19 Closed technique rhinoplasty
Synopsis
 Nature is predictable – therefore nasal phenomenology can be understood.
Nature is predictable – therefore nasal phenomenology can be understood.
 Rhinoplasty has consistent “behavioral” rules, like all surgery – therefore the surgeon can control the result.
Rhinoplasty has consistent “behavioral” rules, like all surgery – therefore the surgeon can control the result.
 Nasal deformities are not limitless or lawless but follow patterns – therefore their solutions follow patterns.
Nasal deformities are not limitless or lawless but follow patterns – therefore their solutions follow patterns.
 Sequential intraoperative photography teaches nasal behavior and structural interactions.
Sequential intraoperative photography teaches nasal behavior and structural interactions.
 Follow the technical rules that apply to all other surgery: limit dissection, morbidity, and tension on closure.
Follow the technical rules that apply to all other surgery: limit dissection, morbidity, and tension on closure.
 Never forget the patient’s own aesthetic.
Never forget the patient’s own aesthetic.
 Remember that most problems are under the surgeon’s control.
Remember that most problems are under the surgeon’s control.
The anatomy and science necessary for surgical success
Rhinoplasty as a right brain operation
Rhinoplasty is probably the most right-brain operation that plastic surgeons perform.1 Unfortunately, our educational system cultivates the verbal, rational, analytical, and numerical left side of the brain, tending to ignore the right side. This is one of the reasons that most adults draw with the same level of sophistication that they possessed as a 10-year-old child, which is, not coincidentally, the age at which the left brain becomes dominant. The ease with which surgeons can perform rhinoplasty correlates directly with the facility with which they can deliberately involve right-brain function.
The most attractive noses in nature do not look assembled but unified as a whole, in which every part seems to belong. This principle should also apply to rhinoplasty results. The beauty of the assembled result, the finished surgical product, depends on the ability to see the preoperative excesses and deficiencies in shape and proportion as they really are, with no preconceptions; balance them against the patient’s surgical goals; and from those accurate observations create an effective surgical plan. The most direct means to this kind of analysis involves significant right-brain contributions. Right brain function can be cultivated2 by analytic exercises and by studying silhouettes of patient profiles.1
Equilibrium and balance
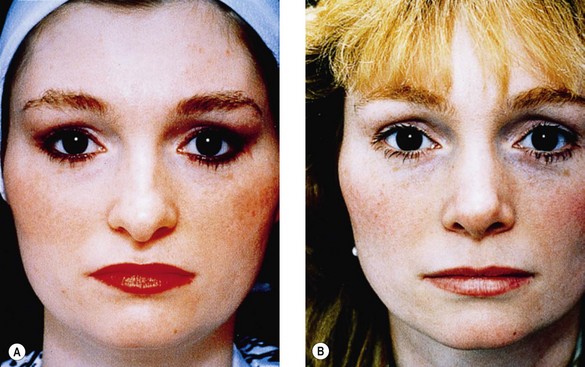
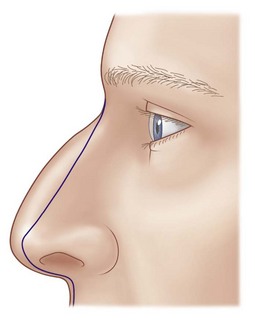
The fact that many post-rhinoplasty nasal configurations, and all “end-stage” noses, look the same, is not a coincidence. The response of any nasal skeleton and its investing soft tissues to reduction is not idiosyncratic but consistent and therefore predictable.3,4 These forces collapse the upper nose caudally and medially over the pyramidal bony and upper cartilaginous vaults, cephalad at the columella, posteriorly over the maxillary arch, and posteriorly and concentrically around the tip lobule (Fig. 19.1). Whether the nasal skeleton has been reduced by surgery, trauma, congenital anomalies, or Wegener’s granulomatosis, the essential external appearance remains the same. Variations occur because of disequilibrium between skeletal and soft tissue volumes or differences in soft tissue thickness and elasticity, not because biologic forces differ from patient to patient.
If the reaction of the nose to reduction rhinoplasty is not idiosyncratic but predictable, it follows that the surgeon who can predict the nasal response to certain interventions can control the result. Useful here is the concept of a dynamic equilibrium,3 in which preoperative nasal shape represents not a static structure but rather a dynamic equilibrium, the sum of balanced, opposing forces between the nasal soft tissues and their underlying support. At the start of a rhinoplasty, the nose is equilibrated. Skeletal reduction during rhinoplasty disrupts this preoperative skeletal and soft tissue equilibrium; the nose in disequilibrium at the conclusion of a rhinoplasty cannot remain the same. Soft tissue and skeletal contraction occurs until the nose re-establishes its internal equilibrium or until contraction can no longer occur. The degree of disequilibrium at the end of the procedure thus determines the amount of redraping and contraction that will occur postoperatively: the bigger the disequilibrium, the bigger the postoperative surprise. The surgeon therefore ideally controls the postoperative equilibrium by permitting soft tissue contraction only where it is most predictable (e.g., over the bony and cartilaginous dorsa) and by minimizing contraction where it is less predictable (e.g., the lower nasal third). In doing so, the surgeon immediately gains a powerful tool for influencing postoperative nasal contour.
The structural nasal layers
In understanding structural nasal interrelationships, it is helpful to conceptualize the nose as a system of two interrelated layers (Fig. 19.2). The outer layer, like a soft, elastic sleeve, slides over the inner semi-rigid layer and contains the entire investing nasal soft tissues plus the alar cartilages and their associated lining. The inner layer contains everything else (the bony and upper cartilaginous vaults, the nasal septum, and their associated linings). This two-layer concept associates those structures that behave together anatomically and functionally, and provides an explanation for the “global” manifestations of some surgical changes (e.g., the effect of dorsal reduction or augmentation on nasal length).5
Upper cartilaginous vaults
The width and stability of the upper cartilaginous vault (formed by the upper lateral cartilages and the anterior septal edge), the critical area of the internal nasal valves, depend not only on the width of the bony vault but also on the height and width of the middle vault roof.6 Resection of the middle vault roof during hump reduction removes this most critical anterior stabilizing force on the upper lateral cartilages, which will fall medially and produce a characteristic “inverted V” deformity and consequent narrowing at the internal valves.7 Middle vault collapse virtually always occurs when the cartilaginous roof has been resected, whether or not osteotomy has been performed, but may not be visible if the overlying soft tissues are sufficiently thick; when the nasal skin is thin, the deformity can be impressive (Fig. 19.3). To avoid middle vault collapse and internal valvular incompetence, the surgeon should plan to reconstruct the normal distracting forces by a substantial dorsal graft or by spreader grafts,8,9 which provide the same degree of functional mean nasal airflow improvement (see below).
Middle and lower cartilaginous vaults
The point of intersection of the upper and lower lateral cartilages creates the “watershed” area between the internal and external nasal valves, and aggressive surgery in this area also affects external valvular competence, particularly in patients whose alar cartilage lateral crura are cephalically rotated.9,10 Compared with caudal support of the upper lateral cartilages, however, anterior support provided by the intact cartilaginous roof is more profound and more critical to airway function.
Dorsum and tip
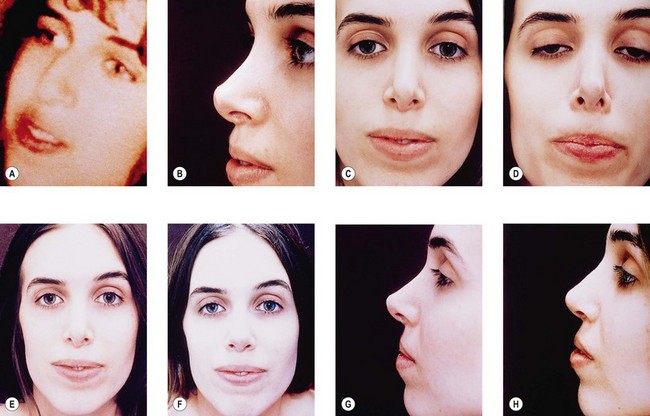
Tip projection, that is, the intrinsic ability of the alar cartilages to support the tip lobule independent of dorsal height, depends on alar cartilage middle crural size, shape, and substance (Fig. 19.4). Tip projection extremes are the easiest to identify. The nasal tip in Figure 19.4A depends not on bridge height but on intrinsic alar cartilage strength and projection. Conversely, the nasal tip in Figure 19.4B is partially dependent on bridge height; therefore, tip projection will decrease when the dorsum is resected. It is important to identify inadequate tip projection preoperatively so that its correction can be incorporated in the surgical plan; inadequate tip projection cannot be rendered adequate by bridge resection alone. The tip, the lower nasal skin, and in many patients nearly the lower half of the profile line depend on suspension of the skin sleeve by the alar cartilages, which, like a “cap” anterior to the remaining skeleton, maintain lower nasal tension. The alar cartilages carry a larger responsibility for caudal nasal support than may at first be evident.
Four common anatomic variants that predispose to unfavorable results
A great deal can be learned about primary rhinoplasty by observing the deformities of secondary rhinoplasty patients seen in consultation and by examining one’s own unsatisfactory results. A retrospective study of 150 consecutive secondary rhinoplasty and 50 primary rhinoplasty patients suggests that only four anatomic variants in the primary nose particularly predispose to unfavorable rhinoplasty results: low radix/low dorsum, narrow middle vault, inadequate tip projection, and alar cartilage malposition.11
Low radix or low dorsum
Low radix or low dorsum begins caudal to the level of the upper lash margin with the patient’s eyes in primary gaze; this variant was present in 93% of the secondary patients and 32% of the primary patients in the series.11 First described by Sheen, the low radix is one of several primary causes of nasal imbalance: an upper nose that seems too small for its lower nasal component.12 When the radix begins lower than the upper lash margin, dorsal length is therefore shorter and so nasal base size appears larger. The classic imbalance may take the form of the depression or notch in the upper nasal third, or a low, straight dorsum may accompany a large nasal base. Whether or not the patient has a dorsal convexity, the surgeon often hears the same complaint: “The tip of my nose sticks out too far.” The surgical dilemma is as follows: If the surgeon reduces the nasal dorsum, the patient’s preoperative skeletal and skin sleeve maldistribution will worsen: the lower nose will appear even larger. The surgeon fortunately has two other choices: either limit tip reduction or raise the dorsum segmentally or entirely to balance the nasal base. Variations of the latter option are generally safer because they require less contraction of the thicker nasal base tissues.
Narrow middle vault
A narrow middle vault (Fig. 19.5) is arbitrarily defined as any upper cartilaginous vault that is at least 25% narrower than the upper or lower nasal third (present in 87% of the secondary patients and 38% of the primary patients in this series). This variant was described by Sheen in conjunction with short nasal bones13 but has been discussed subsequently by that author and others11,14–16 as a trait that places the patient at special risk for internal valvular obstruction, which can exist preoperatively or may be produced by dorsal resection. Descriptions of valvular collapse had appeared earlier in the rhinoplasty literature17–19 but the missing puzzle piece had been the link between resection of the cartilaginous roof and postoperative internal valvular collapse, a phenomenon previously attributed to traumatic or surgical avulsion of the upper lateral cartilages from the nasal bones. Resection of even 2 mm of the cartilaginous roof during hump removal ablates the stabilizing confluence that braces the middle vault, 80 which can now collapse toward the anterior septal edge, restricting airflow at the internal valves and producing a characteristic inverted “V deformity”. Rhinomanometric studies indicate that valvular obstruction is 4 times more common than pure septal obstruction in primary rhinoplasty patients and 12 times more common in secondary patients; reconstruction of incompetent, internal valves by dorsal or spreader graft doubles nasal airflow in most patients.8,20
Inadequate tip projection
A tip with inadequate projection is defined as any tip that does not project to the level of the anterior septal angle. Inadequate tip projection was present in 80% of the secondary patients and 31% of the primary patients in the reported series.11 Despite its common use and importance, the term “tip projection” has been used to connote different things by different authors. Some surgeons assess tip projection by measuring the distance of the most projecting point of the tip from a facial parameter,21,22 and others by the relative proportion of the nasal base segments anterior and posterior to the upper lip23,24 or the relative lengths of the nasal base and upper lip.25 Although these definitions apply in some cases, there are patients whose nasal bases are large but whose tip cartilages are nevertheless poorly projecting (i.e., reflecting a disproportionate amount of lower nasal skin, rather than excessive cartilage size). In these individuals, tip projection may be inaccurately assessed as “adequate” or even “excessive”, even though the alar cartilages still lack the substance required to create a straight bridge line.
An alternative functional definition of tip projection is the relationship of the tip lobule to the anterior septal angle. Alar cartilages sufficiently strong to support the tip to the level of the septal angle are “adequately projecting” (Fig. 19.4A); alar cartilages too weak to do so are “inadequately projecting” (Fig. 19.4B). The practical value of this definition lies in its ability to define treatment: adequately projecting tips do not need increased support, whereas inadequately projecting tips do. Further, by defining tip projection relative to the septal angle, the surgeon can distinguish between two associated but distinct entities: (1) intrinsic anterior supporting strength supplied by the alar cartilages and (2) skin sleeve volume and distribution in the lower nasal third. Inadequately projecting nasal tips often appear to “hang” from the septal angle (Fig. 19.4B). Because an inadequately projecting tip owes its position to dorsal height and not only to tip cartilage support, the surgeon must employ some tip-strengthening method (sutures, struts, or grafts) to create that support. A straight dorsum requires the nasal tip to support itself independent of bridge height; that is, to be adequately projecting. Interestingly, rhinoplasty pioneer Jacques Joseph instinctively solved the problem of inadequate tip resection in his patients by limiting bridge resection and deliberately leaving a dorsal convexity.26
Alar cartilage malposition
“Alar cartilage malposition” describes cephalically-rotated lateral crura whose long axes run on an axis toward the medial canthi instead of toward the lateral canthi, the position of orthotopic lateral crura (Fig. 19.6). This anatomic variation was first recognized by Sheen27 as an aesthetic deformity that produced a round or boxy tip lobule with characteristic “parentheses” on frontal view. Initially believed to be a rare variant, malposition is present in approximately 50% of primary patients and 80% of secondary patients.28 Malposition also has two additional ramifications that are not aesthetic. First, the abnormal cephalic position of the lateral crura places them at special risk if an intercartilaginous incision is made at its normal intranasal location.10 This maneuver could transect the entire rotated lateral crus instead of only splitting the intended cephalic portion: the entire lateral crus may thus be inadvertently removed. Resected or whole, most malpositioned lateral crura do not provide adequate external valvular support, and so malposition is not only associated with boxy or ball tips but also the leading cause of external valvular incompetence.20 The secondary deformity is characteristic.
The adequate treatment of cephalic rotation of the lateral crura requires resection and replacement of these structures, relocation of the lateral crura to support the external valves, or supporting the areas of external valvular collapse with autogenous grafts.29 Prior reports have indicated that approximately 50% of patients presenting with external valvular obstruction have alar cartilage malposition. Correction of external valvular incompetence doubles mean nasal airflow in most patients (2.5 times for primary patients; 4.0 times for secondary patients).8
None of these four anatomical variants (low radix or low dorsum, narrow middle vault, inadequate tip projection, and alar cartilage malposition) always requires treatment. For example, the low radix must always be assessed relative to nasal base size: if the base is small, a low radix may create the best balance. But they do supply cautionary notes. At least one of these four anatomic traits was present in each of the 150 secondary patients in the reported series.11 Some 78% of the secondary patients and 58% of the primary patients had three or all four of the traits. The most common grouping in both primary and secondary patients was the triad of low radix, narrow middle vault, and inadequate tip projection (40% and 28%, respectively) (Fig. 19.7). The second most common grouping was the association of all four anatomic traits (27% of secondary and 28% of primary patients). None of these anatomic variants is adequately treated by classic reduction rhinoplasty, emphasizing the importance of careful preoperative diagnosis.
The effect of rhinoplasty on the airway
Traditional concepts and clinical observations
For many years, the following concepts provided the basis for analysis of the obstructed nose:
1. The bony and cartilaginous septal partition, deformed by congenital or traumatic causes, may obstruct the nasal airway.
2. Compensatory hypertrophy of the contralateral inferior turbinate frequently occurs, so that both airways eventually become obstructed.
Although many patients with obstructed nasal airways undoubtedly improve after septoplasty or submucous septal resection with or without inferior turbinectomy, not all do. Adding to the frustration of patient and surgeon are three other common observations: that there is often a poor correlation between a patient’s symptoms and the apparent site of clinical septal or turbinate obstruction; 18–21 that patients frequently breathe better (preoperatively and even postoperatively) on the “narrower” side (ipsilateral to the septal deviation); and that many patients who have undergone prior successful septoplasty and turbinectomy or who have straight, unoperated nasal septa still complain of airway obstruction.
These apparently inconsistent observations are more understandable if the reader considers airway size to be the product of at least four factors: (1) mucosal sensitivity to the environment or hereditary factors; (2) inferior turbinate hypertrophy from many causes; (3) septal deviation; and (4) position and stability of the lateral nasal wall during the dynamic process of ventilation.8,9,20 Thus, any congenital or acquired weakness or instability of the upper or lower lateral cartilages or their investing soft tissues (which compose the internal and external nasal valves, respectively), along with septal and inferior turbinate size and position, becomes a factor that may profoundly influence the ability to draw adequate volumes of air through the nose.
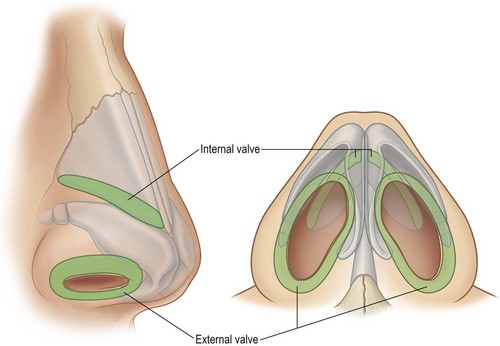
The internal nasal valve is formed by the articulation of the caudal and anterior (or dorsal) edges of the upper lateral cartilages with the anterior septal edge (Fig. 19.8); the external nasal valve is composed of the cutaneous and skeletal support of the mobile alar wall (the alar cartilage lateral crura with their associated external and vestibular skin coverings). The watershed area between the valves occurs at the transverse portion of the alar crease and at the articulation of the caudal edge of the upper lateral cartilages with the alar cartilage lateral crura.
Until recently, efforts to correlate nasal airflow with clinical symptoms had yielded equivocal conclusions. There was argument on both clinical and rhinomanometric grounds that septoplasty either did or did not improve nasal airflow.30–34 For such a commonly and easily diagnosed clinical problem as septal deviation, one would expect to find more unanimity in clinical series. These observations were clouded further by the fact that some 80% of nasal septa in the population at large are “deviated”, and by imprecision in distinguishing internal from external valvular incompetence. Nevertheless, mounting clinical evidence indicates that obstruction at either set of valves may profoundly obstruct the airway, even in the absence of septal deviation.35–58
Results of an airway outcome study in 600 patients
To help resolve some of these conflicting clinical observations and to provide quantitative measurements of the relative increases in nasal airflow after functional septal or valvular surgery, an ongoing study of 600 consecutive patients undergoing surgery for airway obstruction was undertaken between 1991 and 2008.8,59 Patients with septal perforation, atopic patients, and patients requiring turbinectomy were excluded. The diagnosis of septal obstruction or valvular incompetence was made by observation of the nasal airway and nasal sidewall movement with and without a nasal speculum during quiet and forced inspiration (Fig. 19.9). When a flaccid or collapsible valve was supported during inspiration by a cotton-tipped applicator, the patient could usually appreciate an immediate improvement in nasal airflow, thus directing the surgeon toward appropriate operative treatment. Septoplasty and valvular reconstruction by cartilage or bone grafts were performed as indicated. Anterior, active, mask rhinomanometry was performed according to the method of Mertz et al.42,60–62
The data may be summarized as follows:
1. Overall, septal and valvular surgery corrected the airway obstruction in more than 95% of patients in a single operation.
2. Although septoplasty improved airflow ipsilateral to the obstruction, there was no significant improvement in total (geometric mean) nasal airflow after septoplasty alone.
3. Internal valvular reconstruction by dorsal or spreader grafts doubled nasal airflow.
4. Spreader grafts and dorsal grafts were equally effective in supporting the internal nasal valves.
5. External valvular reconstruction doubled mean nasal airflow.
6. The largest postoperative improvement was seen in patients after correction of both internal and external valvular incompetence (more than four times preoperative values).
7. Septoplasty in addition to valvular reconstruction did not significantly improve nasal airflow over the results obtained by valvular reconstruction alone, even in patients observed more than 100 months postoperatively.
8. These results were produced without performing inferior turbinectomy in any of the patients.
9. In those patients with lateralized symptoms, the septum was contralateral to the more obstructed side in 45% of cases.
10. Of the 384 secondary rhinoplasty patients, 94% had previously undergone an adequate septoplasty but were still symptomatically obstructed. Within this group, valvular reconstruction alone corrected the airway in 97% after one operative procedure.
11. When primary and secondary rhinoplasty patients were stratified, the improvement in primary patients equaled or exceeded the improvement achieved in secondary rhinoplasty patients in six of the eight obstructed sites examined.
12. Valvular obstruction was 4 times more common than pure septal obstruction in primary rhinoplasty patients and 12 times more common than pure septal obstruction in secondary rhinoplasty patients.
13. When the entire 600-patient group was stratified, the greatest improvement was observed in those patients observed more than 12 months, supporting the view that the airway continues to enlarge as edema resolves.
14. By measuring airflow during quiet and forced inspiration, sidewall stiffness could be quantified and shown to increase following valvular reconstruction.
15. A decrease in nasal airflow was not obligatory, even in patients with preoperative airway obstruction.
In conclusion, current rhinomanometric data support the concept that lateral nasal wall movement caudal to the bony arch (containing both the internal and external valves) constitutes a cause of airway obstruction equal to or greater than septal deviation in many rhinoplasty patients. By visualizing the airway as a structure in which the septal partition forms one side and the mobile lateral nasal wall forms the other, and in which the turbinates and mucosa function as additional dynamic structures, the inconsistencies in prior clinical observations become easier to explain. Inspired airflow passes through the nose at 15–65 km/h, the latter equivalent to gale force winds.63 The size of any nasal airway thus depends not only on the position and configuration of the septum and turbinates but also on the stability and competence of the nasal valves under significant transmural pressures. The observation that patients often breathe worse on the side contralateral to the septal obstruction (45% of our patients with lateralized obstructions) can therefore be explained through valvular incompetence. In patients with septal deviation and valvular incompetence, the side with the greater airflow and greater transmural pressure (i.e., the side contralateral to the septal deviation) will collapse first and therefore become more symptomatic.
This study did not answer the question of where turbinates fit into the hierarchy of nasal obstructions. In atopic individuals with gross turbinate hypertrophy or polyps, turbinectomy is unquestionably valuable.64 However, the mean nasal airflow improvements listed above were achieved without turbinectomy, even in a population of patients with secondary deformities, valvular incompetence, prior intranasal scars, webs, or vestibular atresia, suggesting that turbinate obstruction in many patients may be largely reactive or secondary and that radical turbinectomy may not often be necessary if septal and valvular factors have been addressed properly.
Patient selection
Fallacies of planning: two false assumptions that lead to unsatisfactory results
Two principles underlie the logic of reduction rhinoplasty, neither of which is always valid:
• FALSE ASSUMPTION NUMBER ONE: The nasal soft tissue cover has the infinite ability to contract to the shape of any underlying skeleton.
If the nose is being reduced, the entire surgical result depends on the validity of this assumption. The nasal skin sleeve does contract according to its quality, thickness, and preoperative distribution, but not necessarily to the shape of the surgically reduced skeleton. The vectors of skin sleeve contraction are related to, but independent of, the volume and contour of the underlying skeleton; the end stage of these vectors is the classic shape of supratip deformity (Fig. 19.1). If this assumption was always true, supratip deformity would never occur and augmentation would not correct it.65
The classic application of this assumption is a preoperative plan in which the surgeon plans to resect all nasal dorsum anterior to a straight line drawn from the nasal radix to the point of the tip (Fig. 19.9). Underlying this strategy is the assumption that dorsal reduction affects bridge height alone. Changes in the nasal skeleton are not independent, however, but rather have global effects outside their areas. Resection of the nasal bridge affects nasal width and length, apparent nasal base size, middle vault support, alar rim contour, and columellar position. Similarly, alar cartilage reduction can affect tip support and projection, nasal length, alar rim contour, and external valvular support. These structural interdependencies are not just regional. Recognizing them is necessary to preoperative planning, interpretation of intraoperative nasal appearance, postoperative success, and the correction of secondary deformities.
The interview
The patient must therefore be guided to understand that every rhinoplasty is a compromise between the patient’s preferred aesthetic goals and the limitations that a predetermined preoperative skeletal and soft tissue configuration imposes. Donor materials vary in quantity, character, and composition, determining their usefulness.66 Finally, many preoperative noses already have some desirable features; patient and surgeon should be careful not to destroy them.
Differences in primary and secondary candidates
Primary and secondary rhinoplasty patients differ in three characteristic ways. First, the secondary patient’s scarred, contracted soft tissues will not tolerate aggressive dissection, multiple incisions, or tight dressings. Second, graft donor sites may have already been harvested, necessitating the use of more difficult (distorted septum or concha), painful (costal), or frightening (calvarial) donor sources. Third, the secondary rhinoplasty patient’s morale is often more fragile. Having already invested money, time, discomfort, and emotion in one or more unsuccessful procedures, what secondary rhinoplasty patients fear most and need least are additional disappointments. The surgeon should be careful to construct a plan that is based on a clear understanding of what is possible and founded on sound surgical and biologic principles that maximize the airway and respect the patient’s aesthetic goals.67
The preoperative examination
Valves
Sidewall collapse with inspiration at one or both of the nasal valves is surprisingly common. It is important to determine why valvular incompetence exists (e.g., prior surgery, intrinsic weakness, or alar cartilage malposition). If sidewall collapse occurs, occlude one nostril and ask the patient to compare flow through the unobstructed airway with and without supporting the collapsing area with a cotton-tipped applicator soaked in 1% Pontocaine hydrochloride for the patient’s comfort. Patients with valvular incompetence will notice an obvious and gratifying increase in airway size. The surgeon may observe valvular collapse and substantial airway obstruction, even in the patient with a straight, unoperated septum and without turbinate hypertrophy. Here, septoplasty may be indicated to harvest grafts but by itself will not open the airway; the surgeon must also place appropriate valvular grafts. Reconstruction of the internal and external valves can triple or quadruple airflow in most rhinoplasty patients, even when septoplasty is not simultaneously performed.8
Septum
The septum should be palpated for substance, contour, and mucosal cover (indicating the sequelae of allergy, injury, perforation, or chronic cocaine use). It is also important to assess whether a “high” (i.e., toward the anterior edge) septal deviation exists; because hump removal can unmask a high septal curvature, the surgeon should be prepared to camouflage or correct the septal deflection with unilateral or asymmetrically thick spreader grafts.14,68
Turbinates
Although the turbinates are time-honored causes of airway obstruction and affect the airways of atopic patients or patients with chronic, severe septal deflection (in which the turbinate contralateral to the septal deviation hypertrophies), clinical and rhinomanometric data indicate that obstructing turbinates are relatively low in the hierarchy of common airway obstruction causes in primary and secondary patients. Because turbinates warm and humidify inspired air, the surgeon should plan conservative resections even in atopic patients. Furthermore, histologic studies have shown that turbinate hypertrophy secondary to septal deviation is characterized by bony, not mucosal overgrowth.69 Therefore, most patients who have had good septal and valvular reconstruction can be adequately treated by only turbinate crushing and outfracture (or no treatment at all).70
Basic nasal aesthetics
On frontal view, the upper nose should be narrower than the lower nose; symmetric, confluent, divergent lines should connect the two. On oblique view, there should be no regional discontinuities, the supratip should be flat, and tip lobular mass should fall below the levels of the peaks of the alar cartilage domes. On lateral and oblique views, nasal length and base size should balance each other. Ideal parameters have been suggested.71–75 The practical difficulty in employing many of these guidelines lies largely in the facts that skin sleeve volume and distribution have already been predetermined and that skin contractility is limited, not infinite. Furthermore, ideal aesthetics do not apply to most patients, even most Caucasians.75 If the surgeon had the latitude of reducing skin volume, the size of the postoperative nose could be altered more radically to a patient’s facial measurements, body habitus, or other parameters. In practice, however, the surgeon works within narrower limits. The airway should be patent and stable on forced inspiration. Beyond these basics, the details depend on the patient’s skeletal framework and soft tissue cover and his or her aesthetic goals. Rhinoplasty offers, as much as or more than any other aesthetic procedure, the possibility of individualizing an aesthetic goal.
Parameters of rhinoplasty planning
What limits the applicability of most proposed aesthetic ideals is the character, volume, and distribution of the skin sleeve and the limitations that they impose. Three soft tissue parameters can be used to form any rhinoplasty plan,76,77 and they will therefore apply to both primary and secondary patients.
Skin thickness and distribution
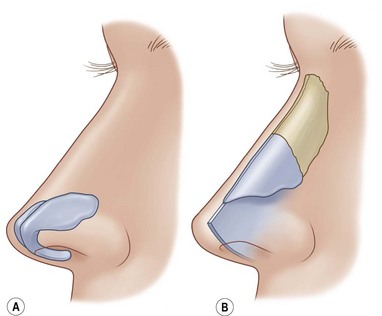
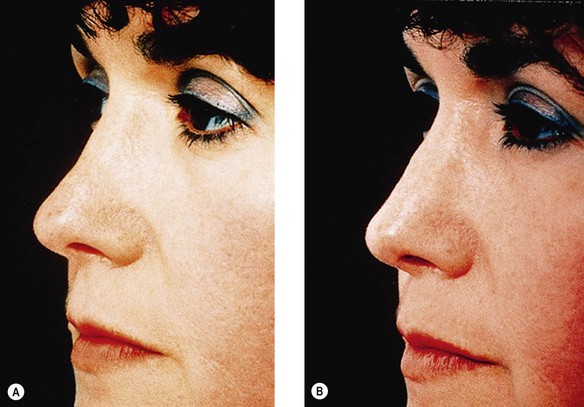
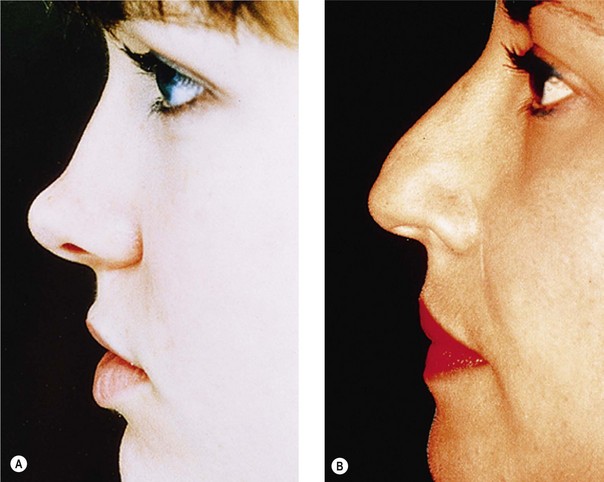
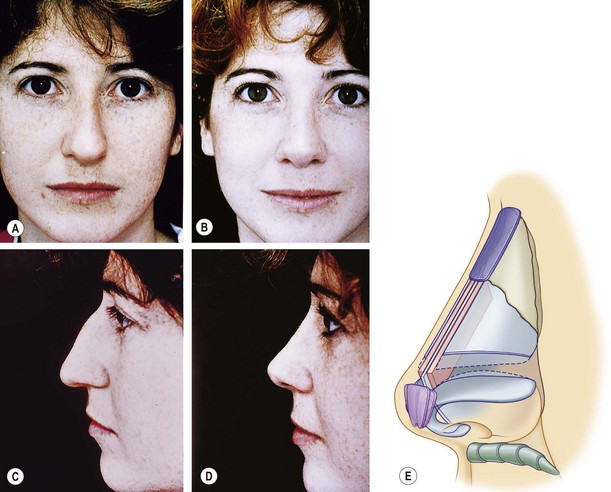
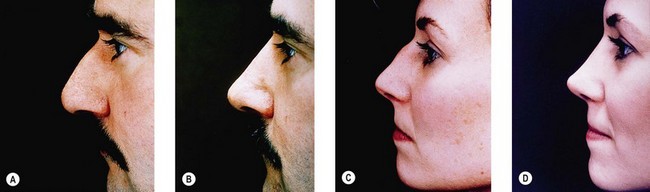
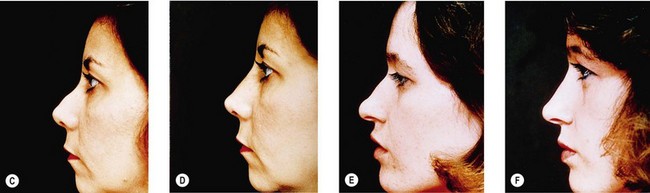
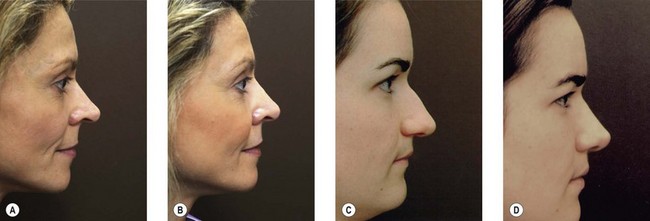
It may be intuitively obvious that skin thickness affects any rhinoplasty plan, but so also does skin distribution. The preoperative large nasal base does not contract into a small nasal base; rather, it contracts to a distorted large nasal base. Skin quality therefore affects both reduction and augmentation. Thicker skin requires more skeletal support and contracts less well; the surgeon must thus be conservative in reduction and will need more substantial grafts to produce a given result (Fig. 19.10A,B). Thinner skin allows greater reduction but requires softer grafts to avoid surface distortions (Fig. 19.10C,D).
Tip lobular contour
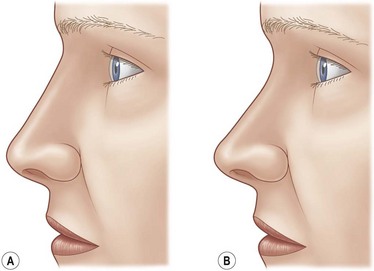
Because the nasal base (the lower nasal third) has a more complex topography than the simpler pyramidal bony and upper cartilaginous vaults, and because the soft tissues are always thicker in the caudal than in the cephalic nose, it follows that the surgeon should select first those maneuvers that provide the best nasal base contours. Ideal tip aesthetics (Fig. 19.11A) require a defined point of greatest projection, a flat supratip, and a tip lobular mass that falls below the point of greatest projection. The poorly shaped tip lobule has the opposite characteristics: a poorly defined, low point of greatest tip projection, a convex supratip, and a tip lobular mass that lies cephalad to the point of greatest projection (Fig. 19.11B).
The balance between nasal base size and bridge height
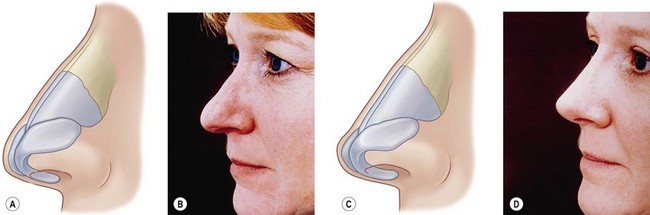
Dorsal reduction or augmentation profoundly affects the apparent size of the preoperative nasal base.77 The higher the dorsum, the smaller the nasal base appears (Fig. 19.12). The reverse is also true: dorsal reduction increases apparent nasal base size. This powerful illusion has its most important practical application in: (1) patients who believe that preoperative nasal base size is excessive, in whom the aesthetic goal may best be reached by a change in balance instead of only size (Fig. 19.13A,B); and (2) patients whose soft tissues are thick, and who therefore may be more successfully treated by the combination of reduction and augmentation (Fig. 19.13C,D), a paradoxical principle that most patients and many surgeons have to see to believe.
Why this author still prefers endonasal rhinoplasty
These are the common objections, but not necessarily the most important ones. First, by separating columellar skin from the medial crura, the surgeon loses an important component of tip stability and projection, which therefore requires some method (suture fixation or columellar strut) to support the medial crura so that a new nasal tip can be made. The strut can impart rigidity to the columella and increases graft requirements. In primary patients, this consideration may be unimportant, but in secondary patients, whose donor sites are already depleted, every bit of graft material counts. Though incisions are limited, endonasal rhinoplasty is not a blind operation. Most procedures are performed under direct vision with greater access than endoscopic surgery permits. The operative strategy, making skeletal changes through limited incisions and judging progress by feeling the surface, is precisely the same discipline required by suction-assisted lipectomy. Limited pocket dissection minimizes the need for graft fixation and simplifies some procedures. Solid or crushed grafts can be used in ways that would be tedious or impossible by the open approach,78 although some solutions have been described.79
The decision to operate
1. Can I see the deformity? This question eliminates delusional patients or those with minimal defects that may not e surgically correctable.
2. Can I personally fix it? This criterion will vary from surgeon to surgeon and must be based on operative experience and ease in correcting specific problems.
3. Can I manage the patient? A patient who is unacceptably nervous, impossible to examine, or unwilling to comply with preoperative and postoperative instructions is a poor candidate, even if all other conditions are met.
4. If there is a complication, will the patient remain controlled and cooperate with treatment? No patient enjoys a complication, but there are those who, although disappointed, quietly understand and will await the proper time for revision. There are others who become hysterical, angry, disruptive, or accusatory and want an immediate correction. From the author’s experience, the personal stress of operating on the latter group and anticipating the outcome if something goes wrong is agonizing. More than that, patients whose emotions are so poorly controlled are in no position to withstand the additional trauma of surgery.
5. Does the patient accept the margin of error inherent in surgery? This is the most important criterion. Some patients (and even some surgeons) have unrealistically optimistic opinions about the degree to which any surgeon can control wound healing; the quality and availability of building materials, the patient’s immune competence, and the myriad other factors, currently known and unknown, that influence surgical outcomes. The patient’s willingness to accept the imperfection that is inherent in surgery is a willingness to accept the imperfection that is inherent in being human.