CHAPTER 11 Immune Regulation by the Peripheral Lymphatics
KEY POINTS
Lymphedema is characterized by numerous immune deficits, including altered wound healing and increased susceptibility to infection, which develop after an initial lymphatic insult. This connection between immune dysfunction and peripheral lymphatic dysfunction is also mirrored in most human inflammatory disorders, suggesting a crucial immune role for the lymphatics.
The peripheral lymphatics are a highly integrated component of the immune system capable of mediating their effector function in response to pathogenic and immunogenic stimuli in the local immune microenvironment.
After activation by a diverse range of stimuli, the lymphatics upregulate a range of immune modulatory molecules capable of mediating cell migration and local immune function. Activation also induces lymphangiogenesis to facilitate cell migration and enhance antigen, cytokine, and fluid clearance from the affected site.
As a key immune organ, the lymphatics represent a promising target for future lymphatic-directed immuno-modulatory therapies and a crucial area of interest for future research into human disease.
This chapter discusses the active roles of the lymphatics in an immune response. Active immune regulation by the peripheral lymphatics is essentially defined as the ability of the lymphatics to dynamically and specifically respond to immune mediators and subsequently modulate the function of peripheral immune cell populations in a stimulus-specific manner. Although these active immune roles remain poorly understood, they are critically important to the understanding of immune functioning as a whole.
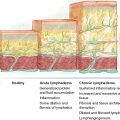
The peripheral lymphatic system plays several diverse roles in the human body, including the regulation of fluid balance and fatty acid transport from the gastrointestinal tract. Peripheral lymphatic dysfunction can lead to lymphedema, which is generally considered a circulatory condition characterized by abnormal fluid distribution (initially) and fat deposition (later stages). However, evidence of concurrent immune dysfunction in lymphedema suggests that lymphedema could also be classified as a functional immune disorder. The multiple immune deficits in lymphedema include excessive fibrosis and local inflammation, poor wound healing, increased susceptibility to infections, and increased risk for the development of malignancy in the affected area. 1 , 2
The appearance of immune dysfunction resulting from lymphatic dysfunction in lymphedema strongly supports a relationship between the lymphatics and the rest of the immune system. This relationship is reciprocal rather than linear, because lymphatic dysfunction is also observed as a result of immune dysfunction in primary immune conditions. For example, chronic inflammation in patients with chronic obstructive pulmonary disease is characterized by multiple immune defects, including abnormalities in the phenotype and density of lymphatic vessels. 3 Moreover, altered patterns of lymphangiogenesis (the formation of new lymphatic vessels) have been identified in most human inflammatory conditions, including psoriasis and chronic airway inflammation. 4 Finally, peripheral lymphatic vessels also play key roles in transplant rejection and tumor metastases. 5
Collectively, these observations highlight the complex and important interconnection between the lymphatic and immune systems. However, despite the identification of peripheral lymphatic involvement at a gross level in many diseases, the specific pathways and mechanisms underlying lymphatic-mediated regulation of immunity remain relatively poorly understood. The primary immune role for the lymphatics is the regulation of cell migration, which is mediated through both the expression of molecules involved in cellular trafficking and the formation of new lymphatic pathways by means of lymphangiogenesis. However, very few studies have directly investigated the immune role of the lymphatics in human disease, and thus the mechanisms of immune regulation remain poorly understood. In addition to this limited understanding, the lymphatics are also still often perceived as passive conduits for fluid, fatty acids, and immune cells. This perception inherently reduces the importance of the lymphatics as an immune organ, and this at least partially explains the omission of the lymphatics in most studies investigating immune function.
Lymphatic Activation by the Immune Microenvironment
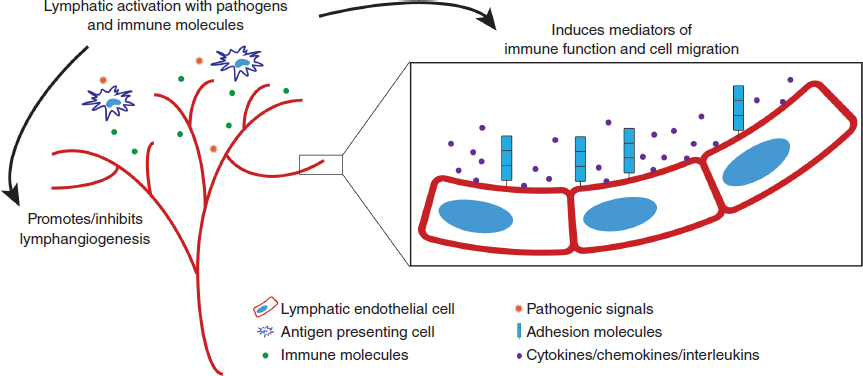
Compared with a passive component, the essential characteristic of an active component of the immune response is the ability to respond to diverse stimuli and change effector function in response to these diverse stimuli. For example, different activation signals induce different activation programs in macrophages and T cells, such as the classic M1/M2 macrophage responses or Th1/Th2 T-cell responses. This differential integration of signals allows functional specialization and active immune regulation. In contrast to their perceived role as a passive conduit for immune cells, the lymphatics can respond to a wide range of pathogenic stimuli (the socalled primary activation signals) and immune molecules (the socalled secondary activation signals), all of which induce different functional outcomes in immune cell activation/migration and lymphangiogenesis (Fig. 11-1).
PRIMARY ACTIVATION SIGNALS
Lymphatic endothelial cells express functional toll-like receptors (TLRs) and thus can respond to pathogenic stimuli, including lipopolysaccharide (via TLR4) or lipoteichoic acid (via TLR2). 6 , 7 As with other immune cell populations, stimulation via TLR4 (or any of the pathogen recognition receptors) induces the expression of a range of inflammatory mediators involved in effector function. 6 , 7 In vitro stimulation of lymphatic endothelial cells with lipopolysaccharide induced the expression of a large number of molecules, including interleukin 6 (IL-6), IL-8, chemokine (CC motif) ligand 21 (CCL21), vascular cell adhesion molecule 1, and intercellular adhesion molecule 1 (ICAM-1). 8 Although CCL21, vascular cell adhesion molecule 1, and ICAM-1 are related to the well-appreciated role of the lymphatics in cell migration, IL-6 and IL-8 are classic proinflammatory mediators involved in regulating the function of multiple immune populations, 8 suggesting that the lymphatics may also regulate local immune homeostasis.
In addition, lymphatic activation does not occur in isolation (as with an in vitro culture model) but instead in a complex, integrated immune milieu. This is perhaps best highlighted in TLR-deficient mouse models, which show concurrent deficiencies in lymphatic architecture/function and macrophage activation/migration. The macrophage deficits are at least partly caused by the reduced ability of the dysfunctional peripheral lymphatics to regulate macrophage recruitment, maturation, and function (and vice versa). 9 , 10
The other crucial observation that highlights the integration of the lymphatics into the normal immune response to pathogens is the ability of lymphatic endothelial cells to uniquely respond to distinct pathogens (as is observed in all immune cell populations). This stimulus-dependent specificity is observed not only in simplified in vitro restimulation assays with various TLR agonists 6 – 8 but also in more physiologic models of inflammation. For example, large differences in the transcriptional expression of key chemokines and integrins were observed in lymphatic endothelial cells isolated from models of oxazolone-induced contact hypersensitivity and complete Freund’s adjuvant-induced inflammation. 11 These differences in immune mediator expression by lymphatic endothelial cells contributed to gross differences in inflammation, cell activation/migration, and edema formation between models. 11 The large number of differentially expressed genes (>1000) in the inflammation-activated lymphatics also implies a far greater complexity than currently appreciated. 11 Thus different pathogenic stimuli induce distinct functional programs in the lymphatics, allowing their active regulation of different immune responses (see Fig. 11-1).
SECONDARY ACTIVATION SIGNALS
Although external pathogenic signals may be responsible for the primary initiation of an immune response, a diverse range of interleukins, cytokines, and chemokines (secondary activation signals), produced by activated immune cells, serves to modulate the resulting response. As an integrated component of the immune response, the lymphatics are capable of responding to a wide range of chemokines, interleukins, and other immune mediators with a unique pattern of activation depending on the stimulus. 4 , 12 The effector molecules induced by many of these immune mediators overlap with those induced by pathogenic stimulation, 8 , 13 suggesting that changes in lymphatic phenotype/function in the context of in situ human disease are directed by an inseparable combination of primary and secondary activation signals (see Fig. 11-1). Indeed, it is becoming increasingly clear that activation of all immune populations (now including the lymphatics) in the immune microenvironment is complex and multifaceted and thus likely poorly reproduced in vitro.
One of the important consequences of lymphatic activation by secondary immune mediators is the promotion or inhibition of lymphangiogenesis either directly or indirectly through the induction in the expression of lymphangiogenesis-promoting molecules in other immune populations. 4 , 12 In inflammation, lymphangiogenesis increases the density and size of the lymphatic vessels to increase fluid drainage, cell migration, and antigen clearance from the affected site and is intrinsically linked to the resolution or progression of inflammation. 14 , 15 Activated macrophages, T cells, mast cells, and dendritic cells can also directly produce vascular endothelial growth factors (VEGFs) A, C, and D, the canonical mediators of lymphangiogenesis. 4 , 12 Given these strong links between multiple immune modulators and lymphangiogenesis, it is not surprising that altered patterns of lymphangiogenesis are commonly observed in human disease.
Collectively, the ability of the lymphatics to regulate their effector function in response to secondary immune mediators emphasizes the lymphatics as an integrated component of the immune response (see Fig. 11-1).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree