CHAPTER 40 Lymphatic Microsurgical Preventing Healing Approach Concept
KEY POINTS
The lymphatic microsurgical preventing healing approach (LYMPHA) is a surgical technique for the primary prevention of arm lymphedema in patients with breast cancer, although it can also be used in patients with melanoma localized to the trunk.
The learning curve for the LYMPHA procedure is approximately 30 cases.
Lymphovenous shunts are used in the LYMPHA technique because they are faster, more physiological, and do not require lymph node or lymph vessel harvesting.
Axillary reverse mapping (ARM), which uses a small amount of blue dye, was developed to identify the lymphatic pathways draining the arm.
Lymphaticovenous anastomosis prevents lymphedema and reduces early lymphatic complications resulting from reduced regional intralymphatic pressure.
A side effect of axillary lymph node excision and radiotherapy for breast cancer is arm lymphedema in about 25% of patients (range 13% to 52%). Sentinel lymph node (SLN) biopsy has reduced the severity of swelling to nearly 6% (from 2% to 7%), but in a patient with a positive SLN, complete axillary dissection is still required. This is why the axillary reverse mapping (ARM) method 1 , 2 was developed—to identify and preserve the lymphatics draining the arm. The procedure consists of an intradermal and a subcutaneous injection of a small quantity (1 to 2 ml) of blue dye at the medial surface of the arm, which helps locate the lymphatic pathways draining the arm. The ARM technique assists in identifying the variable clinical anatomy that differs from what is already generally appreciated, that is, the most common location of arm lymphatics below and around the axillary vein. In about one third of patients, blue lymphatics extended to within 3 to 4 cm of the vein, the site at which the SLNs were easily located. This explained the occurrence of lymphedema after only SLN biopsy. 3 The ARM procedure also showed that blue nodes were almost always placed at the lateral part of the axilla, under the vein, and above the second intercostobrachial nerve. 4 The risk of arm lymphedema was decreased if the lymph nodes related to arm lymphatic drainage remained in place, but the main risk of not retrieving all the nodes was leaving metastatic disease in the axilla. On the contrary, the arm lymphatic pathways when they enter the axilla intuitively cannot be the site of a metastatic breast tumor, and their preservation certainly brings about a significant decrease in the incidence of lymphedema. Based on long-term experience with lymphaticovenous anastomosis 5 , 6 for lymphedema treatment, we developed and performed preventive lymphaticovenous anastomosis during nodal dissection.
This chapter was adapted from Boccardo F, Casabona F, De Cian F, Friedman D, Murelli F, Puglisi M, Campisi C, Molinari L, Spinaci S, Dessalvi S, Campisi C. Lymphatic Microsurgical Preventing Healing Approach (LYMPHA) for primary surgical prevention of breast cancer-related lymphedema: over 4 years followup. Microsurgery 34:421-424, 2014; with permission from the publisher.
We 7 reported on the long-term outcome of our experience in the prevention of breast cancer–related lymphedema with the lymphatic microsurgical preventing healing approach (LYMPHA), which consisted of anastomosis of the arm lymphatics to a collateral branch of the axillary vein.
Clinical Experience
From July 2008 to December 2012, 78 patients underwent axillary nodal dissection for breast cancer treatment, 74 of whom underwent the LYMPHA procedure (Table 40-1). The LYMPHA procedure could not be performed in four patients because no afferent lymphatics could be found (three patients) and because of massive metastatic disease (one patient).
Variable | Patient Group |
Age (yr) | 57 (42-69) |
BMI | 24 (21-33) |
Number of lymph nodes retrieved | 19 (12-21) |
Number of metastatic lymph nodes | 3 (0-4) |
Type of surgery (mastectomy/tumorectomy cases) | 34/40 |
Radiotherapy | 35 underwent radiotherapy; 39 did not |
Cellulitis | 14 had cellulitis; 60 did not |
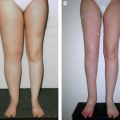
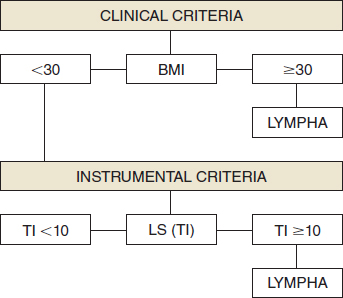
The indications for the LYMPHA technique were based on clinical and lymphoscintigraphic parameters (Fig. 40-1). It is well known that obesity predisposes an individual to lymphedema, and therefore patients with a BMI higher than 30 were considered at high risk for lymphedema and candidates for LYMPHA. Patients with a normal BMI were studied with lymphoscintigraphy, which indicated latent lymphatic impairment still not evident clinically.
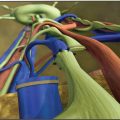
Regarding lymphatic arm drainage, blue nodes were identified in almost all patients after blue dye injection at the medial surface of the arm. All blue nodes were resected, and two to four main afferent lymphatics from the arm were prepared and used for anastomoses. Lymphatics were introduced inside the vein cut-end by a U-shaped stitch. Several other stitches were inserted to fix the lymphatic adventitia to the vein wall (Fig. 40-2). The operation usually took only 20 minutes on average, because both the lymphatics and vein were prepared during nodal dissection. Drain tubes were removed after about 10 days (range 7 to 12 days). No bandages were necessary on the arm, and patients did not wear any elastic garment.

Volumetry was performed preoperatively in all patients and after 1, 3, 6, and 12 months and once a year. Lymphoscintigraphy was performed in 45 patients preoperatively and also in 30 patients postoperatively after at least 1 year, comparing the preoperative and postoperative lymph transport indexes. 8 Statistical analysis was performed by Fisher’s exact test (p <0.05).
Long-Term Outcome
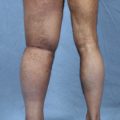
Seventy-one patients had no sign of lymphedema, and volumetric measurements were the same as before surgery (Table 40-2). In three patients from the initial clinical experience, lymphedema occurred 8 to 12 months after surgery, usually with the appearance of lymphangitic attacks. Radiotherapy caused temporary edema of the arm in eight patients, and the edema became permanent in three patients. Thus the incidence of secondary arm lymphedema after the LYMPHA technique was 4.05%.
Lymphoscintigraphy showed the patency of the lymphaticovenous anastomosis at 1 to 4 years after surgery. When we compared the lymphatic transport index with preoperative conditions, the results showed a significant improvement in all patients with early liver tracer uptake (Fig. 40-3). Lymphoscintigraphy was always performed in the same department, which studied the superficial lymphatic circulation by injecting the tracer into the dorsum of the hand.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree