CHAPTER 35 Lymphaticovenular Anastomosis
KEY POINTS
Excision of a lymph node in the extremities causes dilation of the peripheral lymphatic vessels and degeneration of smooth muscle cells within a short period.
Although smooth muscle cells regenerate, they remain small unless full restoration of lymph flow is achieved.
Combination therapy that consists of the reconstruction of lymph reflux function by microscopic lymphatico-venular anastomosis and compression is an effective treatment for this condition.
Although anastomosis is effective as a preventive and early stage application, it is also effective for patients with chronic edema (lymphedema).
In the future, a method will be developed to reconstruct the reflux function based on vascularized lymphatic implantation, in addition to the promotion of preventive anastomosis.
Both conservative therapy and surgical procedures (for example, tissue ablation and classic lymph–vascular conduction surgery) have been performed to treat lymphedema. Conservative therapy is directed at slowing the progression of edema, and surgical interventions attempt radical treatment by redirecting the flow of retained lymph back to the venous system by an artificially prepared bypass. However, it is difficult to significantly improve prolonged edema with either of these methods. 1 , 2
With the recent advent of the supermicrosurgery technique (vascular anastomoses 0.5 to 0.8 mm in diameter) in plastic and reconstructive surgery, it has become possible to create an anastomosis with extremely tiny blood vessels. 3 – 6 Based on the microscopic anastomosis technique, a new method of lymphaticovenular anastomosis (LVA) has been developed that surpasses the performances achieved by conventional macroscopic lymph–vascular vein conduction 1 , 2 , 7 or LVA. 8 – 10
This chapter is adapted from Koshima I, Narushima M, Mihara M, et al. Treatments for leg lymphedema. J Jpn Soc Gynecol Oncol 27:10-18, 2009.
Currently patients undergoing surgical treatment in combination with anastomosis and compression therapy have been followed for more than a decade. 3 – 8 As a result, the conventional notion that “lymphedema is incurable” is shifting to a concept that the disease can be cured by early or preventive anastomosis or by combination therapy based on anastomosis and compression.
The Fate of Lymphatic Vessels With Lymphedema
Lymphedema develops in patients with congenital hypoplasia when there are few collateral pathways in the lymphatic system to drain an area. For this reason, the removal of even a few lymph nodes on the medial side of an extremity may cause rapid dilation and failure in the related and nearby lymphatic vessels (if there are any).
As a result, the smooth muscle cells of the remaining and central lymphatics degenerate soon after the removal of the lymph nodes. Later, the smooth muscle cells may regenerate but often remain in a hypertrophied state. 3 Thus it is impossible to restore strong systolic contractile function of these remaining lymphatic vessels, resulting in a continued reflux disorder.
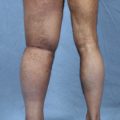
In patients with repeated lymphangitis during this time, occlusion and destruction of lymphatic vessels occur. Furthermore, there is growth of collagenous fibers formed by activated fibroblast cells between the subcutaneous fat tissues and the dermis. However, based on our knowledge of all biopsy findings in lymphatic vessels at the time of surgery of our clinical cases, the morphologic evidence of degeneration and regeneration of smooth muscle cells differs, depending on individual cases and sites within the extremities (Fig. 35-1).
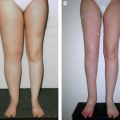
The duration from the onset of edema has no correlation with the degree of regeneration. 3 However, the degree of regeneration of the smooth muscle cells is to some extent correlated with the degree of postoperative reduction in edema and cellulitis. Moreover, even if lymphatic vessels are destroyed throughout the area, lymphatic vessel function in the distal extremity is frequently preserved. As a result, partial improvement is often achieved in many patients with bypass surgery only in the peripheral parts. Also, because some reduction in edema is observed in the femoral region by anastomosis at the ankle joint in some patients, it may be true that valvular insufficiency occurs in the lymphatic vessels in edema patients, causing lymphatic return.
Assessment of Lymphatic Functional Status
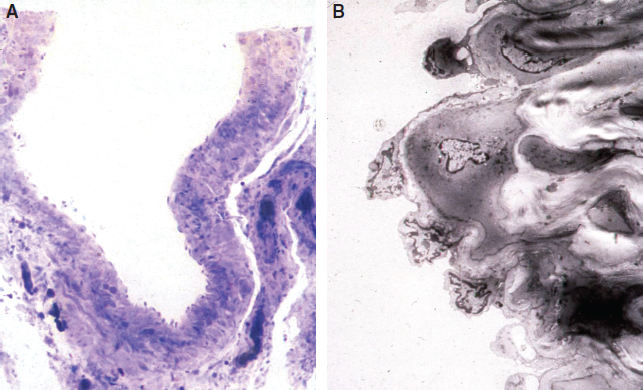
Conventional methods, such as lymphoscintigraphy, can indicate the location of lymph collectors but cannot provide information about the functional state or specific areas of occlusion. As a test for lymph collector functionality, the recently developed fluorescence lymphography shows promise. By applying infrared rays to subcutaneously injected indocyanine green (ICG), it is possible to confirm that the ICG is rapidly flowing back through lymphatic vessels while showing fluorescence in the normal extremities. In the edematous region, it is possible to easily confirm the degree of reflux under direct vision 11 (Fig. 35-2).
In mild lymphedema, whereas fluorescently stained lymphatic vessels are generally not observed in the proximal or medial part of the limb, a few vessels may be recognized in the more peripheral and distal areas. With unilateral lymphedema of the lower limb, these vessels do not fluoresce, even in unaffected lower limbs in many cases. This suggests that the reflux function of the lymphatic vessels may have been significantly lost in patients with lymphedema of the lower limb, including that of the unaffected side at the time edema developed.
Treatment Methods
The major treatment method relevant to LVAs is compression therapy with elastic stockings.
LONG-STRETCH ELASTIC STOCKINGS
Stockings worn to prevent edema formation work by applying external pressure to enhance tissue pressure. This is a core, indispensable therapy, even for mild edema, and can be used daily from the early stages of edema. Patients are instructed to wear them without exception during the day when standing. As the stockings loosen (usually after about 6 months because of frequent and repeated washing), they should be replaced with a new pair. These are often adapted to fit individuals by adding a fastener (zipper) or an elasticized band, as required. For patients who require the stocking larger than standard size or have irregular-shaped limbs, a custom-made stocking is ordered. With the Földi method (which uses compression concomitant with intensive massage therapy based on hospitalization), surgical treatment is a contraindication. 12
LYMPHATICOVENULAR ANASTOMOSIS
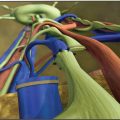
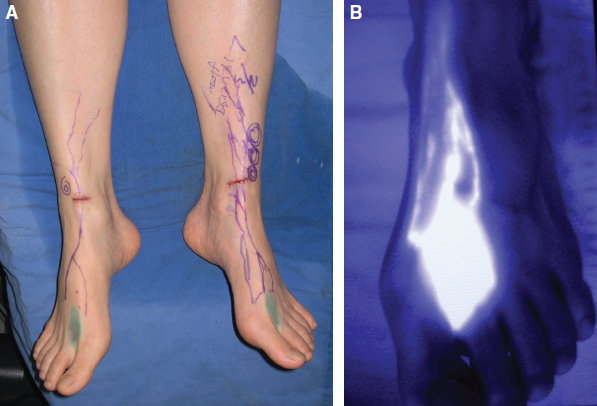
LVA is a permanent solution for the lymphedema, rerouting lymph back to the venous system through a bypass prepared for it with no need for compression therapy. We have performed 20 to 30 LVAs, guided by supermicrosurgical magnification of the site. A fine needle with a 50-micron (1/20 mm) suture (Fig. 35-3) is used to link the vessels.
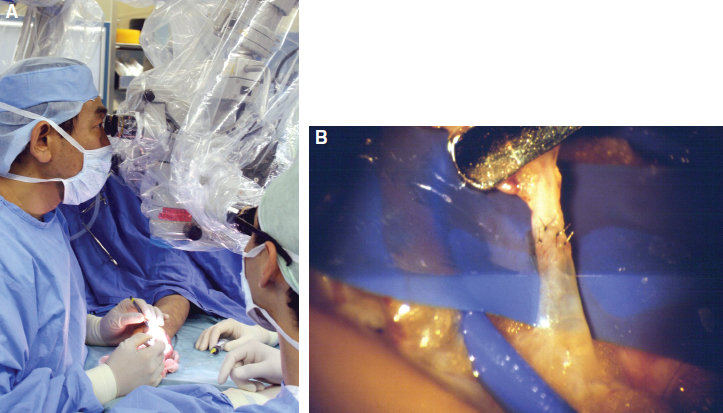
Our operative method is different from the conventional lymphaticovenous anastomosis 13 in that the cutaneous veins to be refluxed are connected with tiny venules immediately beneath the dermis or in the shallow part of the fat layer in an end-to-end anastomosis. 3 – 6 In principle, the anastomosis is performed after the use of conservative therapy (mainly by outpatient treatment) based on intensive continued compression for at least 6 months before surgery. The compression therapy is continued even after surgery (combination therapy). With recently developed fluorescence lymphography, it is possible to identify lymphatic vessels with normal reflux function by observing their presence under the skin. With the use of a local anesthetic during surgery, it is possible to readily expose those lymphatic vessels with good functionality with a small incision. 11 As a result, this operative method, which should be performed only by experienced surgeons, is being increasingly adopted by facilities worldwide.
History of Lymphaticovenular Anastomosis
The first reported experimental lymphaticovenous anastomosis was performed by Laine in 1963. 12 Yamada 14 first performed clinical application of lymphaticovenous anastomosis for obstructive lymphedema in 1967. Degni 7 described end-to-side anastomoses between a saphenous vein and surrounding lymphatic vessels with a single stitch in the inguinal region. O’Brien 8 described the same approach to treat lymphedema referring to Yamada’s procedure. However, these procedures were not widely accepted, because outcomes of the treatments were poor.
Nevertheless, the achievements of these pioneers laid the foundations for the subsequent development of lymphedema surgery. The introduction of supermicrosurgical technique in the 1980s was the critical development in reconstructive microsurgery. This technique was used to provide successful fingertip replantation and contributed to the development of the deep inferior epigastric perforator (DIEP) flap, the initial perforator flap. 15 , 16 Koshima 3 – 6 applied supermicrosurgical technique for LVA to manage lymphedema patients in the 1990s, and further improvement of edema more than before was obtained. This technique made possible the precise anastomosis of vessels smaller than 0.5 mm in diameter. The development of supermicrosurgical instruments and suture materials helped to ensure more accurate surgical approach for lymphedema. Based on our experience, we recommend the use of multiple surgical microscopes at the same time to perform multiple LVAs quickly and to increase the number of anastomosis (see Fig. 35-3). We also recommend the use of elastic stockings after surgery for at least half a year.
LVAs have proved to be effective, especially in patients in the earlier stages of lymphedema (stage 1 or 2), since here the mechanism of degeneration of lymphatic channel smooth muscle cells is well understood. 3 However, LVA only is not fully effective for severe and advanced edema. For those cases, we now use combined surgical treatments with multiple LVAs and functional lymphatic transfer.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree