Key Words
skin tumors, seborrheic keratosis, cysts, syringoma, keratoacanthoma, chondrodermatitis nodularis helicis, skin tag, melanoma mimic, sebaceous hyperplasia, nevus sebaceus, keloid, dermatosis papulosa nigra, acrochordon
Seborrheic Keratosis
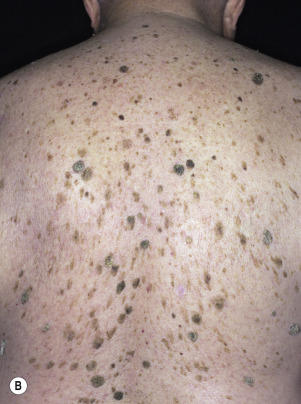
Seborrheic keratosis (SK) and nevi are the most common benign cutaneous neoplasms. SKs are of unknown origin and have no malignant potential. In recent years, somatic mutations in the fibroblast growth factor receptor 3 ( FGFR3 ) gene (more likely seen in histologic acanthotic and adenoid SKs) and p100α subunit of phosphoinositide 3-kinase (PIK3CA) (more likely in histologic hyperkeratotic SKs) oncogenes have been found in SK. The significance of these mutations is not known. One must be familiar with all of the characteristics and variants of these lesions to differentiate them from other lesions and to prevent unnecessary destructive procedures. SKs can be easily and quickly removed and, if the procedure is correctly executed, heal with little or no scarring. Most people develop at least one SK at some point in their lives. SKs appear in a substantial proportion of people younger than 30 years. The term senile keratosis is no longer appropriate for these lesions. The number varies from less than 20 in most individuals to numerous lesions. They can occur on any hair-bearing surface on the face, trunk, extremities, and genitals. They do not occur on the lips, palms, or soles. Patients refer to them as warts, but SKs do not appear to be associated with human papilloma virus.
Surface Characteristics.
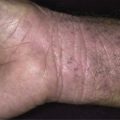
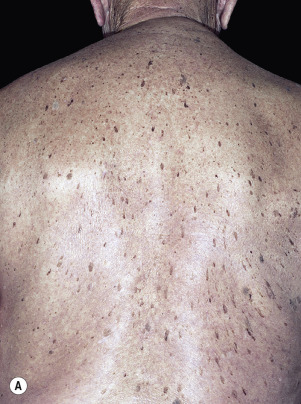
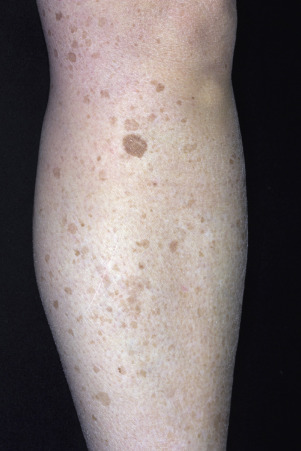
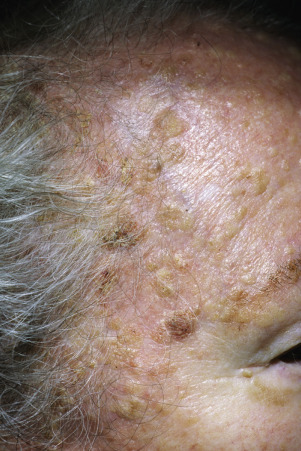
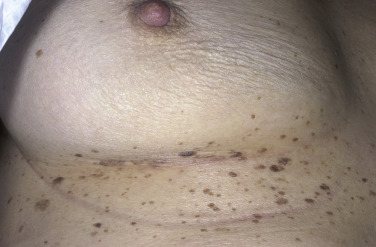
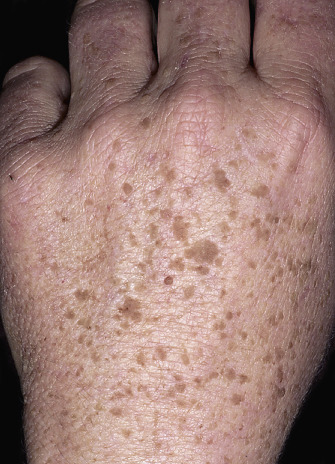
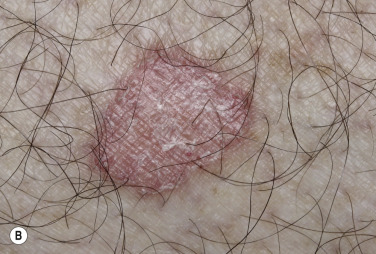
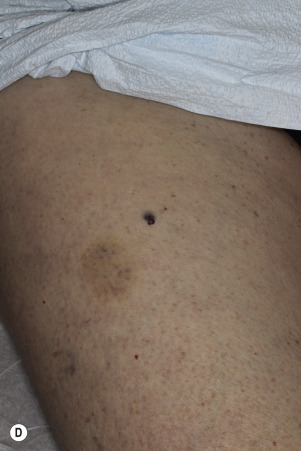
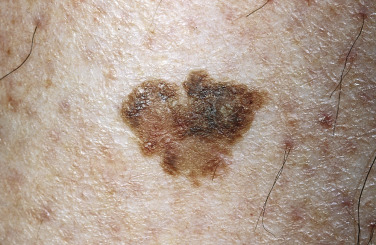
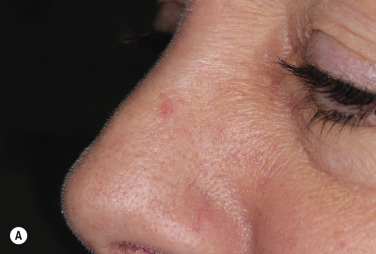
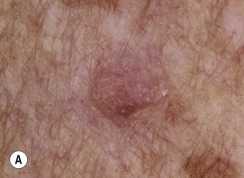
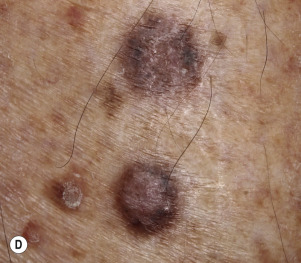
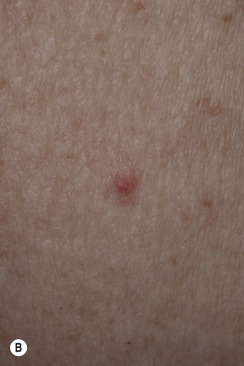
The surface of SKs is either smooth with tiny, round, embedded pearls or rough, dry, and cracked. SKs are sharply circumscribed and vary from 0.2 cm to more than 3 cm in diameter. They appear to be stuck to the skin surface and, in fact, occur totally within the epidermis. The surface characteristics vary with the age of the lesion and its location. Those on the extremities are often subtle, flat, or minimally raised and are slightly scaly with accentuated skin lines. Lesions on the face and trunk vary considerably in appearance, but the characteristics common to all lesions are the well-circumscribed border, the stuck-on appearance, and the variable tan-brown-black color ( Figs. 20.1 to 20.8 ![]() ). When the border is irregular and notched, the SK resembles a malignant melanoma ( Fig. 20.9
). When the border is irregular and notched, the SK resembles a malignant melanoma ( Fig. 20.9 ![]() ).
).
Smooth or Rough Surfaced.
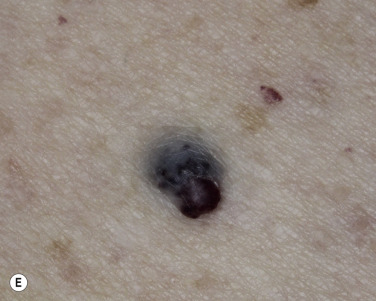
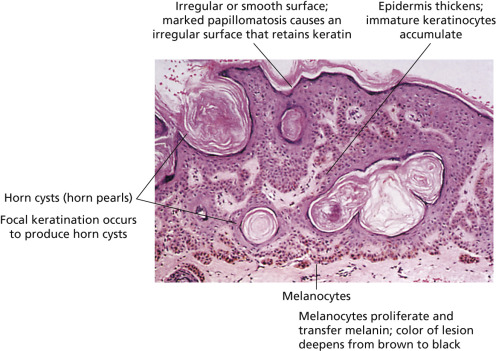
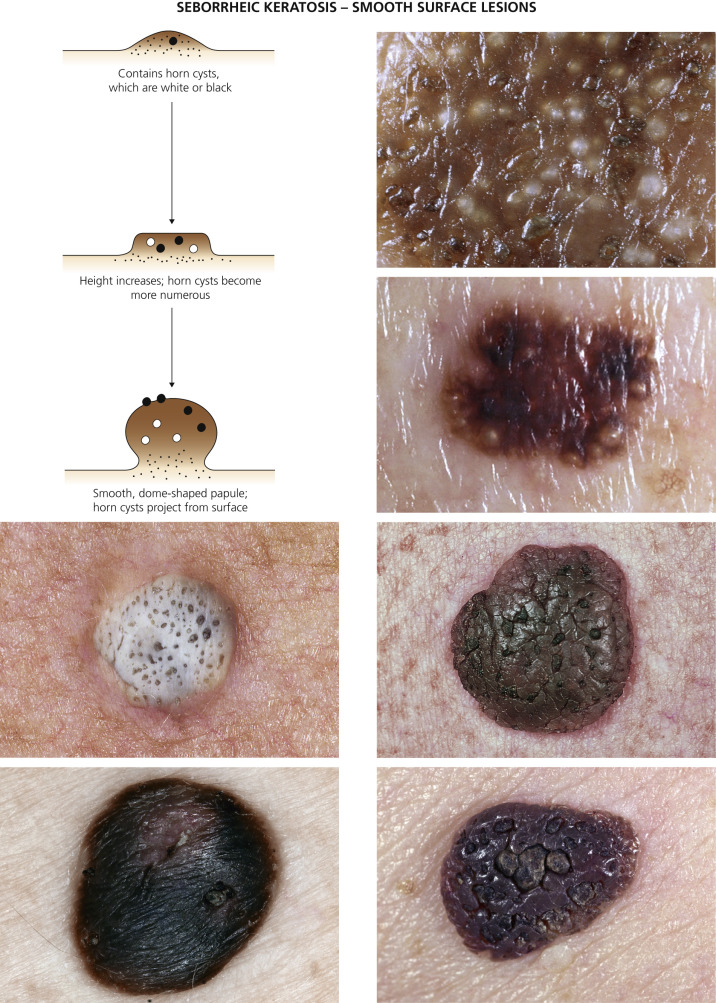
The surface characteristics show considerable variation ( Figs. 20.10 and 20.11 ). Smooth-surfaced, dome-shaped tumors have white or black pearls of keratin, are 1 mm in diameter, and are embedded in the surface. These horn pearls are easily seen with a hand lens. The presence of horn cysts on the surface helps to confirm the diagnosis of an SK. Horn cysts are also found on the surface of some dermal nevi. The rough-surfaced SKs are the most common. They are oval-to-round, flattened domes with a granular or irregular surface that crumbles when picked.

Seborrheic Keratosis Versus Malignant Melanoma.
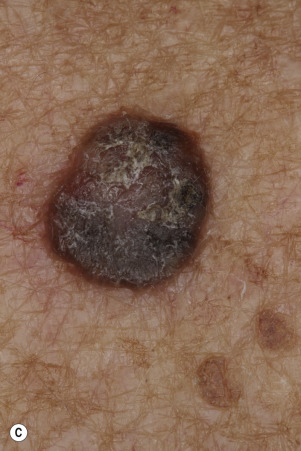
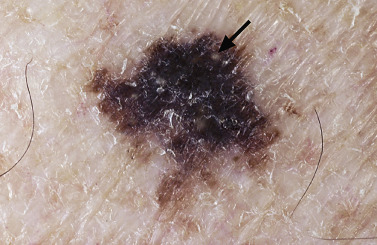
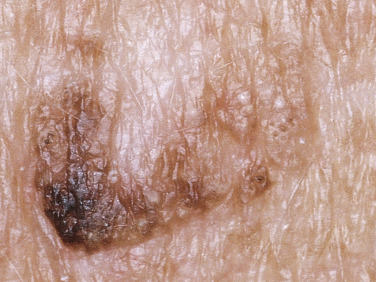
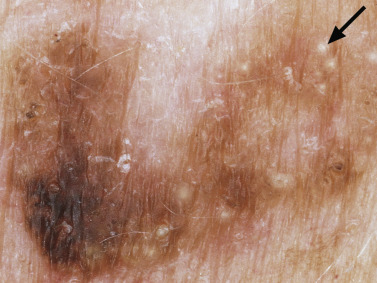
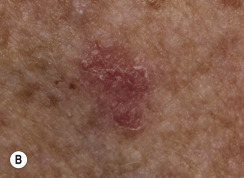
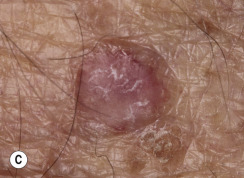
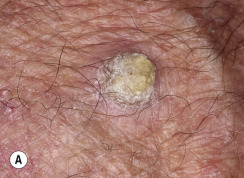
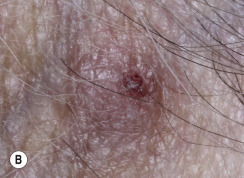
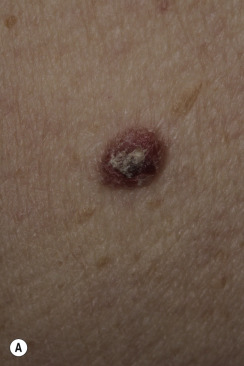
Many patients present with dark, irregular, sometimes irritated SKs and worry that they are melanomas. SKs can show many of the features of a malignant melanoma, including an irregular border and variable pigmentation ( Figs. 20.12 to 20.15 ). The key differential diagnostic features are the surface characteristics. Melanomas have a smooth surface that varies in elevation and in color, density, and shade. SKs preserve a uniform appearance over their entire surface. The presence of horn cysts on the surface helps to confirm the diagnosis of an SK. Examination with a hand lens is very helpful. Many SKs occur in sun-exposed areas.
Microscopic Mimic of Melanoma in Situ.
Seborrheic keratoses with linear basal clear cells mimicking melanoma in situ have been reported. Histopathologically, the lesions exhibit the typical architectural pattern of a seborrheic keratosis with acanthosis, papillomatosis, and hyperkeratosis. Along the basal layer a striking row of clear cells is present. The cells show a small nucleus with abundant clear cytoplasm. No nests are seen. The cells fail to react with S100 protein and Melan-A.
Accuracy of Diagnosis.
When a clinical diagnosis of SK is made with confidence, the lesion is often left untreated or a destructive method of removal is used. The diagnostic accuracy must be high to justify such practices, which do not yield material for histopathologic confirmation. A diagnostic accuracy of greater than 99% was established by dermatologists in a study. The extremely high diagnostic accuracy of greater than 99% justifies common clinical practice. A biopsy should be performed in cases of diagnostic doubt. Submit all specimens that have been removed for histologic examination.
Irritated Seborrheic Keratosis.
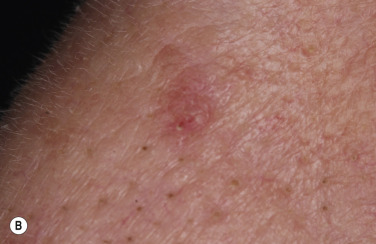
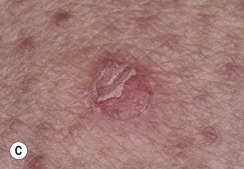
Although generally asymptomatic, SKs can be a source of itching, especially in the elderly, who have a tendency to subconsciously manipulate these protruding growths. Irritation can be aggravated by chafing from clothing or from maceration in intertriginous areas, such as under the breasts and in the groin. When inflamed, SKs become slightly swollen and develop an irregular, red flare in the surrounding skin ( Figs. 20.16 and 20.17 ![]() ). Itching and erythema can then appear spontaneously in other SKs that have not been manipulated ( Figs. 20.18 and 20.19
). Itching and erythema can then appear spontaneously in other SKs that have not been manipulated ( Figs. 20.18 and 20.19 ![]() ) and in areas without SKs. A halo of eczema can appear around SKs; the inflamed border is red and scaly and may represent a localized form of nummular (coin-shaped) dermatitis. The only treatment is to apply topical steroids or to remove all inflamed lesions. With continued inflammation, the SK loses most of its normal characteristics and becomes a bright red, oozing mass with a friable surface that itches intensely and resembles an advanced melanoma or a pyogenic granuloma. SK may appear similar to acantholytic acanthoma, lichenoid keratosis and verrucal keratosis ( Figs. 20.20 to 20.22
) and in areas without SKs. A halo of eczema can appear around SKs; the inflamed border is red and scaly and may represent a localized form of nummular (coin-shaped) dermatitis. The only treatment is to apply topical steroids or to remove all inflamed lesions. With continued inflammation, the SK loses most of its normal characteristics and becomes a bright red, oozing mass with a friable surface that itches intensely and resembles an advanced melanoma or a pyogenic granuloma. SK may appear similar to acantholytic acanthoma, lichenoid keratosis and verrucal keratosis ( Figs. 20.20 to 20.22 ![]() ).
).
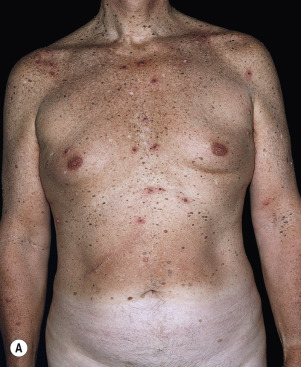
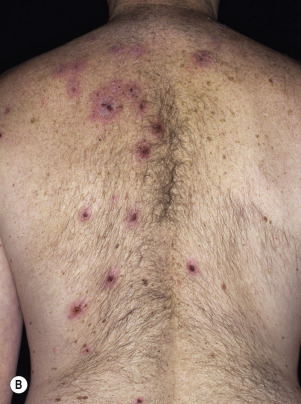
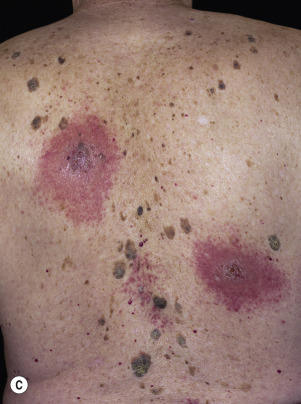
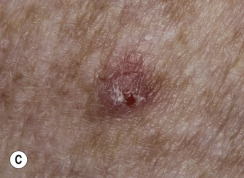
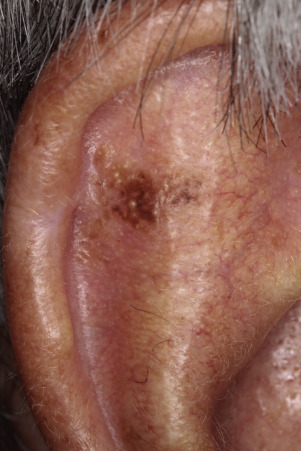
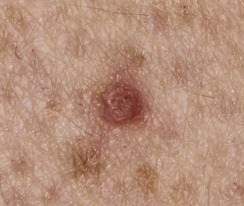
Leser–Trélat Sign (Eruptive Seborrheic Keratosis as a Sign of Internal Malignancy).
The sudden appearance of or sudden increase in the number and size of SKs on noninflamed skin has been reported to be a sign of internal malignancy (see Fig. 26.23 ). The malignancy most frequently associated with this sign was gastric adenocarcinoma. Acanthosis nigricans may accompany eruptive SK as a paraneoplastic syndrome.
Treatment.
Lesions are removed for cosmetic purposes or to eliminate a source of irritation. Since these growths appear entirely within the epidermis, scalpel excision is unnecessary. They are easily removed with cryosurgery or curettage. Lesions to be curetted are first anesthetized with lidocaine. With multiple strokes, a small curette is smoothly drawn through the lesion (see Chapter 27 ). SKs on the face or on other areas with inappreciable underlying support can be softened before curettage with the electric needle. Monsel’s solution controls bleeding, and the site remains exposed to heal. Some lesions are tenaciously fixed to the skin and resist curettage; others are on sites that are difficult to curette, such as the eyelid. These can be dissected with curved, blunt-tipped scissors. Cryosurgery is effective for thin SKs but posttreatment hyperpigmentation or hypopigmentation is a possible side effect.
Stucco Keratoses
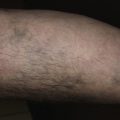
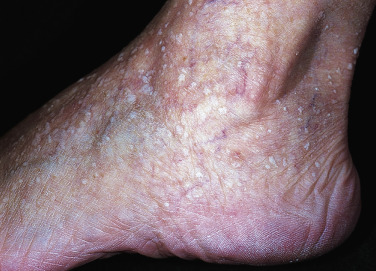
Stucco keratoses, sometimes referred to as barnacles, are common, nearly inconspicuous, papular, warty lesions occurring on the lower legs ( Fig. 20.23 ), especially around the Achilles tendon area, the dorsum of the foot, and the forearms of the elderly. The 1- to 10-mm, round, very dry, stuck-on lesions are considered by most patients to be simply manifestations of dry skin. The dry surface scale is easily picked intact from the skin without bleeding, but it recurs shortly thereafter. The lesions can be removed with curettage or cryosurgery.
Skin Tags (Acrochordon) and Polyps
Skin Tags.
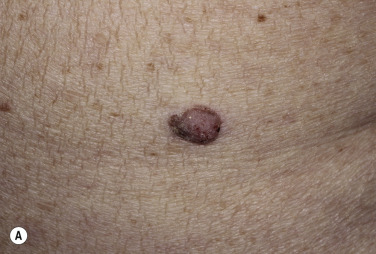
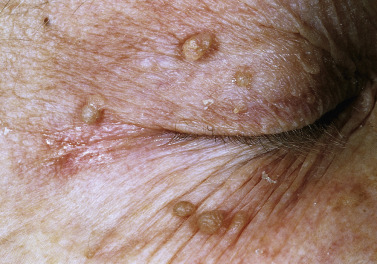
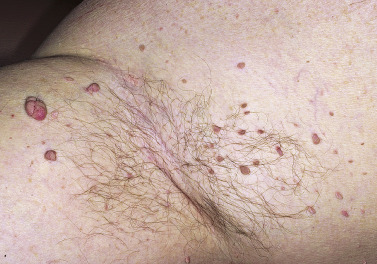
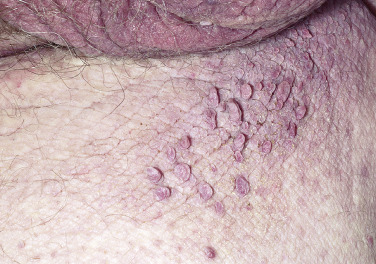
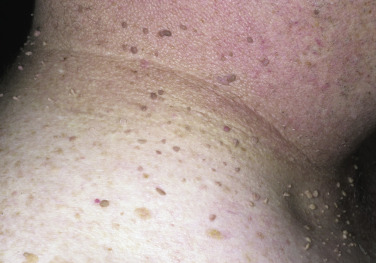
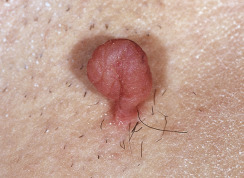
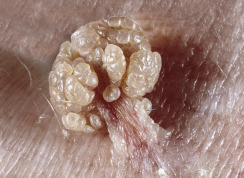
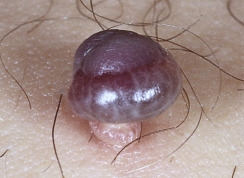
Skin tags are common pedunculated papules found in approximately 25% of males and females. They occur more often and in greater number in obese patients. The most frequently affected area is the axilla (48%), followed by the neck (35%) and inguinal region. The majority of affected individuals (71%) have no more than three skin tags per location. They can begin in the second decade, with a steady increase in frequency up to the fifth decade; older than this age range, there is no further growth. The skin tags begin as a tiny, brown or skin-colored, oval excrescence attached by a short, broad-to-narrow stalk ( Figs. 20.24 to 20.27 ![]() ). With time, a skin tag can increase to 1 cm as the stalk becomes long and narrow. Jewelry and clothing may catch and rub skin tags and annoy patients. The stalks are easily removed with iris scissors or with low-level electrocautery. Local anesthesia is usually not necessary.
). With time, a skin tag can increase to 1 cm as the stalk becomes long and narrow. Jewelry and clothing may catch and rub skin tags and annoy patients. The stalks are easily removed with iris scissors or with low-level electrocautery. Local anesthesia is usually not necessary.
Polyps.
Skin polyps have a long, narrow stalk and a broad tip ( Figs. 20.28 to 20.30 ). Sometimes they become twisted and compromise the blood supply. Lesions then turn dark brown or black. This sudden change is alarming to patients (see Fig. 20.30 ). Polypoid growths may be skin tags, nevi, or melanomas. Polyps occur on the eyelids (see Fig. 20.24 ), groin, axilla, or any skin surface except the palms and soles.
Skin tags and associated conditions. Skin tags may be a sign of diabetes mellitus and metabolic syndrome, especially accompanied by acanthosis nigricans. Patients with Crohn disease may have perianal skin tags and patients with Birt–Hogg–Dubé syndrome have the triad of skin tags (skin tag–like lesions), trichodiscomas, and fibrofolliculomas.
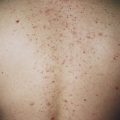
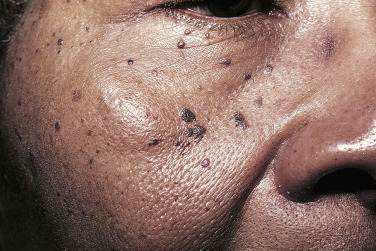
Dermatosis Papulosa Nigra
Young and middle-aged black people may develop multiple brown-black, 2- to 3-mm, smooth, dome-shaped papules on the face ( Fig. 20.31 ) in a photodistribution. The average age of onset is 22 years. There is a female predominance and family predisposition. They probably represent a type of SK. Patients who desire removal should be informed that white, hypopigmented scarring may result. The patient’s response should be determined by curetting or freezing one or two lesions and permitting them to heal completely. EMLA (a mixture of lidocaine 2.5% and prilocaine 2.5%) may be applied under occlusion for 30 minutes to provide anesthesia before electrodesiccation of the superficial skin lesions.
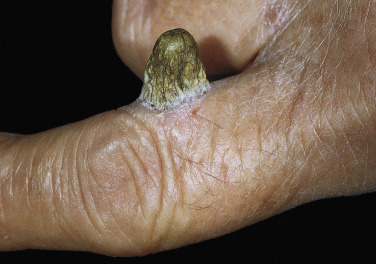
Cutaneous Horn
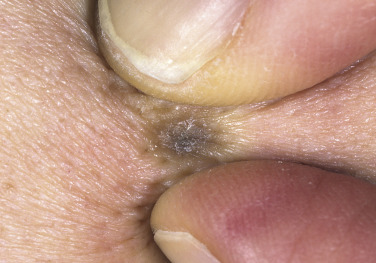
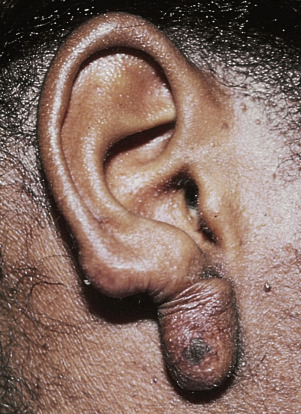
Cutaneous horn refers to a hard, conical projection composed of keratin and resembling an animal horn. It occurs on the face, ears, and hands ( Fig. 20.32 ) and may become very long. Warts, SKs, actinic keratosis, tricholemmoma, and squamous cell carcinoma (SCC) may all retain keratin and produce horns. Treatments include cryosurgery, iris scissor snip, or surgical excision.
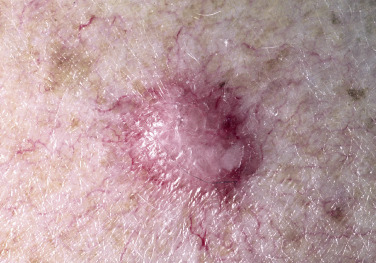
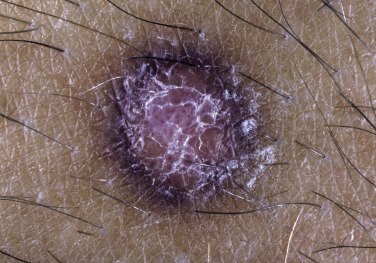
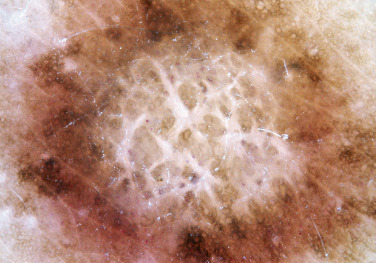
Dermatofibroma
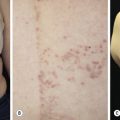
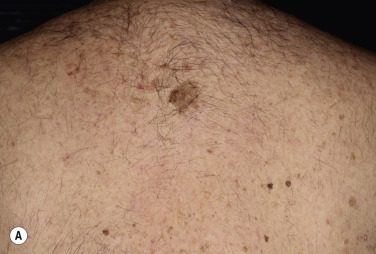
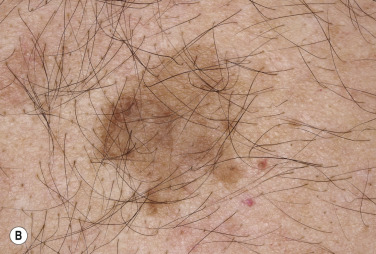
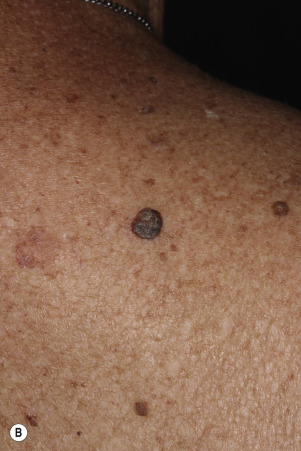
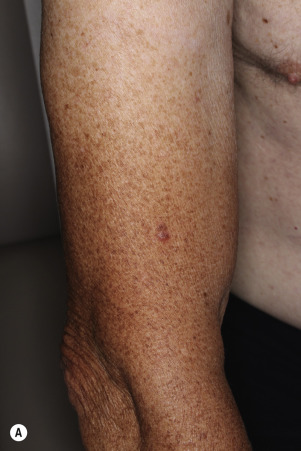
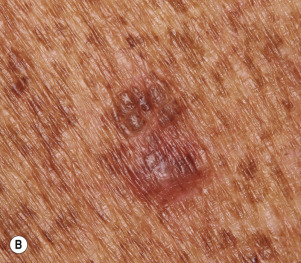
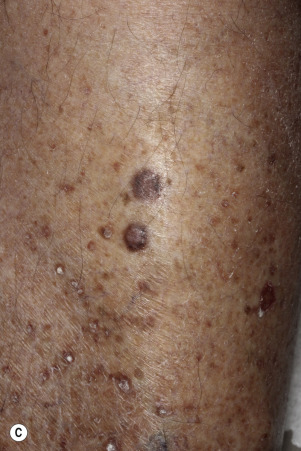
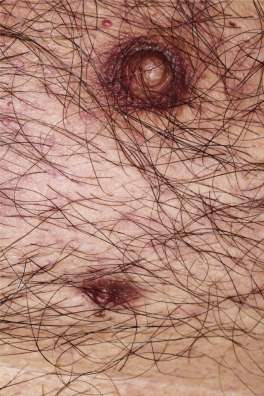
Dermatofibromas are common, benign, asymptomatic to slightly itchy lesions occurring more frequently in females. They vary in number from 1 to 10 and can be found anywhere on the extremities and trunk, but they most likely occur on the anterior surface of the lower legs. Dermatofibromas represent a fibrohistiocytic reaction to trauma, a viral infection, or an insect bite. They appear as 3- to 10-mm, slightly raised, pink-brown, sometimes scaly, hard growths that retract beneath the skin surface during attempts to compress and elevate them with the thumb and index finger ( Figs. 20.33 to 20.40 ![]() ). Multiple dermatofibromas (i.e., more than 15) are very rare but have been reported with systemic lupus erythematosus, with and without immunosuppressive therapy. Over 80% of patients with eruptive multiple dermatofibromas have an underlying immune-mediated disorder such as myasthenia gravis, Sjögren syndrome, atopic dermatitis, pemphigus vulgaris, HIV, psoriasis, sarcoidosis, and cutaneous T-cell lymphoma. Dermatoscopic examination shows a central, white, scar-like patch and a delicate pigment network at the periphery (see Fig. 20.35 ).
). Multiple dermatofibromas (i.e., more than 15) are very rare but have been reported with systemic lupus erythematosus, with and without immunosuppressive therapy. Over 80% of patients with eruptive multiple dermatofibromas have an underlying immune-mediated disorder such as myasthenia gravis, Sjögren syndrome, atopic dermatitis, pemphigus vulgaris, HIV, psoriasis, sarcoidosis, and cutaneous T-cell lymphoma. Dermatoscopic examination shows a central, white, scar-like patch and a delicate pigment network at the periphery (see Fig. 20.35 ).
Treatment.
Some patients object to the color of the lesion and therefore request excision. These lesions are most commonly found on the lower legs, where elliptic excisions closed with sutures may result in wide, unsightly scars. An alternative is to shave the brown surface with a no. 15 surgical blade and allow the wound to granulate and reepithelialize. The healed area remains hard because a portion of the fibrous tissue has remained. The brown color may reappear in some lesions. Conservative cryosurgery may also eliminate the color and part of the tumor.
Hypertrophic Scars and Keloids
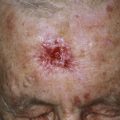
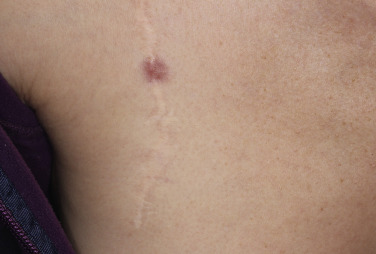
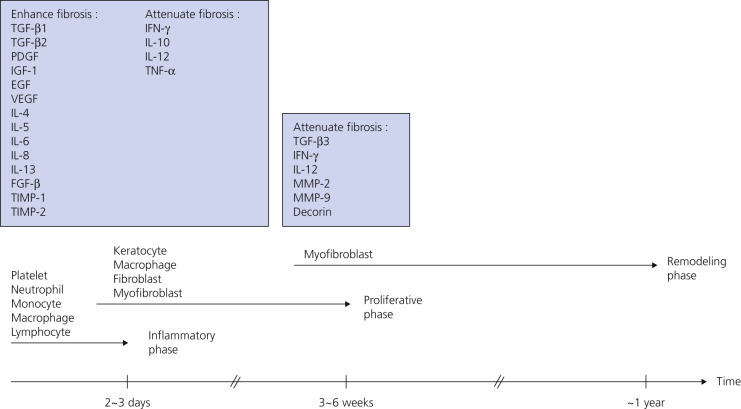
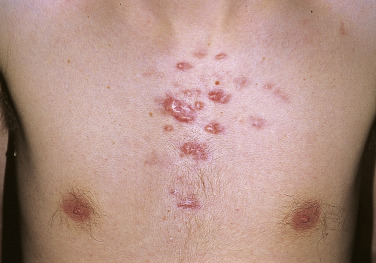
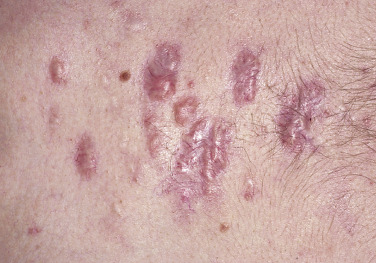
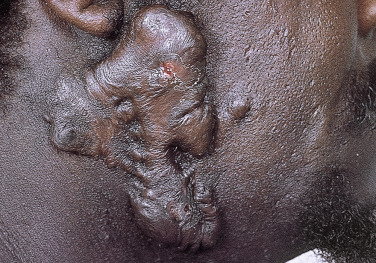
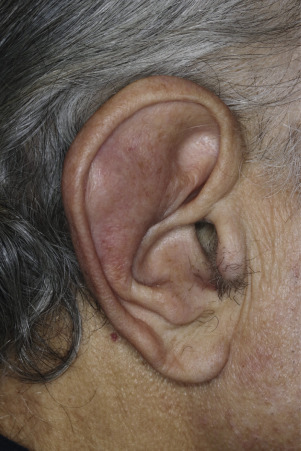
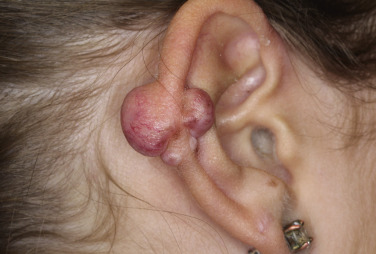
Injury or surgery in a predisposed individual can result in an abnormally large scar. The mechanism of hypertrophic scars and keloids has not been completely elucidated, but involves an alteration in normal wound healing (inflammatory, proliferative and remodeling phases) ( Fig. 20.41 ). A hypertrophic scar is inappropriately large but remains confined to the wound site and in time regresses; a keloid extends beyond the margins of injury ( Figs. 20.42 to 20.47 ![]() ) and usually is constant and stable without any tendency to subside. Hypertrophic scars may regress with time and occur earlier after injury (usually within 4 weeks); keloidal scars may begin later, even years after the event. There are histologic differences between hypertrophic scars and keloids. Large collagen bundles occur in keloidal scars but not in hypertrophic scars. Keloids are often symptomatic, and complaints arise because of tenderness, pain, and hyperesthesia, particularly in the early stages of development. Keloids are most common on the shoulders and chest, but they may occur on any skin surface. Black people are more susceptible and sometimes develop facial keloids. Some patients with cystic acne of the back and chest form numerous keloidal scars.
) and usually is constant and stable without any tendency to subside. Hypertrophic scars may regress with time and occur earlier after injury (usually within 4 weeks); keloidal scars may begin later, even years after the event. There are histologic differences between hypertrophic scars and keloids. Large collagen bundles occur in keloidal scars but not in hypertrophic scars. Keloids are often symptomatic, and complaints arise because of tenderness, pain, and hyperesthesia, particularly in the early stages of development. Keloids are most common on the shoulders and chest, but they may occur on any skin surface. Black people are more susceptible and sometimes develop facial keloids. Some patients with cystic acne of the back and chest form numerous keloidal scars.
Treatment.
There is no routinely effective therapy for all keloids. Many modalities reduce mediators of inflammation and may be used in the active treatment and prevention of keloids ( Table 20.1 ). The standard of care for keloids is intralesional triamcinolone, excision, and destruction.
| Categories | Modalities | Suggested Mechanisms | Use |
|---|---|---|---|
| Prophylaxis | Tension-free closure |
|
|
| Taping or silicone sheeting |
|
| |
| Flavonoids |
|
| |
| Pressure therapy |
|
| |
| Treatment (current) | Corticosteroids |
|
|
| Scar revision |
|
| |
| Cryotherapy |
|
| |
| Radiotherapy |
|
| |
| Laser therapy |
|
| |
| 5-Fluorouracil |
|
| |
| Treatment (emerging) | MSC * therapy |
|
|
| Fat grafting |
|
| |
| Interferon |
|
| |
| Human recombinant TGF-β3/TGF-β1 or 2 neutralizing antibody |
|
| |
| Botulinum toxin type A |
|
| |
| Bleomycin |
|
|
* MMPs, matrix metalloproteinases; MSC, mesenchymal stem cell; SMAD, the fusion of Caenorhabditis elegans Sma genes and the Drosophila Mad; TGF, transforming growth factor.
Intralesional Steroid Injections.
Proposed mechanisms of action include inhibition of collagen synthesis, inhibition of keloid fibroblast growth, and degeneration of fibroblasts. Fresh, small, and narrow lesions are treated with intralesional injections of corticosteroids every 2 to 4 weeks. Early keloids have softer proliferating connective tissue and are more likely to improve with intralesional injections than are older, inactive lesions. When the lesion shrinks to near the skin surface, the frequency and concentration of injections should be decreased to avoid skin atrophy and telangiectasia. Intralesional triamcinolone acetonide (Kenalog) 10 to 40 mg/mL is adequate for most small lesions; a 27- or 30-gauge needle on a Luer-Lok is used. The 40-mg/mL concentration is preferred for most patients . A commonly used program is to use triamcinolone acetonide given with four injections at 1- to 4-week intervals, and is dependent on lesion surface area (1 to 2 cm 2 , 20 to 40 mg per course; 2 to 6 cm 2 , 40 to 80 mg; 6 to 12 cm 2 , 80 to 120 mg; with courses repeated if necessary).
To distribute the suspension evenly, the triamcinolone should be injected while continuously advancing the needle. Particles of steroid that have not been properly dispersed remain visible as white flecks in the scar tissue. The pressure of the injection should be firm until the lesion blanches. Light cryosurgery before the injection facilitates the process. Nitrogen is applied briefly for 2 to 4 seconds until the skin frosts. The keloid is injected 10 to 15 minutes later. This allows better dispersal of the steroid and minimizes deposition into surrounding normal tissue. Intralesional steroids also result in symptomatic relief.
Surgery and Intralesional Steroid Injections.
Surgical excision removes the bulk of the scar and has the potential to replace a broad-based scar with a thin scar. Surgical removal alone is associated with a 55% to 100% recurrence rate, but better results are realized when intralesional steroids are used following surgery. A typical treatment program involves injecting triamcinolone acetonide 10 to 40 mg/mL into the wound edges after excision. Treatment of the healed site is repeated at 2- to 4-week intervals for 6 months.
Silicone Gel Sheeting.
The effectiveness of silicone gel sheeting (see Formulary) and other occlusive dressings in treating keloidal and hypertrophic scars is uncertain. Limited studies suggest that some beneficial effect may exist. It is claimed that these dressings can prevent keloids from recurring after surgery. Silicone gel sheeting is soft, self-adhesive, and semi-occlusive. The mechanism of action is unknown; it might act as an impermeable membrane that keeps the skin hydrated. Sheets are used to cover the entire scar for periods of at least 12 hours each day, and ideally 24 hours for at least 2 months. Beneficial effects are not related to pressure.
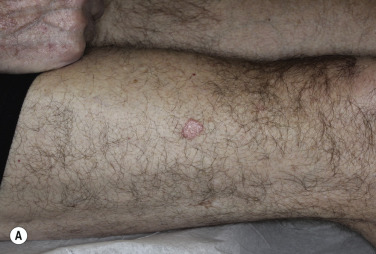
Keratoacanthoma
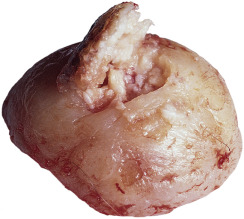
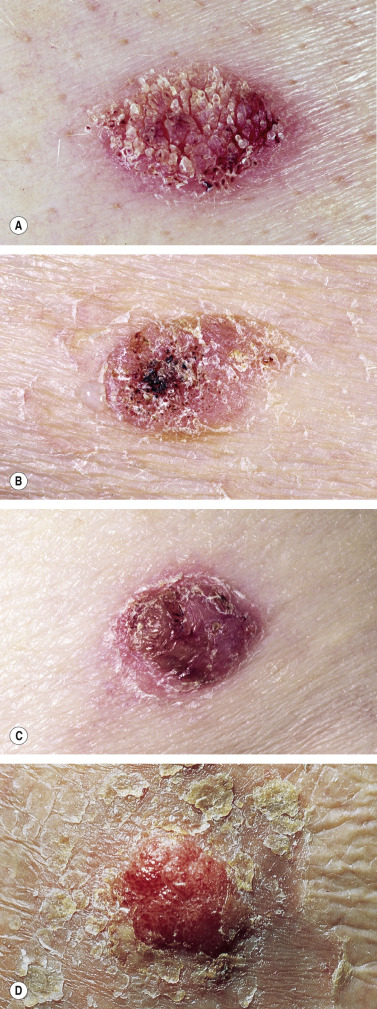
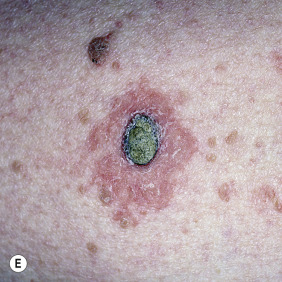
Keratoacanthoma (KA) is a relatively common, benign, epithelial tumor that was previously considered to be a variant of SCC. The etiology is unknown. Human papillomavirus-DNA sequences have been detected in lesions. It is a disease of the elderly (mean age, 64 years) with an annual incidence rate of 104 per 100,000. It is not associated with internal malignancy. There may be a seasonal presentation (summer and autumn) of KA that suggests that ultraviolet radiation has an acute effect on the development of KA. Moreover, KAs are more common in patients who have defects in ultraviolet radiation DNA repair mechanisms (xeroderma pigmentosum). KAs may develop in sites of previous trauma, even minor trauma, such as bites, razor cuts, venous puncture, and surgical procedures. Skin disorders with breaks in the epidermis such as dermatitis, psoriasis, lichen planus, prurigo nodularis, and rosacea have been associated with KAs. There are reports of KAs occurring in tattoos, especially red ink tattoos (mercury sulfide). In point of fact, medical treatments such as laser, chemical peels, cryotherapy, imiquimod, and photodynamic therapy that result in skin barrier disruption and inflammation may predispose to KAs in susceptible individuals. KAs may form 1 to 2 months after the inciting event. KAs and KA-like SCC occur with BRAF inhibitor (sorafenib, vemurafenib) treatment.
Most cases are the “crateriform” type, which grow rapidly and then undergo spontaneous regression. Less than 2% belong to the rare destructive variants with no regression and persistent invasive growth. These are referred to as KS marginatum centrifugum and mutilating KAs and can lead to severe defects; they are very rare.
Muir–Torre syndrome is a rare autosomal dominant genodermatosis that is characterized by the presence of at least one sebaceous gland tumor and a minimum of one internal malignancy. Keratoacanthomas have been noted in 23% of patients. The most commonly associated neoplasms are colorectal (61%) and genitourinary (22%).
Clinical Presentation.
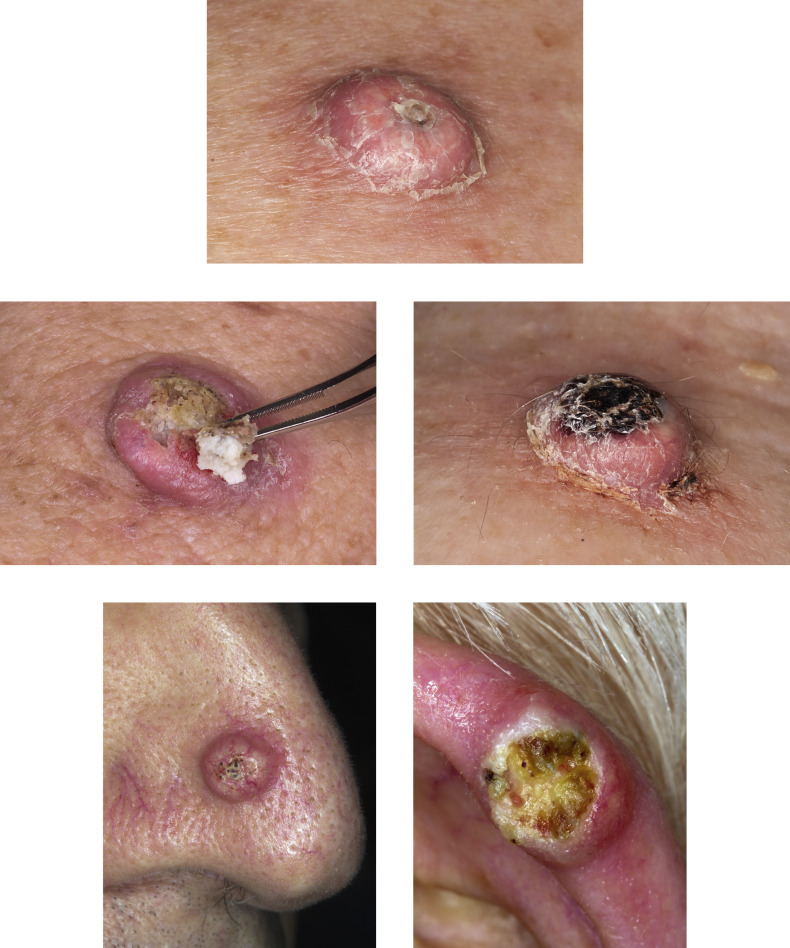
KA begins as a smooth, dome-shaped, red papule that resembles molluscum contagiosum. In a few weeks the tumor may rapidly expand to 1 or 2 cm and develop a central keratin-filled crater that is frequently filled with crust ( Fig. 20.48 ). The growth retains its smooth surface, unlike SCC. When untreated, growth stops in approximately 6 weeks, and the tumor remains unchanged for an indefinite period. In the majority of cases it then regresses slowly over 2 to 12 months and frequently heals with scarring. The limbs, particularly the sun-exposed hands and arms, are the most common site; the trunk is the second most common site, but KA may occur on any skin surface, including the subungual and anal area. On occasion, multiple KAs appear, or a single lesion extends over several centimeters. These variants resist treatment and are unlikely to undergo spontaneous remission. Familial KAs have been reported.
Differentiating Squamous Cell Carcinoma From Keratoacanthoma.
Squamous cell carcinoma (SCC) and KA are sometimes difficult to distinguish by histopathologic examination, since cytologic features are similar in both tumors. Many of the criteria commonly used for the differential diagnosis of SCC and KA are not reliable. In the future, gene expression technology (e.g., microarray chips) may aid in the differentiation of KA from SCC, since SCC has more chromosomal instability and markedly different gene expression than KA. At the present time, atypical or difficult cases should be considered and treated as SCC.
Treatment.
There is no advantage to waiting for spontaneous regression to occur, since most KAs ultimately heal with scarring. KAs are treated surgically or medically. KAs may recur.
Surgery.
Electrodesiccation and curettage or blunt dissection ( Figs. 20.49 and 20.50 ) (see Chapter 27 ) is efficient and effective for smaller lesions. Excision is effective for large tumors.