CHAPTER 37 Reverse Lymphatic Mapping
KEY POINTS
The risk of donor-site lymphedema is relevant to the harvest of groin, axillary, and supraclavicular lymph nodes.
Reverse lymphatic mapping enables the surgeon to identify lymph nodes draining the extremity so that they can be avoided during lymph node transfer.
Anatomic landmarks do not always correlate with physiologic lymphatic drainage patterns, which can be identified with reverse lymphatic mapping.
Reverse lymphatic mapping provides physiologic information that can alter the surgical plan and improve the safety of lymph node transfer.
Vascularized lymph node transfer (VLNT) has become increasingly popular for the treatment of lymphedema, because it is conceptually appealing and uses techniques familiar to every microsurgeon. Although VLNT has been effective in the treatment of lymphedema, the potential for creating lymphedema at the donor site is the primary concern of both the surgeon and patient. Reports of donor-site lymphedema have surfaced in the literature, even when surgery was performed by very experienced microsurgeons with the use of traditionally accepted anatomic landmarks. 1 – 5 This risk is a real one, and at the same time, it would be premature to dismiss VLNT entirely, because it has yielded significant improvement in numerous patients who had previously coped with a disabling and unrelenting disease. 6 – 8 The question now becomes, how can we perform this technique while minimizing the potential risk of donor-site lymphedema?
We developed reverse lymphatic mapping for VLNT to specifically address the risk of donor-site lymphedema. We have used reverse lymphatic mapping to harvest lymph nodes from the inguinal, axillary, and supraclavicular regions to avoid harvesting lymph nodes that drain the adjacent limb.
Concept
Reverse mapping was first conceived by Thompson et al 9 and Klimberg 10 to reduce the risk of iatrogenic lymphedema after sentinel lymph node biopsy or axillary lymphadenectomy for breast cancer treatment. This technique, described as axillary reverse mapping, involves the injection of blue dye in the upper extremity and technetium in the breast. Avoiding blue lymph nodes that drain the upper limb during sentinel lymph node biopsy or axillary lymphadenectomy (unless they share drainage with the breast) potentially reduces the risk of developing lymphedema.
We 11 have applied this concept and modified it for VLNT to minimize the risk of donor-site lymphedema. Technetium is injected into the hand or foot, depending on where the donor site of the lymph node flap is located. A gamma probe is used intraoperatively to locate the extremity-draining lymph nodes, which are then avoided. Indocyanine green is also injected into the trunk, and with nearinfrared imaging, the target lymph nodes draining the trunk can be identified and included in the flap. This approach provides valuable physiologic information that can alter flap harvest, as opposed to solely using anatomic landmarks as a guide. We have observed several aberrant lymphatic drainage patterns with reverse lymphatic mapping. By using reverse lymphatic mapping, we have identified and avoided lymph nodes draining the leg that typically would have been harvested if anatomic landmarks alone were used, thus potentially reducing the risk of iatrogenic lymphedema.
One limitation that we encountered in our early experience with blue dye injection was that the lymph nodes could only be seen when directly exposed in the operative field. At this point, the delicate lymphatic network and vascular supply may be jeopardized, and harvesting compromised lymph nodes may lead to a failed lymph node transfer. The use of indocyanine green lymphangiography with a nearinfrared detector allowed us to see the lymph nodes before they were directly exposed in the field, which enabled the preservation of the lymph nodes in the flap with maximum viability.
Reverse Lymphatic Mapping in Vascularized Groin Lymph Node Transfer
ANATOMY
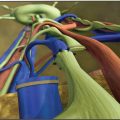
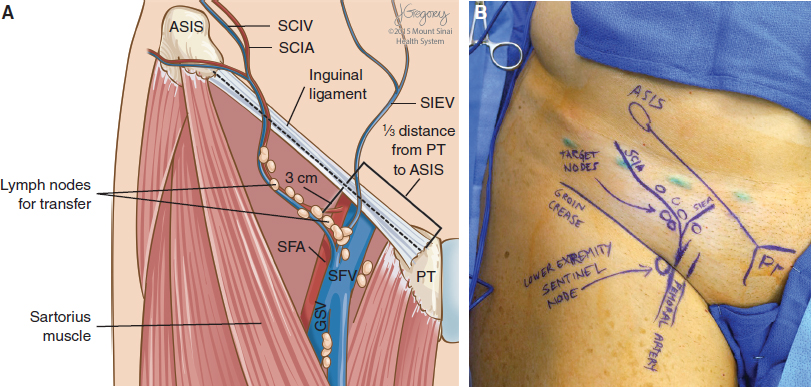
The superficial inguinal lymph nodes are typically harvested based on the superficial circumflex iliac artery (SCIA). This artery commonly arises from the common femoral artery in the femoral triangle, although it may sometimes have an anomalous takeoff from the deep circumflex iliac artery (DCIA). The venous drainage of this flap can be through the venae comitantes or an alternative superficial vein.
Based on our previous anatomic study, 12 the SCIA-based lymph nodes are concentrated at the junction of the superficial circumflex iliac and superficial inferior epigastric veins. Anatomically, this corresponds to the region between the inguinal ligament and groin crease, which is approximately one third the distance from the pubic tubercle toward the anterior superior iliac spine (Fig. 37-1). The superficial lymph nodes lie just deep to Scarpa’s fascia. Although these nodes predominantly drain the lower abdomen, on occasion there can be SCIA-based lymph nodes that significantly contribute to lower extremity drainage, and these can be identified with reverse lymphatic mapping. Lymph nodes below the groin crease consistently drain the lower limb and should be avoided.
SURGICAL TECHNIQUE
Approximately 1 hour before surgery, the patient is injected with 0.2 ml of filtered technetium sulfur colloid (0.2 mCi/0.2 ml) in the first and second web spaces of the foot on the same side as the planned donor site. After the patient is prepared and draped, 0.1 ml of indocyanine green is injected intradermally across the lower abdomen in four areas halfway between the region of the umbilicus and inguinal ligament. The area is massaged to facilitate flow of the dye into the lymphatics and inguinal lymph node basin (Fig. 37-2).
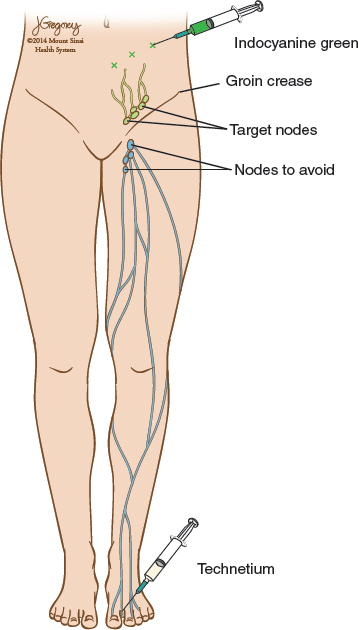
At this point, the inguinal crease is marked, as well as the course of the inguinal ligament between the pubic tubercle and anterior superior iliac spine. A handheld Doppler is used to confirm the course of the SCIA and superficial inferior epigastric artery. The gamma probe is now used to locate the sentinel lymph node draining the lower limb, which typically lies along the femoral vessels slightly below the inguinal crease. There will always be a signal in the region of the flap before dissection, because the probe will pick up the technetium in the deeper iliac lymph nodes. Thus this should not be a cause for concern. Near-infrared imaging is now used to visualize the lower abdominal lymphatic vessels and the corresponding lymph nodes targeted for harvest. Because the lymph nodes are deeper, these nodes will appear as a faint blush but will become obvious once the flap elevation begins.
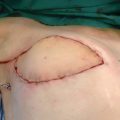
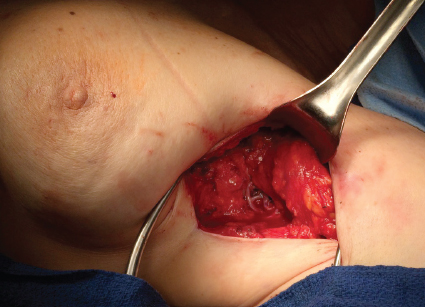
Most commonly, we harvest a lymph node flap without a skin island from the groin for use in the treatment of breast cancer–related lymphedema. The skin is incised over the SCIA, and subcutaneous flaps are elevated superficial to Scarpa’s fascia, which exposes the superficial surface of the lymph node flap. The nearinfrared imaging is used once again to confirm the location of the target nodes. The flap is then elevated from lateral to medial, typically superficial to the sartorius muscle fascia. There can be variations in the origin and course of the SCIA. Therefore preoperative imaging may be beneficial. During lateral-to-medial flap elevation, the gamma probe is placed on the undersurface of the flap and aimed toward the ceiling to avoid signal artifact from the lower limb or deeper iliac nodes. This is a critical step in confirming the lack of signal in the lymph nodes based on the SCIA. If there is a high signal present, the decision is typically made to abort the flap and either to stop or switch to an alternate donor site. We secure consent from all patients for an alternative donor site if such a scenario occurs. On two occasions to date we have found significant technetium-99 uptake from the lower extremity by an SCIA-based lymph node, which resulted in a modification of the procedure or the use of an alternate site. The patient should be informed before surgery regarding the potential need for an alternate approach if this situation arises.
After the pedicle is identified, the next decision is to determine the medial border of the flap. This is an important point in the case, because there are medial lymph nodes in the same dissection plane that do significantly contribute to lower extremity drainage and are typically medial to the superficial inferior epigastric vein. However, as mentioned previously, static landmarks can be misleading. Thus the gamma probe is used again to interrogate the more medial lymph nodes. The surgeon takes the time necessary to make this decision; the goal is to capture as many viable nodes as possible without sacrificing drainage to the lower limb. If there is any doubt, the more conservative route is always taken and the lymph node is spared. An unsuccessful lymph node transfer is clearly preferable to iatrogenic lymphedema. Finally, the medial border of the flap is then defined, and the flap dissection is completed.
After flap harvest, a 10-second count of the sentinel lymph nodes of the lower extremity and lymph node flap is separately recorded for documentation and further study. Near-infrared imaging can be used to assess the content of the lymph nodes in the flap if desired. The flap is then transferred, and intravenous indocyanine green is administered after anastomosis to confirm good perfusion of the flap (Fig. 37-3).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree