CHAPTER 42 Interventional Timing
KEY POINTS
Although there is no cure for lymphedema, early treatment improves outcomes.
Some patients respond to conservative treatment.
Patients should be referred early for surgery if they do not respond to conservative measures.
It has been made clear throughout this book that there is currently no cure for lymphedema. Although some treatments can yield spectacular results practically amounting to a cure, such results are inconsistent and exceptional. Evidence shows that early intervention leads to better outcomes. 1 For this reason, it is important to develop a reasoned approach regarding which treatments to offer our patients, and when. The sooner a diagnosis can be made, it is more likely that we can develop a strategy and treatment plan before the condition becomes indolent and chronic. We also know that before lymphedema becomes clinically recognizable, there is a preclinical stage in which even the patient is unaware of the looming problem. Clearly, early detection is advantageous. Despite controversies in the literature, there is good evidence indicating that bioimpedance analysis (see Chapter 24) can detect lymphedema in this preclinical stage. 2 It is possible that more widespread use of this technology as a screening tool may lead to earlier detection and consequently potentially better outcomes. Currently bioimpedance is considered experimental in many countries. The detection of preclinical lymphedema may also be possible with indocyanine green (ICG) lymphangiography. 3 However, although it makes sense intuitively, much work must be done to confirm the link between early detection, early treatment, and better outcomes. This is particularly important in an era of constrained resources and greater accountability in health care. It is also essential to remember that some patients with postoperative lymphedema improve on their own, and invasive treatment should be avoided in this group of patients, although the use of conservative therapy at this stage reduces the risk of chronic lymphedema. 4
However, should we look at our patients even before they undergo cancer surgery and try to determine which patients are most at risk of developing lymphedema? True, some individuals may develop lymphedema after sentinel node biopsy, whereas others do not, even after lymph node dissection and radiotherapy. There is some evidence in the literature that predicting which patients are at risk is not only possible but useful. 5 One proposed approach to preventing the problem is to identify the draining lymph nodes of the arm or leg and preserve them during lymphadenectomy. This approach involves the concept of reverse lymph mapping and has been shown to be feasible in the upper 6 and lower 7 extremity. Some surgeons argue that we should treat patients prophylactically with lymphaticovenular anastomosis (LVA) at the time of lymph node dissection, and there is some proof to confirm that this may work. 8 However, because the incidence of lymphedema after lymph node dissection is nowhere near 100%, such an approach means that we would be treating a large proportion of patients unnecessarily. Furthermore, the associated costs of such treatment (for example, resource use, intensity, and demand) make it untenable.
It may be more reasonable to adopt such an approach in patients recognized as being at high risk of developing lymphedema. How do we identify this group of patients, and what treatment options do we offer them? The lymphatic microsurgical preventive healing approach (LYMPHA) procedure has been proposed as one such method in which the draining lymph nodes of the upper extremity are identified, and when removed, the afferent lymphatic channels to those nodes are anastomosed to a branch of the axillary vein (see Chapter 40). 9 Although this is an attractive proposal, its utility is limited by the general lack of availability of such expertise and resources.
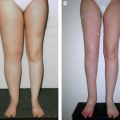
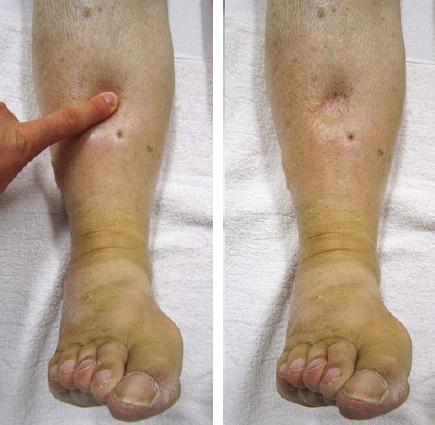
Excluding prophylactic treatment, what other early treatment options can we offer our patients who have either preclinical or early stage clinical lymphedema? Conservative treatment is the first line of active therapy, and it is reasonable to suggest that every patient with early stage lymphedema should be offered a trial of conservative decongestant management. This is especially true, because lymphedema will resolve in a proportion of such patients. The big question is, when should we intervene with more radical treatment? Obviously, the patient who has tried and failed conservative treatment will be a potential candidate for surgical management, but how long do we wait to see if conservative treatment works? Furthermore, there is clear evidence that certain treatments are best managed at a particular stage of the disease. For example, liposuction (see Chapter 32) is indicated only after the chronic changes of fibrosis and increased fat deposition have become established. If the patient continues to have pitting edema (Fig. 42-1), indicating that there is a significant fluid element to the swelling, liposuction is not indicated. Conversely, we know that LVA is best performed before the stages of fibrosis and increased fat deposition, while there are still functioning lymphatic channels and a reasonable chance that LVA will work. We also know that the collecting lymphatic channels undergo a spectrum of changes ranging from normal through the ectatic, contracting, and sclerosing types and that these changes are indicative of the likely success of LVA. 10 Akita et al 11 conducted a prospective cohort observational study of 100 consecutive patients with gynecologic cancer who underwent pelvic lymph node dissection with ICG lymphangiography to assess early lymphatic flow patterns and risk factors. Of the patients who developed transient dermal backflow, those patients with a splash pattern of distribution tended to improve, whereas those with a stardust pattern did not. The authors concluded that perhaps these patients should be targeted for early treatment. 11
For LVA to be successful, we must have functioning lymphatic channels. What do we offer the patient who does not have functioning lymphatic channels? Generally speaking, these are patients with established stage II or III lymphedema. Should we just offer them excisional procedures, including liposuction? We know that liposuction helps with limb volume reduction in these patients, even if they are still destined for lifelong compression. 12 Vascularized lymph node transfer (VLNT) is another potential treatment. Can we reasonably expect transplanted lymph nodes to drain a limb in which there is no evidence of functioning lymphatic channels?
There is controversy in the literature regarding where best to place the lymph nodes. Should they be placed distally where the edema is maximal 13 or proximally, as proposed by Dr. Becker (see Chapter 36)? Cheng et al 14 performed some elegant studies to show that lymph nodes act like sump pumps, and there is rapid transit of fluid from a flap containing lymph nodes into the draining vein of that flap. Regardless, VLNT is increasingly being used. However, a recent comprehensive review of the literature concluded that although the results with the use of VLNT for the treatment of lymphedema have largely been positive, further exploration into standardized protocols for the diagnosis, treatment optimization, and patient outcomes assessment is needed. 15 With VLNT, there is much diversity in practice not just regarding placement of the lymph nodes but also whether to include a lymphaticolymphatic anastomosis, anastomosing an efferent lymphatic from the limb to an afferent lymphatic to the lymph node. Is that enough? How does the lymph drain from there? Does it drain to the bloodstream, or does it continue to drain to the main lymphatic pathways, and if so, how does it do that? Thus there are many unanswered questions, and our current recommendations may change as our knowledge increases.
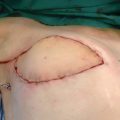
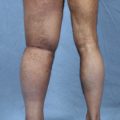
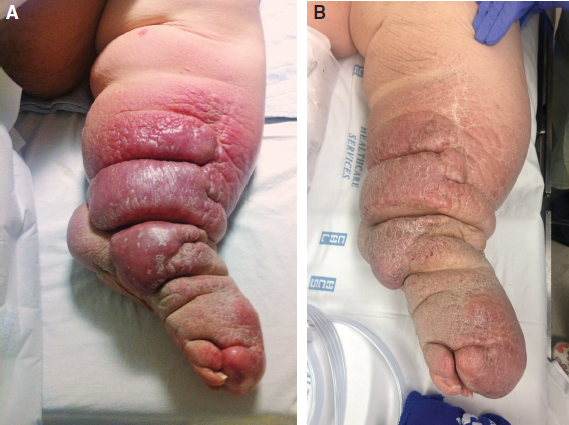
Our approach to patients with lymphedema is similar. Every patient who presents needs a diagnosis with many different methods of evaluation as already outlined in this book. It is also important not to rely on any one modality. The patient in Fig. 42-2 has long-standing lymphedema with a history of many episodes of infection and trophic skin changes. Although it appears that she would not be a candidate for a bypass procedure, we found functioning lymphatics with ICG, performed several LVAs, and had a surprisingly positive outcome. Although we did not cure her lymphedema, she lost considerable limb volume, had a much lighter leg, significantly reduced her incidence of cellulitis, and overall was considerably improved. At the time we performed the LVA, she also had a direct excision of a festoon of lymphedematous tissue in her proximal thigh.
We have already mentioned that patient history is of paramount importance. It is important to differentiate between primary and secondary lymphedema, postsurgical or postradiation lymphedema (or both), duration of the edema, previous treatments, and the patient’s response to previous treatments. This information can be gleaned from the history. The physical examination is also important and may guide the practitioner to the most appropriate treatment.
In our practices we use imaging and consider it an important part of the evaluation, either with magnetic resonance lymphangiography (MRL) or ICG or both. The advantage of MRL is that it provides excellent information not only about the superficial lymphatics but also about the deep system. Furthermore, it helps to visualize where the edema is maximal and the state of fat deposition in the limb. Accurate volume measurements can also be gained from the MRL. The problem of identifying which is a lymphatic structure and which is a venous one has largely been eliminated. 16 The downside of the technique is that it is time-consuming, technically demanding for the radiologist, and uncomfortable for the patient.
Fluorescent lymphangiography with ICG is an extremely useful tool and is used extensively in lymphatic surgery. In fact, it is difficult to perform lymphatic surgery without ICG. In our practices it is used extensively both in diagnosis and treatment. The advantages of ICG are several. It is an easy technique and involves injection of ICG into the web spaces of the fingers or toes, excitation by a laser light source, and then examination with a nearinfrared camera. When ICG is injected subcutaneously it is taken up by the lymphatics, making it a very useful tool for mapping subdermal lymphatic vessels.
The one situation in which we proceed to schedule surgery is in patients with lymphedema who have a history of node dissection and radiotherapy. These are mostly postmastectomy patients. Some of these patients want to have their lymphedema treated and their breast reconstructed. In those patients we perform a deep inferior epigastric perforator flap in combination with lateral inguinal VLNT. Alternatively, in patients who do not want any reconstruction, we perform radical scar excision and VLNT, either from the inguinal region or from elsewhere, as described in Dr. Becker’s chapter on VLNT (see Chapter 36).
Conclusion
Numerous options are available for both conservative treatment (see Chapter 29) and surgical treatment (see Chapters 30 through 37). There is good evidence that early intervention yields the best results. It is equally important to choose the correct modality of treatment. This choice is facilitated by a thorough history and preoperative imaging.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree