for 12 weeks. Those on a stable dose of methotrexate for 8 weeks before baseline were allowed to continue with results showing those on a combination treatment of etanercept and methotrexate were more likely to achieve a PGA score of 0 to 2 at week 24 compared with not being on methotrexate. In fact, many of the patients were able to decrease their weekly methotrexate dosage or even eventually suspend its use.
Another small case series in high-need patients with psoriasis showed the efficacy of combination therapy for maximal therapeutic effect without significant difference in laboratory findings or AEs compared with methotrexate monotherapy.
Pharmacologically, the greater response seen with combination therapy has yet to be explained. Studies of lesional and nonlesional skin with other combination therapies suggest increased targeting of inflammatory markers with such treatment compared with monotherapy alone.
In fact, large rheumatologic studies, such as TEMPO (Trial of Etanercept and Methotrexate with Radiographic Patient Outcomes) and COMET (Combination of methotrexate and etanercept in active early rheumatoid arthritis), assessing etanercept and methotrexate combination therapy found no significant differences in terms of serious adverse events (SAE) reported or serious infections.
Caution is advised because few trials have analyzed the potential for increased AEs with combination therapies. One exception, the CORRONA (Consortium of Rheumatology Researchers of North America) database for rheumatoid arthritis patients, found that combination therapy of methotrexate with TNF antagonists, particularly adalimumab, etanercept, and infliximab, was not associated with higher risk of infection compared with infection rates of monotherapy of any agent alone. Finally, a Swedish registry for psoriatic arthritis patients found similar SAE rates for those on combination therapy compared with methotrexate alone. Another source chronicled the potential use of etanercept with methotrexate coupled with subsequent tapering of methotrexate to a minimal effective dose. This combination lowered the risk of toxicity. In fact, this combination therapy has been shown to be effective in rheumatoid arthritis, juvenile idiopathic arthritis, and psoriatic arthritis.
Thus, etanercept and methotrexate combination therapy seems to be a successful alternative to treatment of recalcitrant disease with multiple studies and reports outlining its successful use.
Cyclosporine
There are limited studies on the use of cyclosporine with biologics. The information in later discussion highlights the data currently available on combination therapies with cyclosporine.
Cyclosporine and Etanercept
Yamauchi and Lowe evaluated 8 patients who received cyclosporine 200 mg twice a day until reaching PASI-50, at which point etanercept was started at 50 mg weekly while cyclosporine was concomitantly tapered. This combination therapy maintained response during tapering as well as 12 weeks after tapering concluded. Lee and colleagues administered cyclosporine 200 mg daily with etanercept 50 mg weekly to 7 patients until symptom improvement with subsequent dose reduction of both etanercept and cyclosporine. All 7 patients showed progress with PASI improvements at 94.9% after conditioning therapy lasting an average of 6.85 weeks and 93.2% after maintenance therapy lasting an average of 56.5 weeks. Finally, 1 small pilot study for patients with psoriatic arthritis (n = 11) added cyclosporine 3.0 mg/kg/d to those with inadequate dermatologic response to etanercept. Nine patients achieved PASI-75 by week 24 with AEs, including worsening of hypertension and elevated creatinine. Other combination therapy studies have evaluated patients already on cyclosporine treatment who require eventual cessation once disease control has been achieved with concomitant etanercept treatment. Although few and small, these studies highlight the potential for cyclosporine and etanercept as effective combination therapy, although larger clinical trials are needed.
Cyclosporine and Adalimumab
The combination of cyclosporine and adalimumab was investigated in an open-label trial for patients with psoriasis and psoriatic arthritis refractory to methotrexate. This study showed a PASI-50 rate after 12 months of treatment in 95% of patients on combination therapy compared with an 85% and 65% PASI-50 rate in those receiving adalimumab and cyclosporine alone, respectively. Another case series investigated 5 patients who transitioned without flare from cyclosporine to adalimumab with a cyclosporine taper ranging from 6 to 11 weeks.
Cyclosporine and Apremilast
A recent case report highlighted the use of both cyclosporine and apremilast in a 45-year-old man who had previously failed or developed adverse reactions to methotrexate, phototherapy, ustekinumab, adalimumab, and etanercept. Although taking certolizumab for joint pain, the patient was started on apremilast 30 mg twice daily. He eventually discontinued his certolizumab without return of joint symptoms but continued to have generalized psoriasis. He restarted cyclosporine at 100 mg twice a day, after a “cyclosporine holiday” due to 2 years of continuous dosing at 400 mg daily and achieved almost complete clearance. Thus, the patient was able to achieve a greater level of treatment success with combination therapy and a lower dose of cyclosporine than previously achieved. Although more data are certainly needed, this case report shows the potential use of combination therapy with apremilast.
Acitretin
Acitretin is an oral retinoid used as monotherapy in psoriasis treatment in both pustular and plaque psoriasis. Because acitretin monotherapy is not as effective as most other psoriasis treatments, it is usually prescribed in combination with other treatments, especially phototherapy.
Smith and colleagues evaluated 15 patients in a retrospective chart review detailing combination therapy with acitretin and biologics. Overall, 29% showed complete clearance, 43% showed PASI 90, 14% showed PASI 75, and 7.1% showed no change. More data and clinical trials are certainly needed, but this study highlights the potential for therapeutic success with acitretin combination therapy.
Acitretin and Etanercept
A 24-week, randomized trial by Gisondi and colleagues included 60 patients randomized to either combination etanercept (25 mg weekly) and oral acitretin (0.4 mg/kg daily) (n = 18), etanercept alone (25 mg twice weekly) (n = 22), or oral acitretin (0.4 mg/kg daily) alone (n = 20). At week 24, PASI-75 was seen in 44% of patients receiving combination therapy, 45% of patients receiving etanercept monotherapy, and 30% of patients with acitretin monotherapy. Body surface area improvement at week 24 was 78% in the combination group, 80% in the etanercept group, and 46% in the acitretin group. Safety profiles were the same in all 4 groups. This study suggested using etanercept in combination with acitretin was equally as effective as using etanercept monotherapy. Of note, the etanercept monotherapy and combination therapy groups both had greater improvement than acitretin alone. Other smaller clinical case series on the combination of etanercept and acitretin showed good disease control with no increase in AEs. One study examined possible cases in which this combination would be recommended, such as in those with a history of nonmelanoma skin cancer in whom phototherapy would not be recommended. Because acitretin has potential protective properties against the development of nonmelanoma skin cancer, it may warrant use in a patient with a history of nonmelanoma skin cancer.
Acitretin and Apremilast
One case report of complete clearance of palmoplantar psoriasis in a 66-year-old woman was reported. The patient had previously failed various topical corticosteroids, combination betamethasone dipropionate and calcipotriene ointment, and tazarotene 0.1% gel. She was subsequently started on systemic acitretin treatment at 10 mg daily, which was increased to 20 mg daily, combined with 10 months of biweekly excimer laser therapy. After 10 months, the patient was unable to continue excimer laser therapy and was started on apremilast with standard dosing escalation to 30 mg twice daily. After 3 months of combined acitretin and apremilast therapy, complete resolution of all symptoms was reported.
Biologics and Phototherapy
Etanercept in combination with narrowband UV-B (NBUVB) has been evaluated in multiple studies all showing a low rate of AEs. Kircik and colleagues showed etanercept (50 mg twice weekly) combined with NBUVB (3 times per week) evaluated in 86 patients allowed for a PASI-75 rate of 84.0% at week 12, with PASI-90 and PASI-100 being achieved by 58.1% and 26%, respectively.
Park and colleagues conducted a study during which 30 patients all received therapy with etanercept, 50 mg twice weekly, for 12 weeks, after which half received treatment with NBUVB 3 times per week for 12 weeks and a once weekly dose of 50 mg etanercept. At week 24, 53% of patients in the combination group had achieved PASI-75, whereas only 47% on monotherapy achieved similar results. Of note, it was found that combination therapy with etanercept did not show significant improvement compared with etanercept monotherapy.
Lynde and colleagues studied etanercept, 50 mg per week, with NBUVB in 75 patients who had failed to reach PASI-90 improvement after 12 weeks of etanercept monotherapy. At week 16, of those patients adhering to thrice weekly phototherapy sessions, 43% reached PASI-90 improvement compared with 3% in the monotherapy group ( P <.05).
In a 1-arm, open-label study, De Simone and colleagues evaluated 33 patients receiving 50 mg weekly of etanercept and NBUVB 3 times weekly for 8 weeks, then followed by etanercept monotherapy for 4 weeks. After week 8, 15% of patients had achieved a PASI-90, and after week 12, 58% achieved a PASI-90.
Wolf and colleagues evaluated the use of NBUVB in those patients failing to achieve PASI-75 after 6 weeks of etanercept monotherapy. Mean PASI reduction was 89% for the combination therapy group compared with 68% for those undergoing etanercept monotherapy ( P <.05).
Bagel evaluated the effectiveness of adalimumab with NBUVB in 20 patients with all receiving adalimumab, 80 mg at week 0 and then 40 mg every other week, and NVUVB 3 times weekly. At week 12, 95% of patients had reached PASI-75 with 55% achieving PASI-100. From weeks 12 to 24, patients received no treatment, with 65% of patients maintaining their PASI-75.
In another study, Wolf and colleagues treated 4 patients with standard dosing of adalimumab with half the body treated with 311-nm NBUVB once per week for 6 weeks. After 6 weeks, there was a statistically significant mean PASI reduction of 86% on the phototherapy-treated side compared with 53% on the untreated side.
Wolf and colleagues also completed a similar study with ustekinumab therapy. Ten patients were treated with ustekinumab at weeks 0 and 4 along with 311-nm NBUVB 3 times per week. There was a statistically significant 82% reduction in PASI on the half also treated with phototherapy versus a 54% mean reduction in PASI score on the side without phototherapy.
Thus, phototherapy appears to be an effective adjunctive treatment to biologic treatment.
In summary, combination therapy achieves greater improvements in psoriasis. Ideally, combination therapy would allow dosage reduction of some individual treatments. Conversely, additive immunosuppression is a risk for increased malignancy and infection and should therefore be minimized.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree