CHAPTER 33 Multiple Lymphaticovenous Anastomoses and Multiple Lymphatic-Venous-Lymphatic Anastomoses
KEY POINTS
Microsurgical derivative and reconstructive lymphaticovenous anastomoses (multiple lymphaticovenous anastomoses and multiple lymphaticvenouslymphatic anastomoses) can provide an effective functional repair of the lymphatic pathway by diverting the lymph into adjacent veins proximal to the source of the lymphatic obstruction.
Lymphatic microsurgery is appropriate for both primary and secondary obstructive lymphedemas and as a preventive approach during other general surgeries in which there is the risk of disrupting the lymphatic pathways.
Lymphatic microsurgery is most effective when it is performed early in the disease course and in conjunction with a combined treatment protocol such as complete lymphedema functional therapy (CLyFT).
Advanced stages of lymphedema can be appropriately treated with multiple lymphaticovenous anastomoses or multiple lymphaticvenouslymphatic anastomoses to relieve lymph stasis but may require a complementary procedure to remove residual excess fibrous and adipose tissue contributing to poor lymphatic drainage.
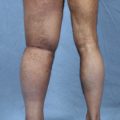
Chronic lymphedema is a progressive and relatively painless swelling of any peripheral tissue, including the limbs, head and neck, breast, trunk, or genitals; it results from a reduced transport capacity of the lymphatic system. Chronic lymphedema can be classified as primary or secondary, depending on the cause. In patients with secondary lymphedema, a specific external cause (filariasis, previous surgery, radiation, malignancy, infection or inflammation, or trauma) can be identified and is believed to impact on a presumed previously normal functioning lymphatic system by causing an obstruction to the lymphatic flow.
Most of the clinical conditions that are considered primary lymphedema are the result of truncal lymphatic malformations that develop during the final stages of lymphovasculogenesis 1 , 2 when the lymphatic trunks, vessels, and nodes are formed. 3 , 4 These malformations result in hypoplasia, hyperplasia, or aplasia of the lymphatic vessels and/or lymph nodes and may clinically manifest as an obstruction or dilation.
BOX 33-1 Staging of Lymphedema Based on Immunohistologic Criteria, Lymphoscintigraphic Findings, Clinical Symptoms, and Degree of Physical Disability
Stage 1
A Latent lymphedema without clinical evidence of edema but with impaired lymph transport capacity (provable by lymphoscintigraphy) and initial immunohistochemical alterations of lymph nodes, lymph vessels, and extracellular matrix
B Initial lymphedema, totally or partially decreasing by rest and draining position, with worsening impairment of lymph transport capacity and immunohistochemical alterations of lymph collectors, nodes, and extracellular matrix
Stage 2
A Increasing lymphedema with vanishing lymph transport capacity, relapsing lymphangitic attacks, fibro-indurative skin changes, and developing disability
B Column-shaped limb fibrolymphedema with lymphostatic skin changes, suppressed lymph transport capacity, and worsening disability
Stage 3
A Properly called elephantiasis, with scleroindurative pachydermatis, papillomatous lymphostatic verrucosis, and no lymph transport
B Extreme elephantiasis with total disability
To provide a comprehensive classification system of lymphedema that encompasses the immunohistopathologic criteria, level of clinically evident edema, lymphoscintigraphic findings, and level of physical disability, we developed a three-stage model 5 , 6 (Box 33-1). In clinical practice, stages 1A, 1B, 2A, and 2B can be considered early manifestations of disease and stages 3A and 3B (usually known as elephantiasis) are chronic and advanced stages. Lymphedema is a progressive disease and can move rapidly between the stages without adequate treatment. 7 – 10
Preoperative Considerations
DIAGNOSIS
The diagnosis of lymphedema and other lymphatic disorders should be performed by clinicians and lymphologists experienced in this area. The essential diagnostic criterion is evidence of slow, reduced, or completely absent lymphatic flow in the tissue affected by the swelling. Lymphoscintigraphy is generally considered the benchmark procedure to measure this flow, although procedures have not been standardized. In Genoa, Italy, lymphoscintigraphy, which is performed with either technetium-99m (99mTc)-labeled antimony sulfur colloid or 99mTc-nanocolloid human serum albumin (90% of the particles are greater than 80 nm), is used in the diagnostic workup before surgery to determine the eligibility for derivative multiple lymphaticovenous anastomoses (MLVA). Lymphoscintigraphy clearly indicates whether the edema has a lymphatic origin in patients and provides useful data about the etiologic factors and pathohistologic nature of the lymphedema. A transport index is calculated to categorize the lymphatic flow as normal or pathologic (Fig. 33-1).

A score of less than 10 signifies a normal transport index, and a score equal to or greater than 10 signifies a pathologic transport index. Scores are determined bilaterally, even in patients with unilateral swelling. (Other diagnostic and preoperative considerations are discussed in the Surgical Techniques section.)
Surgical Techniques
The first use of microsurgery for the treatment of lymphatic disorders was in 1962 when Cockett and Goodwin 11 performed an anastomosis of a dilated lumbar lymphatic vessel to the spermatic vein to treat a patient with chyluria. As a result of this first procedure, subsequent developments in microsurgical techniques have enabled lymphaticovenous anastomosis to emerge as a feasible and relatively commonplace treatment option for lymphedema.
In 1977 and 1979 O’Brien et al 12 , 13 described their use of microlymphatic surgery for the treatment of secondary obstructive lymphedema. They created three lymphaticovenous anastomoses at or above the elbow (or knee) and observed a reduced incidence of cellulitis after surgery. Their microlymphatic techniques were applicable to both upper and lower limbs and were also used for localized cases of obstructive lymphedema after trauma and congenital constriction bands. They emphasized that considerable experience in microvascular surgery was required to perform the procedure.
The field was further advanced in 1981 when Degni 14 , 15 introduced an original technique of lymphaticovenous anastomosis to divert the lymph into a vein in patients with blocked lymphatic vessels, particularly when lymphographic findings showed good function and intact permeability of lymphatic vessels. At almost the same time, Clodius et al 16 , 17 stated that microsurgery for primary and secondary lymphedema, which consisted of shunts between the lymphatic vessels and veins, was a well-established surgical technique. However, problems arose from irreversible changes in the peripheral lymphatic system and connective tissues and from the obliteration of the deep lymphatics best suited for lymphaticovenous anastomoses in more chronic lymphedema. Therefore Clodius cautioned that lymphaticovenous shunts should be performed early before the appearance of fibrotic tissue changes. Despite advances in microsurgery, the most suitable operation for peripheral lymphedema remains unclear.
DERIVATIVE APPROACHES
The first derivative microsurgical procedures used lymph nodal–venous shunts. These have largely been abandoned because of the high rate of anastomotic closures. 18 Lymph nodal pulp has a thrombogenic effect on the venous blood, and there is also a frequent reendothelialization of the lymph nodal surface, both of which contribute to anastomotic closure. Worldwide, surgeons have moved to the anastomosis of lymphatic vessels directly into the veins to increase the long-term efficacy of the procedures. 19 , 20 More recently, our preferred technique has been telescopic MLVA.
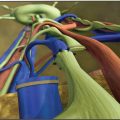
Lymphaticovenous anastomoses involve joining an appropriate lymphatic vessel to a collateral branch of a main vein and ensuring that the vein has competent valvular function and continence. This is essential to prevent reflux of the blood into the lymphatic vessel and thereby thrombosis of the anastomosis. 21 Anastomoses that are created in close proximity to a valve in the vein are able to overcome the difference in pressure between the venous and lymphatic systems. In this way the valvular pumping creates a suction that pulls the lymph immediately through the anastomosis to prevent thrombosis. The Center of Lymphatic Surgery and Microsurgery at the University of Genoa, Italy, has obtained excellent stable clinical outcomes for 40 years with the use of the MLVA and multiple lymphaticvenouslymphatic anastomoses (MLVLA) 5 , 22 , 23 (Fig. 33-2). Anastomoses are performed at a single site; larger lymphatic vessels are attached to collateral branches of the main veins close to vein valves to avoid backflow of blood and the closure of the anastomosis. A single-site approach also minimizes the number of incisions and thereby potential entry sites for infection.

Blue patent violet (BPV) dye, a sodium or calcium salt of diethyl ammonium hydroxide, is used intraoperatively to identify properly functioning lymphatic vessels at the surgical incision. Healthy appearing lymphatics found at the surgical site are directly introduced into the vein by a U-shaped stitch and then fixed to the vein cut end by means of additional stitches between the vein border and perilymphatic adipose tissue. We create the MLVA by using lymphatic vessels with the perilymphatic tissue intact. This ensures that these vessels are functioning and also helps to prevent thrombosis. At the end of the procedure, the first U-shaped stitch is removed to avoid the risk of closure of the lymphatic collectors. With the use of BPV dye, properly functioning lymphatics appear blue, and the passage of blue lymph into the vein branch verifies the patency of the lymphaticovenous anastomosis under the operating microscope when the anastomosis is completed (Fig. 33-3).
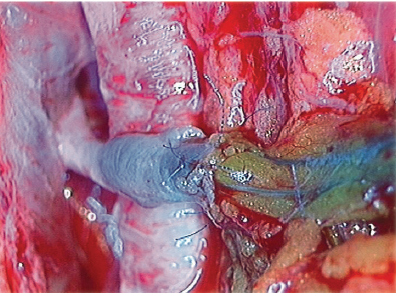
For lower limb lymphedema, the MLVAs are created at the site of a single incision made at the subinguinal region. For upper limb lymphedema, MLVAs are created in the middle third of the volar surface of the arm. Both surface and deep afferent lymphatic collectors that are visualized by the BPV dye are used. The deep lymphatics are generally located between the humeral artery, vein, and median nerve and introduced by a telescopic technique into a patent branch of one of the humeral veins containing a functioning valve.
MULTIPLE LYMPHATICOVENOUS ANASTOMOSES
It is possible to perform multiple lymphaticovenous anastomoses at the same time; the most important requirement is that the vein branch be patent. Duplex scanning is performed in all patients to identify any venous disorders that may contribute to the edema. In most patients, it is possible to correct these venous dysfunctions at the same time as the microsurgery. This is done by performing procedures, such as valvuloplasty, for venous insufficiency with 6-0 nylon sutures. In a few cases, venous disease is an indication for performing a venous graft between the lymphatic collectors above and below the site of lymphatic obstruction, because the valvular incompetence of the diseased veins compromises the efficacy of the anastomoses if MLVAs were performed instead. This venous bridge type of graft is called a lymphaticvenouslymphatic anastomosis. Competent venous segments are harvested from the same operative site or can be taken from the forearm (usually the cephalic vein). The length of the grafted vein section varies from 7 to 15 cm, as needed. It is essential to gather several lymphatic collectors to join to the distal end of the vein to ensure that the vein segment is filled with sufficient lymph to avoid closure from subsequent fibrosis. The competent valves of the vein segment are vital to direct the flow of lymph in the correct direction and to avoid gravitational backflow or reflux. As with the lymphaticovenous anastomosis, the lymphatic collectors are directly introduced into the vein cut ends by means of a U-shaped stitch, which is then stabilized with additional peripheral stitches and ultimately removed as described previously.
Clinical Experience
Between 1973 and February 2014, 3007 patients with upper and lower limb lymphedema and primary and secondary disease received microsurgical treatment for peripheral lymphedema in Genoa, Italy. Patients were followed for a minimum of 5 years, and our maximum follow-up was 25 years after surgery. Our routine follow-up consisted of periodic clinical evaluations at 1, 3, 6, and 12 months after microsurgery and then annually for a minimum of 5 years. This close patient follow-up provided optimal control for the long-term clinical outcome.
Histologic samples taken during surgery showed that primary lymphedemas typically involved lymph nodal dysplasias (leukocyte adhesion deficiency II according to Papendieck’s classifications 24 ) with hypoplastic lymph nodes associated with sinus histiocytosis and a thick fibrous capsule and microlymphangioadenomyomatosis. These dysplasias caused an obstruction to lymphatic flow that was evident in the changes in the afferent lymphatic collectors. These were dilated, swollen, and tortuous, with thickened walls and reduced numbers of smooth muscle cells, which were fragmented by numerous fibrotic elements.
In our clinical experience, secondary lymphedemas are usually related to lymphadenectomy and radiotherapy as part of oncologic treatment (carcinoma of the breast, uterus, penis, bladder, prostate gland, rectum, and seminoma of the epididymis) or to complications from minor surgeries for varicose veins, crural and inguinal hernias, lipomas, tendinous cysts, or inguinal and axillary lymph nodal biopsies. Most of the lymphedemas treated were at stages 2A (38%) and 2B (53%), with 3.5% at stage 1B and 5.5% at stages 3A and 3B, according to the Campisi staging system for lymphedema 25 (see Box 33-1).
Objective clinical measures of lymphedema consisted of limb water volumetry, circumferences, and lymphoscintigraphy. Lymphoscintigraphy, which was performed with either 99mTc-labeled antimony sulfur colloid or 99mTc-nanocolloid human serum albumin (90% of the particles were greater than 80 nm), was used in the diagnostic workup before surgery to determine the eligibility for derivative MLVA. Lymphoscintigraphy clearly indicated whether the edema had a lymphatic origin in patients and provided useful data about the cause and pathohistologic nature of the lymphedema. Lymphoscintigraphy was also used to verify the patency of the microanastomoses in the long term by direct and indirect methods (Fig. 33-4). These included the following:
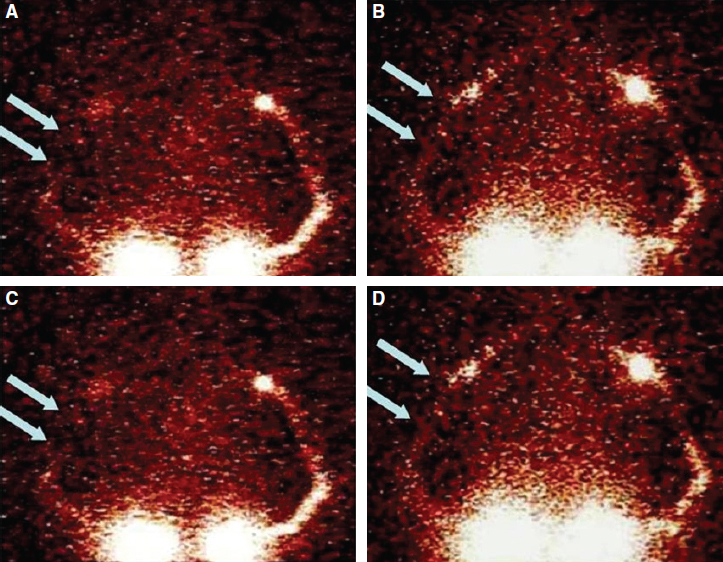
Reduced dermal backflow of the tracer and the appearance of preferential lymphatic pathways not evident preoperatively
The disappearance of the tracer at the site of the lymphaticovenous anastomoses, indicating the passage of lymph into the bloodstream
Earlier liver uptake of tracer compared with preoperative parameters, indicating indirect evidence of the passage of lymph in the bloodstream
Most recently, the evaluation of the superficial lymphatic pathways distal to the surgical site in the affected limb has also been performed with the photodynamic eye (PDE) method of injection of indocyanine green fluorescence. Simultaneous use of PDE with BPV provides two methods of visualizing the lymphatic flow and confirming the patency of the anastomoses intraoperatively. 26 , 27 PDE can be used after surgery, in addition to lymphoscintigrapy, to verify the long-term stability of the surgical outcomes.
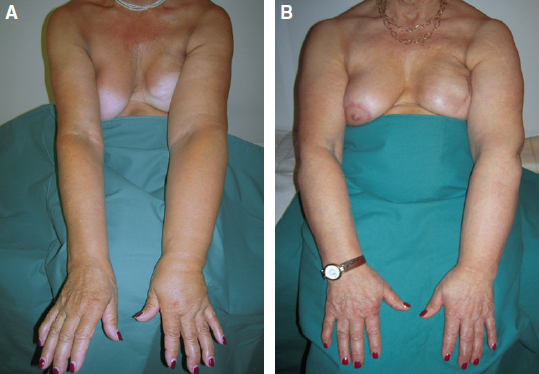
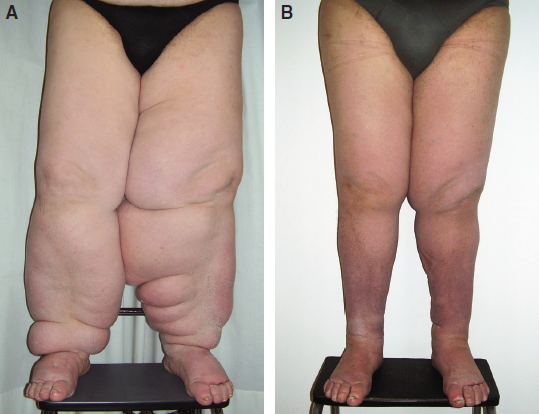
In our series, compared with preoperative conditions, all patients obtained significant reductions in excess limb volume of more than 85%, with an average of 70% as measured by limb water volumetry and circumference. These results were stable over an average of 10 years of followup. Outcomes improved with earlier microsurgical intervention because of no or minimal fibrosclerotic tissue changes in the lymph vessel walls and surrounding tissues. More than 85% of patients with earlier stages of disease (stages 1 or 2A and 2B) progressively stopped using conservative therapies over the follow-up period (Fig. 33-5). Forty-three percent of patients with more advanced lymphedema (stages 3A and 3B) decreased the frequency of physical therapy in the long term (Fig. 33-6). Infective rates, particularly lymphangitis, cellulitis, and erysipelas, dropped by 91% compared with preoperative conditions.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree