CHAPTER 36 Vascularized Lymph Node Transfer
KEY POINTS
Autologous vascularized lymph node transfer is the optimal treatment to reduce the swelling of lymphedema that is resistant to physical therapy.
Preoperative imaging studies to identify the leakage site are critical to the success of lymph node transfer.
To obtain good results, it is imperative that the pathology be well understood and evaluated so that the correct treatment strategy can be determined.
MR lymphangiography helps identify the type of edema and is critical to deciding the best treatment option.
A combination of vascularized lymph node transfer and lymphaticovenular anastomosis is becoming more common and may yield superior results than either modality alone.
Lymphedema is a pathologic condition that results from a disturbance of the lymphatic system, with localized fluid retention and tissue swelling. Primary lymphedema is a congenital disorder caused by a malformation of lymph vessels and nodes. Chronic lymphedema is a progressive condition characterized by a degenerative and inflammatory deterioration of the dermis and subdermis, resulting in diffuse, irreversible fibrosis. Hypotrophy or hypoplasia of the lymphatic system occurs from a lack of lymphatic channel development or lymph nodes. In young adults with congenital lymphedema, swelling of the lower limb is unilateral in 70% of patients and bilateral in 30% of patients. Various forms of lymphedema affecting half of the body (right or left side) also exist.
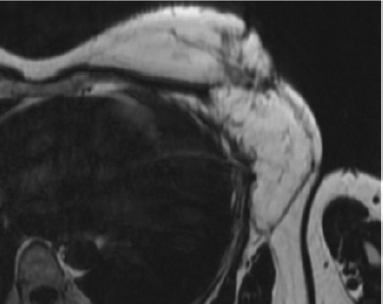
Major progress has been achieved in the radiologic diagnosis of patients with lymphedema. MR lymphangiography with T2-weighted imaging allows visualization of the anatomy of the lymphatic system with greater sensitivity than lymphoscintigraphy and potentially without the need for any injection, 1 , 2 although the use of gadolinium with venous suppression probably yields more consistent images. The ideal treatment of the affected limb should restore both function and the cosmetic appearance. Physical therapy is the most commonly performed treatment for chronic lymphedema, and many consider it the only treatment for long-term management. Certainly, it helps to control to some extent the evolution of the disease, but it is not a curative therapy.
Interruption of regular therapy will invariably result in swelling of the affected segment and continued worsening of the condition.
Lymph nodes express vascular endothelial growth factor C3, which promotes lymphangiogenesis. They also function as a sponge or “sump pump” for extracellular liquid, exchanging some lymphatic fluid with the venous circulation. Furthermore, they contain germinative cells. 3 , 4
Lymphedema presents in various forms, and treatment is not the same for all patients. Physicians must distinguish between secondary or acquired lymphedema and congenital lymphedema. The former may occur because of filariasis, after various cancer treatments, and from trauma and some surgical procedures. The latter may appear at birth, during the first years of the life, after puberty, or even later.
Autologous vascularized lymph node transfer (VLNT) is performed by some surgeons and is becoming increasingly popular. This technique is quite difficult to master and requires not only surgical and microsurgical skill, but also a good knowledge of the pathology involved.
Although many of my cases are the result of combinations of treatment (surgery and radiation), they are labeled as acquired. Although they are acquired, they are also iatrogenic, which is not a label that is commonly used. Iatrogenic lymphedema can appear after lumpectomy or mastectomy combined with radiotherapy and lymphadenectomy. It appears in 20% to 60% of patients, sometimes months or years after those procedures, depending on the number of nodes removed and/or irradiated. 5 The initial evaluation is done by lymphangioscintigraphy and recently by MR lymphangiography, which provides a real high-resolution map of the lymphatic system.
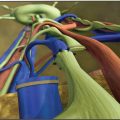
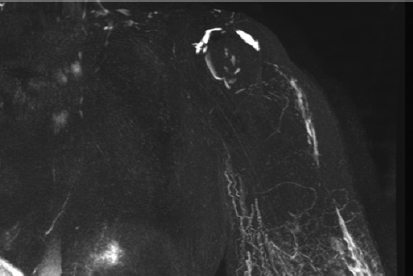
If lymphatic drainage of the limb is completely absent, physical therapy will never resolve the situation. After excision of the scar tissue and placement of a free fatty flap containing some lymph nodes, VLNT is the best alternative. Excision of the scar tissue can be extremely difficult; it demands a great deal of experience and a detailed knowledge of the anatomy of the brachial plexus region (Fig. 36-1). The main indications for VLNT include recurrent infections, extreme fibrosis and even elephantiasis, pain, resistance to physical therapy confirmed by the absence of drainage on MRI, and in combination with breast reconstruction (Fig. 36-2). Although pain is not a major feature of lymphedema, it certainly occurs. 6 Furthermore, patients often complain of discomfort and a feeling of extreme heaviness, which they sometimes equate with pain.
The donor sites are the lateral inguinal region (eventually combined with a deep inferior epigastric perforator [DIEP] or superficial inferior epigastric artery [SIEA] flap) or lateral thoracic and cervical flaps, either submental of supraclavicular. The dissection is difficult and requires an in-depth knowledge of the anatomy to minimize the risk of inducing lymphedema to the donor site.
Iatrogenic lymphedema of the lower extremity can occur after tumor resection and lymphadenectomy in the pelvic area (hysterectomy, prostatectomy, and lymphadenectomy) and/or in the inguinal region, such as after lymphadenectomy for melanoma. It can also occur after nodal biopsies, saphenofemoral ligations, hernia repairs, or aesthetic and reconstructive procedures in the thigh (thigh lift, lipoaspiration, and transverse upper gracilis flaps).
Identifying the problem is very important because it guides the surgeon on the optimal placement of the lymph nodes and which lymphatic pathways must be targeted during physical therapy. MR lymphangiography is the best investigation tool to evaluate the problem. The flap can be sometimes enlarged to reconstruct the breast defect and fibrotic area in the axillary or inguinal region.
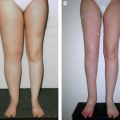
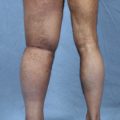
In our series, postoperative results showed improvement of between 40% for the upper extremity and 33% for the lower extremity in practically all patients, with complete resolution in a few patients after 2 years. 7 The ultimate result depends on the quality of the skin, degree of underlying fibrosis, volume of the limb, number of infections (which destroyed the lymphatic vessels), age of the patient, and duration of the lymphedema. Patients with the best results no longer need compression sleeves or any type of ongoing therapy.
In stage 2 or 3 lymphedema, improvement but not normalization was seen in 98% of patients. 6 The infection rate, pain, and discomfort were significantly reduced. In rare cases of weakness or palsy associated with brachial plexopathy, stabilization of the palsy was observed, and after the volume was reduced, tendon transfers could subsequently be performed. Very localized liposculpture could be performed later after fat deposits were present.
A successful outcome in stage 2 and 3 of the lower extremity was seen in about 50% of patients, but even patients with long-standing and extreme lymphedema had improvement. 8 The worst results were observed in stage 3 and 4 bilateral lymphedema. Better results will be achieved the earlier the diagnosis and surgical intervention.
Congenital lymphedema can present in various forms. Lymphoscintigraphy yields poor results and in our practice has been completely replaced by MR lymphangiography. However, this form of imaging is relatively new and is performed in only a few centers. The syndromes, hypoplastic or hyperplastic forms, and the level of the malformations are evaluated. Some individuals can present simultaneously, with both hypoplastic and hyperplastic forms in different parts of the body. VLNT is indicated in patients with hypoplasia, because lymphangiogenic proteins are contained in the transplanted nodes, and they simultaneously act as sump pumps to transfer some of the lymphatic fluid into the venous circulation. 8
Improvement was seen in most patients, even in those with advanced cases of elephantiasis. Mild cases, especially in children, can become normal. Combinations of VLNT with liposculpture can improve the aesthetic results by removing the fat deposits. 8
In our center we treat both congenital and acquired forms of lymphedema, and we have reviewed our results. Two groups of patients were studied, one iatrogenic and the second congenital.
During the past 20 years, more than 1500 cases have been reviewed. In upper extremity lymphedema, patients were classified based on the stage of the disease, duration of the lymphedema, number of infections, dash score, and associated problems, such as pain and palsy. In patients with lymphedema of the lower extremity, similar symptoms were examined.
A multidisciplinary team from my center selected the candidates for surgery. Regardless of the patient’s previous treatments before referral for reconstructive surgery, disease progression for a minimum of 1 year was confirmed. Patients had preoperative evidence of substantial progression of lymphedema from clinical stage 1 to stage 2 or from stage 2 to stage 3 despite an adequate treatment program over at least 12 months or progressive lymph fluid accumulation as seen on MR lymphangiography (or by lymphoscintigraphy).
For congenital cases, the hypoplastic forms were selected for VLNT. Sometimes combinations with other procedures were performed, mostly in patients with true elephantiasis. Elephantiasis can be caused by the hypoplastic or hyperplastic forms of the condition, and thus the diagnosis must be accurate. However, a combination of lymphatic reconstructive surgery with local excision was often necessary to avoid recurrent mycosis on the folds.
Physical therapy was complementary to the surgical treatment. Compressive bandages were applied, and manual drainage was performed for the first 3 postoperative months and progressively reduced thereafter. Patients were evaluated by perimetry, volumetry, and quality of life, and preoperative and postoperative photographs were taken after 6 and 12 months and 2 and 5 years. Between 1992 and 2011, lymphoscintigraphy was performed preoperatively and 1 and 5 years postoperatively. However, the correlation with the clinical results was not always appropriate. Since 2011, MR lymphangiography has yielded superior information and excellent images of the lymphatic system.
Surgical Technique for Upper Extremity Iatrogenic Lymphedema
For upper extremity lymphedema, whether or not combined with modified DIEP for breast reconstruction, the surgical approach to the axilla was performed through the previous scar. The scar tissue was dissected and adhesions were released as far as the anterior border of the latissimus dorsi. Dissection then continued until the thoracodorsal vessels and subscapular system were identified. This dissection can be very difficult, particularly after radiotherapy, because all the vessels and brachial plexus can be involved in the scar. Injury to these structures is a significant hazard. For this reason, surgical experience and anatomic knowledge are very important. After all the scar tissue was removed, the flap was placed in the apex of the axilla around these vessels. Microvascular anastomosis was made to available vessels, quite commonly the thoracodorsals.
LATERAL INGUINAL FLAP
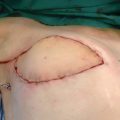
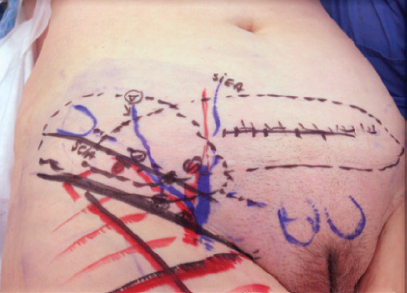
The lateral inguinal flap is supplied by the superficial circumflex iliac vessels, and anastomosis with the deep inferior epigastric vessels was seen in 80% of patients. An oblique incision was made between the iliac crest and the pubis above the inguinal crease (Fig. 36-3, A). The superficial circumflex iliac vessels were identified. The flap was raised on these vessels and included four to five nodes (Fig. 36-3, B). These nodes were dissected, freed, and elevated from lateral to medial at the level of the abdominal wall fascia extending medially if more bulk was required. The inguinal fold must be the lower limit of the flap. If excessive dissection is performed in the thigh, the risk of lymphedema at the donor site is significant. The dissection began by visualization of the superficial circumflex iliac vein. In that region, some lymph nodes are supplied by the circumflex iliac vessels, which mainly drain lymph from the abdominal wall. For this reason the harvesting of this flap does not impair the lymphatic drainage of the lower limb.
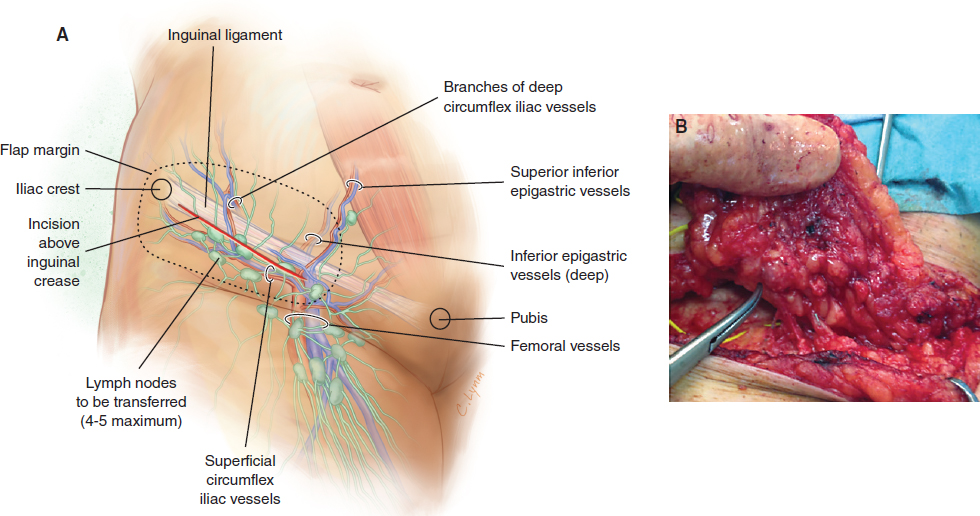
This flap can be transferred with a DIEP or SIEA, or it can be added as a free flap on a branch of another flap, such as the thoracodorsal artery perforator, superior artery gluteal perforator, or inferior gluteal artery perforator, or to a separate donor vessel in the axilla 9 (Fig. 36-4).
The circumflex iliac vessels must be dissected as far as possible to achieve maximal length and caliber. The DIEP can incorporate this flap, but it is important to confirm that the lymph node flap is well vascularized.
In the axillary region, debridement of the scar tissue and individualization of the branches of the thoracodorsal vessels were performed first. If the DIEP anastomosis is done to the mammary vessels, the VLNT must be dissected on the side opposite the DIEP pedicle. In this situation, it should not be assumed that the vascularity of the flap is carried by the DIEP. In most cases, a separate pedicle should be dissected for the lateral inguinal flap. In this case the flap will be anastomosed as the second anastomosis in the axilla (Fig. 36-5).
A second anastomosis was preferred to improve the vascularization of the transplanted nodes. Drains were placed in the defect, which was closed in two layers. The postoperative care of the inguinal region was important. The drains were removed after the second day, and a compression garment such as Spanx was recommended to prevent lymphocele. Perioperative antibiotic therapy was sufficient to prevent infection.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree