CHAPTER 41 Combined Surgical Treatment for Breast Cancer–Related Lymphedema
KEY POINTS
Accurate diagnosis is essential.
A careful history as well as appropriate imaging helps establish the diagnosis.
Only patients with functioning lymphatic channels are candidates for reconstruction.
Patients with advanced lymphedema are better candidates for excisional procedures.
Breast cancer–related lymphedema (BCRL) is a persistent and debilitating complication that affects the health-related quality of life of breast cancer patients. 1 – 6 Findings from recent studies suggest that as many as 30% of women will develop this condition after breast cancer treatment. 7 – 9 Treatment has traditionally been based exclusively on conservative physical and medical therapies, but surgical approaches have emerged over the years in an attempt to provide a more effective or complementary treatment. Most such surgical approaches can be considered either palliative or reconstructive.
Palliative approaches can be excisional or reductive. One of the first techniques described was radical excision, the Charles procedure, 10 described in 1912. It consisted of resecting the skin and subcutaneous tissue and applying a skin graft. Results were poor, and this technique is now indicated only in extreme cases of functional limitation. More recently, vibroliposuction (Brorson’s technique) has been used to reduce hypertrophic adipose and fibrotic tissue, and it has been found to improve the quality of life in affected patients. 11
Reconstructive approaches aim to improve or restore the functionality of an impaired lymphatic system. Those most commonly used to date are autologous vascularized lymph node transfer (ALNT), 12 lymphaticovenular anastomosis (LVA), 13 – 16 and lympholymphatic bypass. 17 Because of a lack of consensus regarding the most effective surgical procedure for lymphedema, 18 in 2005 we chose to perform autologous lymph-node transplantation (ALNT), following Becker’s technique, 12 and LVA following Koshima’s technique. 13 We chose these techniques as we had confidence in their reproducibility and in their rationale. 12 – 14 We did not perform lympholymphatic bypass (Baumeister’s 17 technique) because we considered the risk to develop secondary lymphedema and the scarring and morbidity at the donor site was considerable.
On the basis of our results using these two approaches, we realized that an accurate preoperative assessment was needed to select the most suitable approach for each individual patient. Our combined surgical treatment evolved from this concept and we have followed these techniques since 2009.
To perform preoperative assessment we rely on imaging techniques such as indocyanine green (ICG) lymphography 16 and MR lymphography. 19 , 20 Both these techniques are effective for this purpose. Our surgical approach varies, depending on the results of the preoperative study. In patients who have residual function of their lymphatic system, we perform LVA or LVA in combination with ALNT. In later stages of lymphedema, characterized by excess adipose and fibrotic tissue, we opt for a reductive technique (vibroliposuction 11 ).
The risk of developing lymphedema after breast cancer depends on several factors, such as axillary node dissection, radiotherapy, or obesity, and it is also related to the anatomic variability between patients. ICG lymphography allows visualization of the anatomic patterns of the lymphatic system and their degeneration. It reveals how channels are more functional in some patients than in others. Patients with secondary drainage channels will be able to maintain better activity of the lymphatic system, and their risk of developing lymphedema may be lower. The differences among patients regarding these factors underscore the need to customize treatment to match the needs of individual patients.
Preoperative Assessment
The key concept in decision-making regarding potential candidates for reconstructive surgical techniques for lymphedema is to identify patients who still have a remaining functional lymphatic system. Following an accurate clinical evaluation, assessment with diagnostic imaging techniques is essential to study the functional and morphologic features of the lymphatic system so that each patient can be offered the best therapeutic option.
CLINICAL EVALUATION
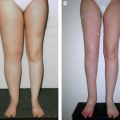
A full clinical examination is crucial in all potential candidates, in addition to a detailed medical history, so the lymphedema stage can be determined according to the International Society of Lymphedema. 21 This evaluation allows differentiation between pitting and nonpitting edema. Nonpitting edema is characterized by severe fibrosis and hypertrophy of adipose tissue and indicates an advanced stage of the disorder. During the clinical examination, limb circumferences are measured at predetermined anatomic levels and photographic documentation is obtained.
DIAGNOSTIC IMAGING TECHNIQUES
Diagnostic imagining techniques are needed to determine an appropriate therapeutic strategy for each patient. Our assessment protocol involves: indocyanine green (ICG) lymphography, lymphoscintigraphy, MR lymphography, and in some cases, CT angiography. For us, the essential technique is ICG lymphography, which we perform as a diagnostic test in the outpatient clinic during the first consultation. Depending on the results, we will plan to do additional imaging studies.
Indocyanine Green Lymphography
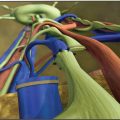
We have found that ICG lymphography provides a decisive test image. This is the first preoperative test in our assessment protocol, since it shows the functionality of the lymphatic system. The procedure consists of injecting 0.1 to 0.2 ml of indocyanine green dye subcutaneously at the second and fourth interdigital web spaces of both arms. ICG is captured by an infrared camera system, and functioning lymphatic channels can be visualized on a display device as fluorescent channels (Fig. 41-1).
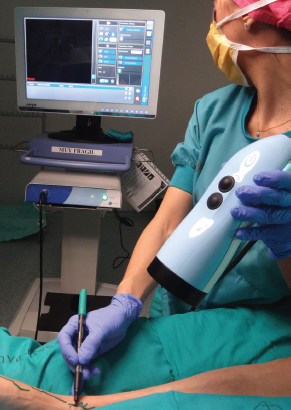
ICG lymphography provides valuable information in our assessment, making it a double evaluation:
First, it allows the examiner to see immediately after injection the velocity of the contrast ascending to the axilla, and after 5 minutes where the contrast medium is stored, and the dermal backflow can be evaluated. Thus the exact location of the active lymphatic channels and transport capacity of them can be determined. With the use of ICG lymphography we have realized that there is a substantial anatomic and functional variability between patients, not only in the distribution and the number of lymphatic channels, but also in the velocity of transport of ICG.
Second, ICG lymphography provides valuable presurgical data about the degree of impairment in the lymphatic system. It identifies the number of enhanced lymphatic channels and their appearance and shows the lymphatic patterns and the presence and appearance of dermal backflow patterns. In addition, the uptake of ICG in a lymphatic channel and its distal to proximal transport means that the channel maintains its contractile function. This information is of crucial importance during the preoperative assessment, because only a patient with functioning lymphatic channels can be considered a potential candidate for LVA surgery.
The preoperative planning assessment with ICG lymphography is done the day before LVA surgery or the same day in the operating room. It provides a map of the lymphatic system, and the lymphatic channels are marked on the patient’s skin surface. We complete the preoperative mapping with the information provided by MR lymphography, as explained later in more detail.
ICG lymphography is also of paramount importance during ALNT surgery helping to understand the lymphatic pattern of the donor site limb. Because we normally use the superficial inguinal nodes as donor nodes for the transplantation in the axilla, during ALNT surgery lymphography allows us to localize the lymph nodes draining the inferior limb. This step reduces the risk to harvest a flap containing these lymph nodes, which would induce an iatrogenic lower limb lymphedema, as recently described. 22
After LVA, ICG lymphography also helps us to assess whether the lymphatic transport function has improved compared with the preoperative examination, to evaluate the patency of an LVA, and to select new functional lymphatic channels when a secondary LVA surgery is planned. After ALNT, ICG lymphography can demonstrate the viability of autotransplanted lymph nodes when they are superficial (less than 2 cm deep).
A considerable limitation of ICG lymphography is that it only allows exploration of the superficial tissue (1 cm), and when there is a diffuse pattern with dermal backflow in the first minutes of injection, a white image is seen that could encumber visualization if there are deeper channels, especially in obese patients or those with fat hypertrophy. As a result, we start to work in combination with MR lymphography.
Lymphoscintigraphy
Lymphoscintigraphy is a standardized imaging test that provides good information on lymphatic function, as well as comparative information for postoperative assessment. Human serum albumin or sulfur colloid is injected into the first and second web space of each hand. The patient is asked to clench and release the fist repeatedly. Images are obtained with the patient in the supine position. Over the first 40 minutes and at 60, 120, and 180 minutes after the injection, dynamic images are taken of both arms, the axilla, and the liver. The main parameters evaluated are the tracer pathway, its appearance time at the axilla, the presence or absence of the major lymphatic collectors, visualization of nodes, and the presence or absence of dermal backflow. If axillary tracer uptake is absent, potential autologous lymph node transplantation to the axilla level could be planned. In a patient with upper limb lymphedema but who has remaining lymph node functionality at the axillary region, there is no reason for lymph node transfer in the axilla. The surgeon should consider some other technique or in some special cases might opt for autologous lymph node transplantation to the cubital fossa, or even to the wrist level.
As an objective comparative follow-up method, further lymphoscintigraphy is performed 12 months after surgery to assess any improvement in drainage and the viability of the transplanted lymph nodes. Postoperative lymphoscintigraphy is also useful to verify the long-term patency of LVA. Indirect findings in respect to preoperative lymphoscintigraphy, such as a decrease in dermal backflow or the disappearance of the tracer in a site of an LVA from tracer passage into the blood circulation, can demonstrate patency of the LVA.
Despite its advantages, lymphoscintigraphy does not provide detailed morphologic information about the lymphatic system, and findings must be complemented with ICG lymphography and MR lymphography.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree