I. OVERVIEW AND EPIDEMIOLOGY
A. Isolated cleft palate (CP) must be differentiated from cleft lip and palate (CLP)
B. CP
1. 0.5 in 1,000
2. Involves the secondary palate only (posterior to incisive foramen)
3. No ethnic variation in incidence
4. *Often syndromic
a. DiGeorge syndrome (Shprintzen)
i. Most common
ii. Cardiac defects
iii. Chromosome 22q deletion
b. Stickler syndrome
i. Autosomal dominant
ii. Mutation in type 2 collagen
C. CLP (see Cleft Lip (chapter 25) for further information)
1. The vast majority of cleft lips arise spontaneously and are not inherited.
2. Ethnic variation in incidence (Asians > Whites > Blacks)
3. Syndromic conditions are rare (e.g., Van der Woude’s syndrome)
4. Predominantly sporadic
5. Always involves the primary palate, with variable involvement of the secondary palate
a. Forme fruste
b. Cleft lip and alveolus
c. Complete CLP
II. NORMAL PALATE ANATOMY
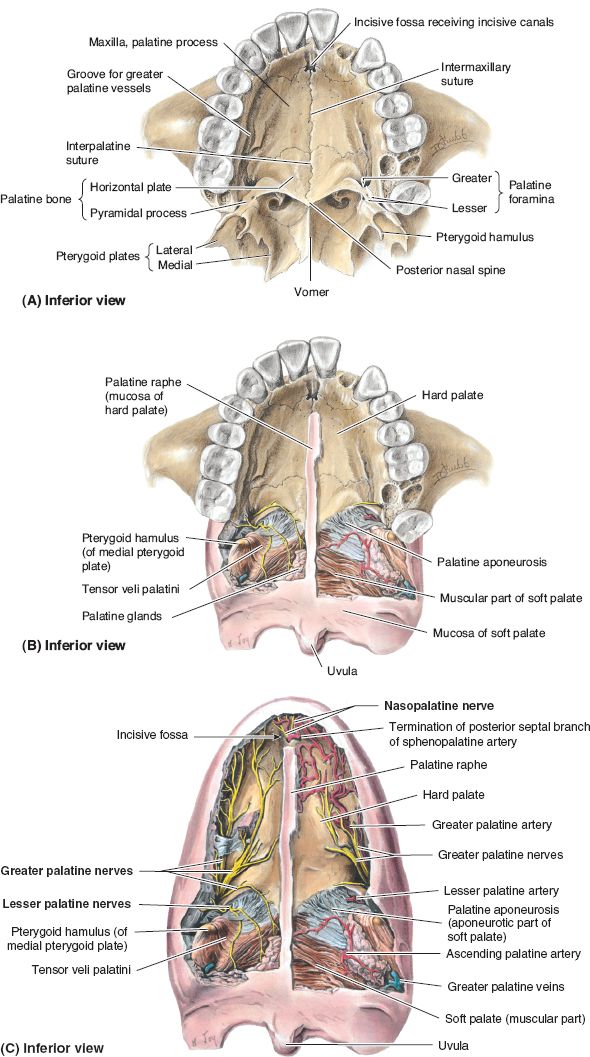
A. Hard palate (bony palate; Fig. 26-1)
1. Incisive foramen, through which passes
a. Nasopalatine nerve
b. Sphenopalatine artery
2. Primary palate
a. Anterior to incisive foramen
b. Forms with fusion of bilateral palatine process of maxilla
3. Secondary palate
a. Posterior to incisive foramen
b. Fusion of bilateral horizontal plates of palatine bone
c. Greater palatine foramen, through which passes greater palatine nerve and artery (blood supply to anterior palatoplasty flaps)
c. Isolated CP involves this region and the soft palate
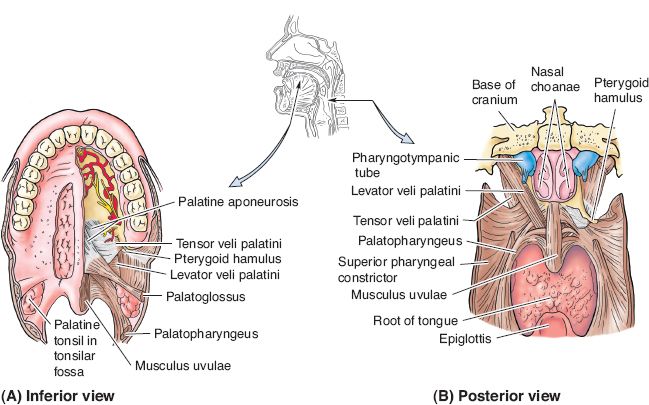
B. *Soft palate (velum; Fig. 26-2): Mucosa and muscles involved in velopharyngeal (VP) closure (with corresponding innervation)
1. Levator veli palatini (LVP, CN X)
a. Lifts velum against posterior pharynx (“genu” action)
b. Key muscle involved in VP closure
______________
*Denotes common in-service examination topics
Figure 26-1. Anatomy of the hard palate. A: Normal palatal anatomy with all soft tissue removed. B: Palatal anatomy with anterior (hard) palate soft tissue removed. C: Palatal anatomy with soft tissue intact, depicting neurovascular supply to palatal muscles and to mucosa. (From Moore KL, Dalley AF, Agur AM, eds. Clinically Oriented Anatomy. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010.)
Figure 26-2. Normal palate muscle anatomy. Views of the palatal muscles and their interaction with the pharyngeal constrictors as shown from (A) inferior and (B) posterior views. (From Moore KL, Dalley AF, Agur AM, eds. Clinically Oriented Anatomy. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010.)
c. Normally oriented transversely across velum to decussate with contralateral muscle in midline, but in CP it is oriented longitudinally; aberrant/anomalous insertion
2. Tensor veli palatini (CN V)
a. Travels around hook or hamulus (sphenoid bone)
b. Tendon often divided in repair to reduce muscle closure (intravelar veloplasty [IVV]) tension
c. Function: Open eustachian tube
3. Palatoglossus (CN X)
a. Originates from the tongue, passes through anterior tonsillar pillar, and inserts on anterior velum
b. Function: Palate depression
c. Used in dynamic sphincter pharyngoplasty (DSP)
4. Palatopharyngeus (CN X)
a. Originates from posterior pharynx, passes through posterior tonsillar pillar, and inserts on velum
b. Function: Palate depression
c. Used for dynamic sphincter pharyngoplasty
5. Musculus uvulae (CN X)
a. Originates from behind the levator to the tip of uvula
b. Function: Uvula shortening and elevation
6. Superior pharyngeal constrictor (CN X)
a. Broad muscle coarses anteriorly within the pharyngeal wall
b. Insertion: Velum
c. Function: Medial movement of lateral pharyngeal wall
C. Vascular supply
1. *Greater palatine arteries are primary blood supply for palatal mucosa
a. Pedicle for most palatoplasty flaps
b. Located medial to maxillary tuberosity in hard palate
2. Lesser palatine arteries: Supplies soft palate
3. Sphenopalatine artery
4. Ascending pharyngeal artery off of external carotid and ascending palatine branch of facial artery
1. Hard palate: Greater palatine (CN V) and nasopalatine nerves (CN V)
2. Soft palate: Lesser palatine nerve (CN V)
III. CP ANATOMY AND CLASSIFICATION
A. Variable severity
1. Bifid uvula only
2. *Submucous cleft triad: Intact mucosa with aberrant musculature
a. Bifid uvula
b. Hard palate notch (palpable on exam)
c. Zona pellucida: Pale midline mucosa
3. Cleft velum only (soft palate, Veau type I)
4. Cleft of velum and bony palate (soft and hard palate, Veau type II)
5. Primary palate clefts (a component of cleft lip and cleft palate)
a. Lip, nostril sill, alveolus, and primary palate
b. May progress to secondary palate posteriorly
i. If unilateral complete, Veau III
ii. If bilateral complete, Veau IV
6. Secondary palatal cleft
B. Anomalous insertion of tensor and LVP
1. Eustachian tube dysfunction and decreased middle ear drainage
a. Recurrent otitis media leading to hearing loss
b. Myringotomy tubes
i. Placed in 95% of CP patients
ii. Often at time of CP repair
2. Instead of decussating in midline, LVP muscles oriented anteroposteriorly, inserting onto posterior margin of hard palate
IV. PALATAL EMBRYOLOGY
A. Primary palate (fifth week)
1. Lip, alveolus, nostril sill, and hard palate anterior to incisive foramen
2. Medial and lateral nasal prominences of frontonasal process migrate and fuse to form median palatine process
3. Median palatine process forms from fusion of bilateral median nasal prominences and becomes premaxilla
B. Secondary palate (5th through 12th weeks)
1. Posterior to incisive foramen and soft palate
2. Bilateral palatine processes develop from medial maxillary process
3. Lateral palatine processes hang vertically then lift horizontally
a. Right lateral palatal process becomes horizontal before left, which may explain higher incidence of left-sided clefts
b. Fusion takes 1 week longer in females, which may explain increased incidence in females
4. Fusion starts at incisive foramen and moves posteriorly. When interrupted, CP results.
V. ETIOLOGY
A. Genetics
1. CP: Autosomal recessive with contributing genes
2. CLP: Polygenic, usually sporadic
B. Environment
1. Smoking: Inconclusive but many studies have implicated its role
2. Teratogens: Alcohol, isotretinoin increases the risk
3. Folate and B6 may be protective
C. Primary retrognathia. Pierre Robin sequence
1. Wide, U-shaped clefts
2. Difficulty maintaining airway
3. Lateral palatine processes unable to fuse due to glossoptosis (posterior displacement of tongue) causing CP
VI. INITIAL EVALUATION
A. Feeding and weight gain
1. Haberman or cross-cut nipple required due to poor oral suction
2. Palate repair is often performed at age 1 year, but may be carried out later if the patient is a poor operative candidate
B. Mandibular anatomy
1. Often normal
2. *Primary retrognathia (Pierre Robin sequence)
a. Prone positioning to relieve glossoptosis and airway obstruction
b. Continuous pulse oximetry and polysomnography to evaluate obstructive sleep apnea (OSA)
c. pH probe to evaluate gastroesophageal reflux
d. Nutrition consultation for failure to thrive, possible feeding tube
e. Direct laryngoscopy to determine additional airway pathology (e.g., subglottic stenosis)
f. Consider early mandibular distraction
i. May perform while intubated, if airway unstable
ii. Distraction until air leak, then subsequent extubation
g. Often delay CP repair for airway concerns until further mandibular growth
h. May require early tracheostomy (secondary strategy), after which CP repair may proceed at standard age
C. Patient examination
1. Use penlight and tongue depressor
a. Crying infant is easier to examine
b. Place child supine and upside-down on parent’s lap
2. Look for bifid uvula and palpate cleft to determine bony involvement
3. Vomer may be visible in nasopharynx in cases of bilateral CLP (Fig. 26-3A)
VII. GOALS
A. Closure of cleft
1. Separate oral and nasal cavities
2. Prevent aerophagia and reflux of oral contents into nasopharynx
B. Speech/VP competence
1. Requires competent VP mechanism
a. Contact of velum against posterior pharynx
b. “Genu” action describes physiologic motion of palate
2. Operative goals: Increased palatal length and muscle repositioning
3. Prevent maladaptive compensatory misarticulations
a. Perform repair at 1 year of age
b. Timing of speech milestone (first words)
4. May lead to OSA in some patients
C. Hearing
1. Otitis media
a. Eustachian tube dysfunction: Abnormal LVP origin impairs “milking” action, which leads to poor venting of middle ear
b. Permanent impairments result with recurrent infection
2. Myringotomy performed at time of CP repair
D. Facial growth
1. *Palate repair in early childhood may adversely affect maxillary growth, but this drawback is outweighed by the improvements in speech achieved by early correction
2. Early secondary palate and delayed primary palate repair has been advocated (controversial two-stage approach)
3. *Patients may require orthognathic surgery in adolescence (Le Fort I advancement; see Chapter 29) for midface hypoplasia and class III malocclusion
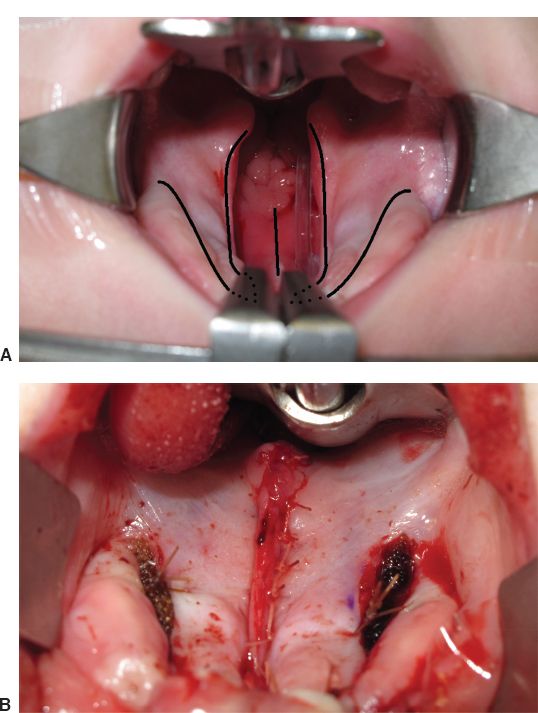
Figure 26-3. Von Langenbeck palatoplasty. A: Preoperative appearance of bilateral cleft palate, surgeons view. Incisions are planned along the cleft margins and in the vomerine mucosa (midline). Lateral relaxing incisions are also shown. The dotted lines anteriorly would be incised to convert this bipedicled technique to a unipedicled island flap (Bardach) repair. In the soft palate, a straight-line repair (IVV) is planned. B: Appearance of palate after closure of oral mucosa, muscle, and nasal mucosa. Cellulose material has been placed into the lateral defects for hemostasis. (Photos courtesy of Dr. Craig Birgfeld.)
VIII. REPAIR TECHNIQUES
A. In CLP, both the primary and secondary palates require repair
1. The entire palate must be elevated for cleft closure
2. Nasal and oral mucosa are repaired in layers over primary palate
B. In isolated CP, only the secondary palate requires repair
1. Often involves dissection in the velum only
2. In wide clefts, total palatal elevation may be required for tension-free closure
3. Nasal mucosa, muscle, and oral mucosa repaired in layers over secondary palate
C. The repair technique used in the primary palate may be chosen independently from the repair technique used in the secondary palate
D. Dingman mouth gag, neck extension with shoulder roll, Trendelenburg positioning all used to maximize exposure
E. Secondary palate repair techniques
1. Straight-line repair or intravelar veloplasty (IVV) (Fig. 26-3)
a. Useful in wide clefts
b. Three-layered closure
c. Relieve aberrant insertion of LVP from hard palate and rotate posteriorly
d. IVV: LVP dissection, disinsertion from hard palate, and muscle repair in soft palate midline
e. Adverse sequelae: Potential for short palate and subsequent VP insufficiency (VPI)
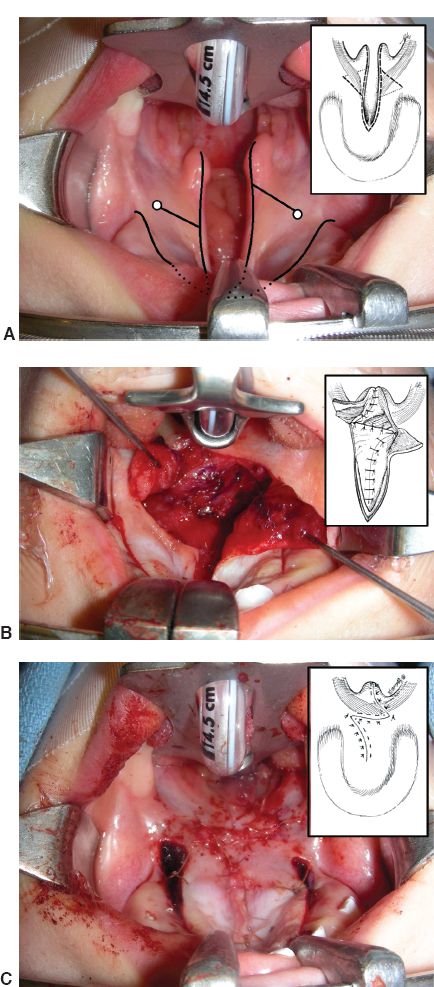
2. Furlow palatoplasty or *double-opposing Z-plasty (Fig. 26-4)
a. Opposing myomucosal Z-plasty flaps for both oral and nasal closure
i. Oral myomucosal flap is on the patient’s left
a) Conventional technique
b) Remember: Easier for right-handed surgeon to elevate the left-sided myomucosal triangle first
ii. Nasal myomucosal flap is on the patient’s right
b. Rotation of triangles will “automatically” bring LVP muscles into anatomic alignment (recall: anomalous LVP insertion)
c. Lengthens soft palate
d. Adverse sequelae: Sacrifice of width in order to gain length; may not be possible in wide clefts (consider IVV)
e. May cause sleep apnea.
F. Primary palate repair techniques
1. Von Langenbeck repair (Fig. 26-3)
a. Bipedicle mucoperiosteal flaps
i. Anterior pedicle: Blood supply via sphenopalatine a.
ii. Posterior pedicle: Blood supply via greater palatine a.
b. Parallel incisions are made along cleft margin lingual to alveolus
c. Lateral relaxing incisions left open to heal secondarily
d. Nasal and oral mucosal flaps are mobilized to midline and sutured in separate layers
e. Adverse sequelae: Potential for maxillary growth restriction, high tension repair, which may lead to dehiscence
2. V-Y pushback (Veau–Wardill–Kilner)
a. Bilateral pedicled posteriorly
b. V-Y advancement posteriorly
c. Lengthens palate for speech
d. Adverse sequelae: Potential for maxillary growth restriction
3. Two-flap palatoplasty (Bardach repair, Fig. 26-4)
a. Requires elevation of entire palatal mucosa: Island flap based on greater palatine a.
b. May be used in wide cases of isolated CP to gain extra mucosa for closure
c. Most common repair in CLP
d. Adverse sequelae: Anterior areas heal secondarily causing maxillary growth restriction
G. Additional maneuvers for wide clefts
1. Infracture of the hamulus can bring lateral elements toward midline
2. Osteotomize medial to greater palatine foramen for increased pedicle length
3. The periosteal sheath around pedicle can be released with meticulous dissection to gain additional pedicle length
IX. ADJUNCTIVE TECHNIQUES
A. Buccal fat pad flaps
1. Small incision made posterior to maxillary tuberosity
2. Fat “teased” from buccal space with gentle spreading motion
3. Can be spread to cover surprisingly large surface area
4. Mucosalize secondarily
Figure 26-4. Furlow double-opposing Z-plasty. A: Preoperative appearance of unilateral cleft palate, surgeons view. Incisions are planned along the cleft margins, with lateral relaxing incisions shown. The hamulus (white circle) has been marked bilaterally so as to design the Z-plasty incisions. The base of the right uvula is the other landmark. Recall that the left Z-plasty flap is the oral myomucosal flap. This will be easiest to elevate for the right-handed surgeon. Inset: diagrammatic representation of incisions and abnormal LVP musculature in a case with isolated CP. B: Appearance of palate after closure of the nasal layer including the right-sided myomucosal flap. The oral layer and left-sided myomucosal flap is retracted. Inset: diagrammatic representation of first Z-plasty closure. C: Final appearance of Furlow palatoplasty after closure of the second Z-plasty. Inset: Normal anatomic relationship of the LVP has been restored. (Photos courtesy of Dr. Craig Birgfeld.)
1. Based superiorly
2. Used for nasal mucosal closure anteriorly in all CLP repairs
C. Turbinate flaps
1. Can be based superiorly or inferiorly
2. Used for nasal mucosal closure of primary palate in CLP repair
D. Facial artery myomucosal flap
1. Intraoral mucosal flap based on facial a.
2. Often used to repair palatal fistulae secondarily
E. Gingivoperiosteoplasty
1. Rarely used
2. May be indicated at time of lip repair to close alveolar segments
3. Only feasible if greater and lesser segments are within ~1 mm of each other
F. Other techniques
1. Two different techniques may be used simultaneously for repair of the primary palate, one on each side
2. For example, a bipedicled Von Langenbeck (bipedicle flap) repair may be used on the lesser segment of the cleft, while a one-flap Bardach (unipedicle island flap) repair is used on the greater segment
X. ALVEOLAR CLEFT BONE GRAFTING
A. *Performed prior to eruption of permanent dentition (~7 years of age)
B. Goals
1. Enables stable support of incoming permanent dentition
2. Provides bony support to deficient cleft nasal alar base
3. Allows for closure of nasoalveolar fistula
a. Often unable to be repaired during cleft lip or cleft palate
b. Prevents nasal regurgitation
C. Requires cancellous bone graft from iliac crest
1. Incision is made parallel and below iliac crest
2. Cartilage cap over ilium is split
3. Curettes used to “scoop out” cancellous bone
D. Intraoral flap dissection
1. Incision made just above attached gingiva/papillae, up into cleft on each side
2. Nasal floor and palatal mucosal closure performed
3. A back-cut is made into unattached gingiva near last molar
4. Subperiosteal dissection along maxilla
5. Cancellous bone is packed into cleft defect
6. Periosteal cuff is then released from the underside of the flap to relieve tension as it is rotated into the cleft defect for anterior labial mucosal closure
XI. POTENTIAL COMPLICATIONS OF CP REPAIR
A. Acute airway obstruction
1. Bleeding/aspiration/laryngospasm
2. Tongue swelling; reperfusion injury from Dingman mouth gag
3. Reintubation in immediate perioperative period is ~1%
4. Place tongue stitch and possibly nasopharyngeal airway postoperatively
5. Pulse oximetry overnight
B. Dehiscence of palatal flaps, may be due to
1. Undue tension
2. Poor flap vascularity
3. Inadequate or overzealous suturing
C. Palatal fistula
1. Reported from 5% to 50%
2. Most common in cases of wide bilateral CLP
3. *Hard/soft palate junction is the most common location
4. Single-layer closure may be risk factor
D. Midfacial growth restriction
1. Intrinsic midface growth problems are present in children with CP or CLP
2. Scarring/secondary healing from palate repair exacerbates maxillary growth restriction
3. May be reduced by avoiding secondary intention healing
a. Limit undermining when possible
b. Use buccal fat pad flaps to close lateral open areas
4. Timing of palate repair: As late as possible to allow maximal growth, but before the emergence of speech
F. Hyponasality: Less common than VPI; related to overaggressive closure of velopharynx
G. VPI
1. Incomplete closure of velum
a. Air escape through nasopharynx
b. Hypernasal speech
2. 20% incidence following palatoplasty
3. Due to inadequate palatal length and/or poor muscle function
4. Patient develops maladaptive compensatory substitutions of abnormal for normal sounds in order to be understood, especially
a. Pharyngeal fricatives
b. Glottal stops
5. Treatment
a. Obturator (prosthesis) to fill areas of tissue deficit
b. Posterior pharyngeal flap (PPF)
i. Static, nonphysiologic technique
ii. Myomucosal flap from posterior pharynx
a) Mucosa and superior pharyngeal constrictor m.
b) Superiorly based and sutured to soft palate
iii. Appears as a tissue “bridge” with two lateral ports
iv. Requires the patient to have movement of lateral walls
c. DSP
i. Dynamic technique
ii. *Superiorly based myomucosal flaps from posterior tonsillar pillar flaps (palatopharyngeus m.)
iii. Crossed and overlapped (to variable degrees) in midline
iv. Indicated with absent or minimal medial excursion of lateral walls
v. Appears as a single port
d. Posterior pharyngeal fat grafting (PPFG)
i. Hand-assisted liposuction
ii. Injection into submucosa of postpharyngeal wall to narrow distance to velum
H. OSA
1. Increasingly diagnosed in CP population
2. More likely following secondary speech operations: PPF, DSP, and PPFG
QUESTIONS YOU WILL BE ASKED
1. What is the effect of palatoplasty on maxillary growth?
Causes maxillary growth restriction
2. What is the most common location for a palatal fistula?
At the junction of hard and soft palate
3. What is the blood supply to the hard and soft palate?
Hard palate: Greater palatine artery. Soft palate: Lesser palatine artery, ascending pharyngeal artery, and ascending palatine branch of facial artery
THINGS TO DRAW
Draw out a furlow palatoplasty.
See Fig. 26-4
Recommended Readings
Fisher DM, Sommerlad BC. Cleft lip, cleft palate, and velopharyngeal insufficiency. Plast Reconstr Surg. 2011;128(4):342e-360e. PMID: 21921748.
Furlow LT Jr. Cleft palate repair by double opposing Z-plasty. Plast Reconstr Surg. 1986;78(6):724-738. PMID: 3786527.
Liau JY, Sadove AM, van Aalst JA. An evidence-based approach to cleft palate repair. Plast Reconstr Surg. 2010;126(6):2216-2221. PMID: 21124164.
< div class='tao-gold-member'>