Abstract
Reconstruction of the skin is challenging and rewarding to the dermatologic surgeon. When second intention or side-to-side linear closure is not possible, more complex movement of local or distant skin in the form of cutaneous flaps is necessary to repair a surgical defect. The basic terminology, physiology, and types of local flaps will be discussed in this chapter.
Keywords
flap, axial pattern flap, random pattern flap, advancement flap, rotation flap, transposition flap, interpolation flap
The reconstruction of wounds has long been a challenge to surgeons. Whereas some wounds may be left to heal by second intention or closed in a simple side-to-side linear fashion, others require more complex movement of local or distant tissue to restore functional and anatomic relationships, and to optimize the cosmetic outcome.
Surgeons have used skin flaps to repair wound defects for centuries. The term “flap” was derived from the Dutch word “flappe” during the 16th century. “Flappe” referred to something that was fastened by one side and hung broad and loose. As early as 700 bce , the Sushruta Samhita first documented a technique of reconstructing a large nasal tip defect with a cheek flap. Since that time, surgeons’ knowledge of biology, anatomy, and physiology has greatly increased. This understanding allows us to significantly decrease the likelihood of surgical complications such as hemorrhage, hematoma, infection, and flap failure. This chapter addresses some basic principles of flap reconstruction including flap design, construction, classification, and practical tips.
Basic Terminology
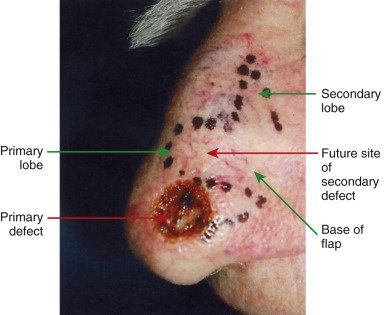
A skin flap is a construct typically consisting of skin and subcutaneous tissue with a partially intact vascular supply that is transferred or repositioned from an adjacent or more distant donor site to a recipient site. The recipient site is called the primary defect and, in dermatologic surgery, is usually the wound resulting from the removal of a cutaneous tumor ( Fig. 2.1 ). The secondary defect is defined as the wound that is created by cutting, lifting, or sliding the flap to fill the primary defect (see Fig. 2.1 ). The base of the flap is the area that remains attached to the skin adjacent to the defect. It is this base, also known as the pedicle of the flap, that contains the vascular supply necessary for initial flap survival. The tip of the flap is the portion of the flap furthest from the flap’s base.
Flaps Defined by Blood Supply
There are many classification schemata for reconstructive flaps. One common method of defining flaps is to determine the flap’s perfusion sources. Flaps may be categorized based on vascular supply as musculocutaneous, fasciocutaneous, axial, or random. A musculocutaneous flap includes muscle tissue in the base of the flap, whereas a fasciocutaneous flap includes only the fascial covering of the muscle in the pedicle. An example of a musculocutaneous flap used in dermatologic surgery is the Keystone flap, which relies on musculocutaneous and fasciocutaneous vascular perforators taken together with venous and neural connections within the flap’s base, which may increase the perfusion, and thus the reliability, of any flap.
The most common flaps used in dermatologic surgery, composed of primarily of subcutaneous tissue and skin (and sometimes superficial muscular fascia), are axial and random perfusion flaps. Axial flaps (also called arterial flaps) have a relatively large-diameter, named artery present within the pedicle of the flap, which directly provides nutrients to the full extent of the flap. This named artery typically lies in the subcutaneous layer superficial to the muscle fascia. On the face, the most commonly used axial flap is the paramedian forehead flap, which depends on the supratrochlear artery (and occasionally branches of the supraorbital artery) for survival. A random flap, the most commonly used flap design in reconstructive dermatologic surgery, depends on the small, unnamed perfusion plexus of highly anastomotic vessels in the dermis to provide nutrients to the flap. Because the arterial input of random pattern flaps is much less predictable, attention to proper flap design and delicate surgical technique is required to ensure surgical success.
Blood flow is the most crucial factor for flap viability. Vascular perfusion pressure is a description of the force of blood flow through a vessel. Perfusion pressure and blood flow in the body’s vessels are analogous to water flowing from a faucet through a garden hose. The greatest pressure and highest flow will be at the faucet head. As water travels farther away from the faucet, its pressure and flow decrease in strength. To maintain sufficient flow to the distal tip of the hose, the perfusion pressure within the hose must exceed the resistance of the hose. Similarly, in the human body, the perfusion pressure of any vessel that supplies blood and nutrients to the skin must exceed the capillary resistance in order to maintain vessel patency and preserve continued blood flow. A critical pressure is required to maintain patency of capillaries. Below this certain pressure, the capillaries will close, and insufficient blood will be supplied to the distal portions of the flap. The greater the distance from the feeding artery or arteriole, the lower the perfusion pressure will be ( Fig. 2.2 ). Thus beyond a certain distance, the flap tip will no longer receive blood nutrients, and it will thus necrose.
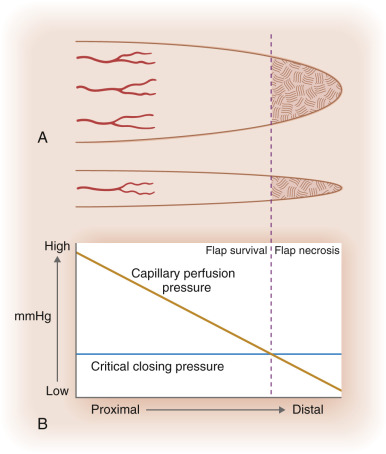
An understanding of perfusion pressures has dictated that flaps be designed with appropriate flap length to pedicle width ratio. Until 1970, it was widely believed that the viable length of a flap was directly proportional to the width of the base. Surgeons believed that in order to double the length of the flap, one would simply need to double the width of the flap base, thereby including a sufficient number of vessels in the pedicle to sustain the flap tip. Many felt that there was no ultimate limit to flap length. In 1970 Milton disputed this conventional hypothesis by publishing his research on axial flaps in the pig model. He discovered that “flaps made under the same conditions of blood supply survive to the same length regardless of width.” Daniel and Williams confirmed Milton’s research on axial flaps and further studied survival of random pattern flaps. Although they concluded that “an increase in width did not result in an increased length of survival,” their data showed a trend toward wider pedicles allowing greater flap length survival. Studies by Stell in a pig model and confirmed when extrapolated to humans suggested that viable flap length was not directly proportional to pedicle width, and that beyond a certain pedicle width, greater recruitment of random vessels cannot support the flap tip (see Fig. 2.2 ). Clinical experience with random flap survival has echoed this survival trend.
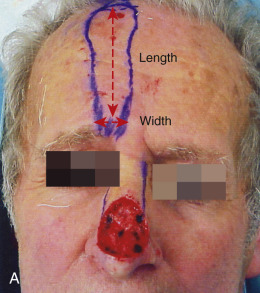
Overall, perfusion pressure limits the ultimate length of both axial and random flaps. The greater the perfusion pressure in the flap’s pedicle, the longer the flap can be without undergoing necrosis. Moreover, the greater the perfusion pressure at the flap’s base, the more narrow the pedicle may be. Arteries always have greater perfusion pressures than distal arterioles and capillaries. Accordingly, the longest viable flaps with the narrowest bases (largest length-to-base ratios) are arterial flaps. Therefore a paramedian forehead flap that uses the supratrochlear artery may be at least four times as long as the flap’s pedicle width (at least a 4 : 1 ratio; Fig. 2.3 ). Musculocutaneous flaps have the next greatest blood supply in the pedicle, followed by fasciocutaneous and random flaps in descending order.
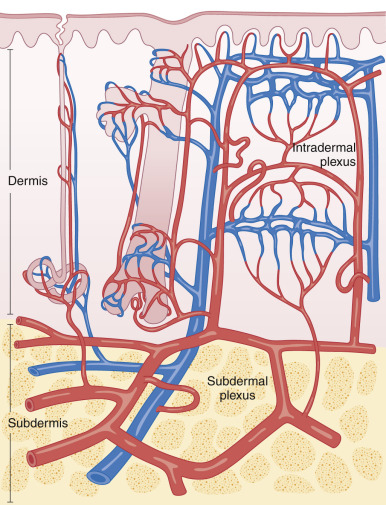
The commonly used random pattern flaps are largely supported by the redundant subdermal vascular plexus in the skin. Fig. 2.4 highlights the innate vasculature of the skin, upon which flap survival depends. The deeper, muscular-based arteries supply the subdermal plexus, which subsequently perfuses the intradermal plexus. The intradermal vasculature alone is usually unable to support tissue viability, due to low perfusion pressures and blood flow within these distal capillaries. However, the subdermal plexus, found in the mid to superficial subcutaneous fat, contains both arterioles and capillaries with sufficient perfusion pressure to sustain tissue viability following flap movement. This anatomy is critical to understand when undermining (dissecting under the flap and its surrounding area to allow tissue movement) a cutaneous flap. If undermining is performed superficial to the subdermal plexus (i.e., within the dermis), the flap has a significantly increased chance of undergoing tissue ischemia.
The perfusion pressures of random cutaneous flaps vary with the flaps’ locations on the body. As soon as a random pattern flap is incised and raised, there is a significant decrease in the perfusion pressure to the distal, disrupted skin. Fortunately at normal skin temperature, the amount of blood flowing through the facial skin is 10 times greater than that needed to supply the skin’s basic metabolic needs. Other areas of the body are not as well supplied with this redundancy of blood supply. A general rule is that the more distant the surgical flap is located from the heart, the less the perfusion pressure will be. Hence a flap on the leg should be designed with a smaller flap length-to-pedicle width ratio when compared with a flap on the face.
As previously discussed, there is a point at which no matter how many extra vessels are recruited at the base, the distal perfusion pressure will be less than the critical closing pressure of the arterioles and capillaries (see Fig. 2.2 ). When the perfusion pressure is less than this intravascular resistance, the flap receives diminished nutrients and necrosis can ensue. Generally, random cutaneous flaps of the face with a maximum length-to-base ratio of 3 : 1 survive. On the trunk and legs, the maximum length-to-base ratio is considered to be 2 : 1. An axial flap on the face may have a 4 : 1 or greater ratio depending on the arterial supply of the pedicle. Remember that these ratios are only guidelines. Patient-related factors must be taken into account when contemplating the feasibility and design of a flap. In addition to location of the body, local perfusion pressures can be altered by a variety of factors such as tobacco use, history of radiation to the area, medical comorbidities (i.e., atherosclerosis, high or low blood pressure, arrhythmias), and use of vasoconstrictors. In addition, flap design must take into account variations in skin laxity based on location, age, and comorbid conditions.
Flap Physiology
Flap survival is dependent on various factors, including blood flow, angiogenesis and vascularization, edema, wound closure tension, postoperative complications such as hematoma/seromas, and infection. Prior to the first incision, the flap skin is fully vascularized and viable using the definition of normal skin. Once the flap is raised, it is immediately ischemic, since the normal vessels supplying that skin are cut and the flap now depends on decreased circulation from the collateral vessels. A flap is always initially viable, since the skin can survive up to 12 to 13 hours of avascularity at 37°F. An ischemic flap can survive even longer, since the blood flow needed to sustain skin is only 2 to 8 cc per 100 g per minute, and normal flow to the skin is 10 times greater than this minimum. Thus Meyers was correct when he commented that a fresh flap is always ischemic but viable.
Once incised and relocated, the flap receives its nutrients from both the cutaneous pedicle and the base of the primary defect. Sufficient blood flow through the base of the flap is essential in the initial 24 to 48 hours after the initial flap creation. In both axial and random flaps, blood flow immediately drops as the flap is elevated. For axial flaps, microvascular flow actually increases to a level greater than the preoperative state within 5 hours. Flow in random pattern flaps, however, starts to improve differentially for up to 4 weeks. Marks used the rat model to show that flow improved on a gradient; flow increased within 14–16 hours to the skin closest to the pedicle, within 24–48 hours to the skin 1 cm distal to the pedicle, and within 96 hours 3 cm distal to the pedicle. All sites recovered approximately 30% of their blood flow per day, with the proximal most portion recovering full blood flow by the end of 7 days and the most distal tip by the end of 14 days. Until 14 days, recovery of blood flow occurred on a gradient that depended on the distance of the skin from the base of the flap. Microvascular flow grew to higher than preoperative levels from 14 to 21 days, and then gradually returned to baseline during the fourth week. The opening of collateral vessels appears to allow this sequential recovery of blood flow. However, there appears to be a limit on how fast these collaterals can open.
This time-dependent opening of collateral vessels may be partially explained by arteriovenous (AV) shunts. These shunts control blood supply to the capillary network that supplies the flap. There are pre-AV shunt sphincters under the control of the sympathetic nervous system. When the flap is incised and undermined, local sympathetic nerve fibers are disrupted and release catecholamines. As a result, there is local vasoconstriction for up to 48 hours, by which time the nerve’s supply of norepinephrine is exhausted. Once sympathetic tone is relaxed, the blood flow to the capillary collaterals is increased to help supply the flap with nutrients. However, this effective sympathectomy cannot fully explain why random flaps have a graded flow recovery. Upon incision, the entire flap should have equal catecholamine release and subsequent equal flow recovery. Other humoral factors such as prostaglandin release may come into play.
Local tissue conditions resulting from surgical trauma also decrease flap perfusion and subsequent survival. Following any local injury, the inflammatory cascade releases the powerful vasoconstrictor thromboxane A2. In addition, free radicals are released, causing direct injury to the flap. Finally, the edema inevitably associated with surgical trauma causes further capillary vessel resistance by increasing manual compression on the skin’s smaller caliber perfusion sources. In addition, fluid collection under the flap in the form of a postoperative hematoma or seroma can further decrease blood flow. All of these negative factors decrease perfusion to the flap and can threaten flap survival.
Conversely, the flap may also benefit from surgical trauma. Relative local hypoxemia and increased levels of metabolic by-products induce opening of precapillary sphincters, thereby promoting increased local blood flow. Moreover, adhesion molecules, such as E-selectin, are activated following exposure to released coagulation cascade molecules such as endotoxin, interleukin-1, and tumor necrosis factor alpha. These adhesion molecules recruit molecules including neutrophils to the flap to clear debris and anabolic waste products. Finally, ischemic tissue attracts endothelial progenitor cells, which allow for the ingrowth of new vascular channels to supply the flap. Flap survival is dependent on the balance of all these factors, ultimately influencing pedicle blood flow.
The nascent flap not only receives nutrients from the pedicle, but it also gains nutrition from the base of the primary defect through angiogenesis, revascularization, and neovascularization. Within the first two days of flap placement, a fibrin layer develops below the flap and provides a suitable environment for angiogenesis. Endothelial cells and macrophages release angiogenic cell factors important in neovascularization, the local growth of new blood vessels into the surgically manipulated skin. Neovascularization is seen as early as 3 days in the rat model, and at 4 days in rabbit and pig models. In staged, pedicled flaps in humans, revascularization adequate for division of the flap pedicle has been demonstrated by the seventh postoperative day. This new vascular growth works in conjunction with the preexisting collateral vessels to nourish the flap.
Tissue edema, wound closure tension, and infection also negatively affect flap blood supply and survival. Although none of these factors can solely lead to necrosis of a well-vascularized flap, each can contribute to further ischemia in a marginally perfused flap. Postoperative tissue edema places external force on small capillaries, resulting in increased capillary resistance. Thus there must be greater perfusion pressure at the pedicle to counter this resistance and ensure flap tip survival. Recent studies in the rat model revealed that significant postoperative edema will not solely cause flap necrosis. However, it can be an additive factor, along with high wound closure tensions and/or infection.
Closing wounds under large amounts of tension can place undue vascular stress on the wound edges and tip of the flap. It is typically recommended that one undermine 2 to 4 cm, or 50% to 100% of the defect width, beyond the wound edge to decrease wound tension. Undermining beyond this distance may be detrimental, since there may be unnecessary vascular compromise, more bleeding, and greater dead space, all of which can lead to surgical complications. High closure tension leads to dehiscence and wound edge necrosis, but it does not usually lead to entire flap necrosis. Wound infection can cause partial or complete flap necrosis. With local infection, there is release of toxic free radicals and greater tissue edema. Infection can also lead to vessel thrombosis. In addition to local tissue destruction and vascular compromise, collagen production and deposition are hindered. Therefore flap adhesion to the wound bed and overall tensile strength are affected. Overall, there are many factors involved in the initial period of wound healing that are critical to the flap’s survival. Predictably acceptable flap results depend on proper flap design, gentle operative technique, and the avoidance of surgical complications.
Flap Biomechanics
In addition to understanding the skin’s vascular supply and physiology, the surgeon must also appreciate the unique biomechanical properties of the skin if flap surgery is to be successful. All materials have characteristic biomechanical properties: stress, strain, creep, and stress relaxation. In regard to skin, stress is the force applied per cross-sectional area, and strain is the change in length divided by the original length of the given tissue, to which a given force is applied. The stress–strain relationship of skin shows that skin, unlike some other materials, is not truly elastic ( Fig. 2.5 ). As a small amount of stress (or tension) is placed on the skin, there is a corresponding change in the skin’s length (strain). At a certain point on the stress–strain curve (see zone III in Fig. 2.5 ), even a large amount of applied force will not result in further incremental skin stretch. This nonelastic property of skin is mainly due to its structural constituents—collagen and elastin. In relaxed skin, collagen is randomly oriented, and elastin is loosely wrapped around and attached to multiple points on the collagen bundles. When a small amount of force is initially applied to skin, the elastin network is first deformed and the skin is easily lengthened (see zone I in Fig. 2.5 ). With sun exposure and intrinsic aging, there is a progressive decrease in the functional elastic fiber network. As a result, the stress–strain curve is shifted to the right so that when applied to aged or sun-damaged skin, less applied force results in greater lengthening. As continued force is applied, the collagen fibers begin to reorient parallel to the direction of the force (rapid transition of the curve; see zone II in Fig. 2.5 ). At the point where the elastin and collagen fibers are maximally stretched, even a large amount of force will only minimally stretch the skin (see zone III in Fig. 2.5 ). Thus the skin is not truly elastic at all levels of applied tension.
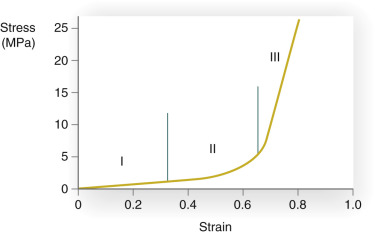
Creep refers to the increase in strain seen when skin is under constant stress. When the skin is held at sufficient tension, elastic fibers fragment and collagen fibers will align parallel to the applied force. As a result of this reorganization of the elastic and collagen fiber networks, interstitial fluid will be displaced and can be seen with the naked eye. Creep typically begins to occur within several minutes of constant force application. While skin demonstrates elastic properties with low loads, skin exhibits “stress relaxation” and creep when larger forces are applied for longer periods of time. Stress relaxation occurs when skin is held under constant tension. In this case the amount of force (stress) required to maintain this tension decreases with time. The skin’s relaxation under stress is closely related to its ability to increase in length when placed under a constant stress (the creep phenomenon). In the case of a cutaneous flap, if held steadily at high tension for 5 to 10 minutes, the skin will lengthen and relax. Surgeons with an understanding of this property may use an intraoperative pulley stitch to take advantage of this mechanical property. As a result of intraoperative creep, a smaller flap can be used to fill a larger defect, but the potential reliance upon secondary motion around the surgical defect must be recognized. When skin is held at a constant tension for several days, stress relaxation occurs. There is a decrease in stress due to an increase in skin cellularity and the permanent stretching of skin components. In clinical practice, serial excision and tissue expansion use the principles of creep and stress relaxation. When a large lesion is partially excised and reconstructed under tension, the skin relaxes and lengthens, allowing further staged excisions.
Understanding the stress–strain curve is essential for optimal wound closure. When reviewing reconstructive options, it is important to avoid any excessive stress on the defect edges that would force the involved skin into the third part of the stress–strain curve; this involves minimizing tension on the wound closure. Unfavorably high wound closure tensions limit recruitment of skin laxity and can be detrimental to wound healing. Specifically, high tension may lead to wound edge necrosis, wound dehiscence, and cosmetically unacceptable scars (i.e., widened, atrophic, or hypertrophic scars). Wound edge tension may be minimized by appropriate undermining; however, care must be taken to understand and minimize the underlying mechanism of stress or restriction. For example, a galeotomy may be used to release galeal fascial restraint and allow for additional movement of the skin and subcutaneous tissue on the scalp; careful dissection of the skin from the underlying orbicularis muscle in the periocular area creates movement of the skin to allow for tension redistribution away from the free margin; and separating the vertical connections within the subcutaneous tissue of the cheek from the underlying superficial muscular aponeurotic system (SMAS) drastically reduces tension on closures and allows for substantial movement of tissue for closure of large cheek defects.
In addition to understanding the role of tension reduction on wound closure, it is important to identify situations where tension redistribution is required. Specifically, when side-to-side linear closure is not possible due to excessive tension or stress on the primary closure, or when linear wound closure abuts a free margin or crosses a cosmetic subunit boundary, the vector of tension may need to be redistributed in the form of a rotation or transposition flaps, discussed later. Overall, by taking advantage of the above-mentioned mechanical properties of skin and designing appropriate flaps, the surgeon takes tension off the wound edges, and returns the skin to the first or second zone of the stress–strain curve. Understanding these principles, as well as mastering techniques for tension reduction and redistribution, allows the closure of skin flaps under low or moderate tension. As a result, wound edges can survive, and a good cosmetic result can be obtained.
Flaps Defined by Movement
In addition to classifying flaps by their vascularity, flaps are often also categorized by their primary movement directions. The three basic flap movements are advancement, rotation, and transposition. With advancement flaps, the major motion of the flap is largely along a single linear direction toward the primary defect ( Fig. 2.6 ). Rotation flaps are those flaps that are rotated in an arc or curvilinear fashion along a pivot point to fill the adjacent primary defect without crossing any intervening skin ( Fig. 2.7 ). A transposition flap is incised and lifted over intact skin and then placed into the wound ( Fig. 2.8 ). A distinct type of transposition flap is called the interpolation flap. An interpolation flap is a flap, potentially with a predictable, axial vascular supply, that involves two steps or stages. The first stage of the flap is transposing the tissue into the wound with the pedicle overlying the intervening skin. The second stage is separating the flap from its origin and therefore dividing its pedicle.
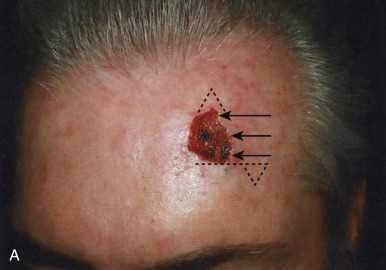
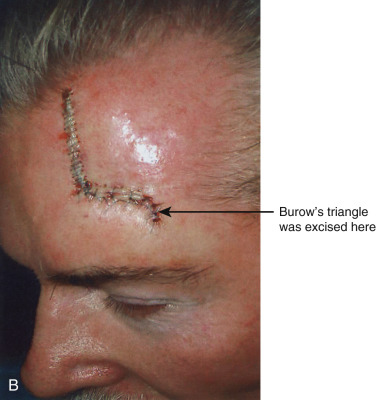
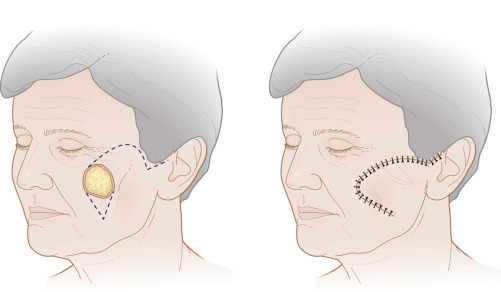
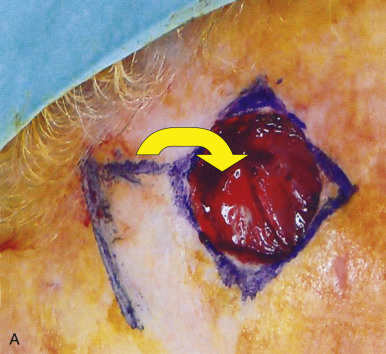
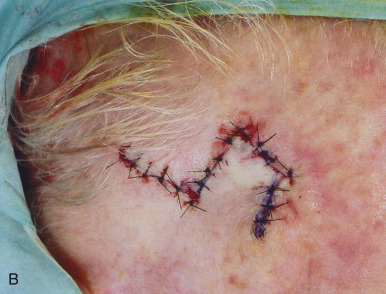
In addition to classifying flaps by their unidirectional motion, some authors, noting the inherent limitations of such schemata, categorize flaps as sliding or lifting flaps. Sliding flaps include advancement and rotation flaps, where tissue is pushed into the primary defect. A lifting flap is the typical transposition or interpolation flap, in which tissue must be lifted over normal skin to fill the wound. These more general classifications recognize that very few flaps move tissue along a single, predictable path. For example, most rotation flaps also incorporate a significant degree of tissue advancement, and vice versa.
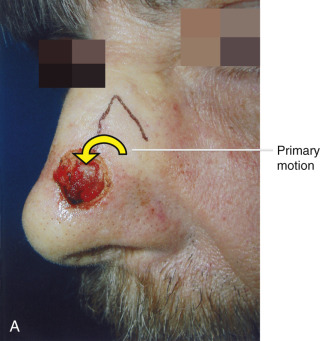
As alluded to previously, when discussing the motion of these flaps, the surgeon must understand the tension vector created by the flap’s closure. This vector depends on the primary and secondary motion of the flap. The primary motion of the flap is the tension placed on the flap tissue as it is moved to fill the wound (primary defect) ( Fig. 2.9 ). The secondary motion, created by the movement of the flap into the defect, is the force placed on the tissue surrounding the primary defect (see Fig. 2.9 ). The combined force created by the primary and secondary motions causes a new tension vector that must be anticipated by the surgeon when deciding which flap can best be used for repair. This final tension vector varies, depending on the flap chosen, and may be appreciated when placing the key stitch. The key stitch is the stitch placed to initially move the flap and accurately place it into the primary defect. If not properly planned, the flap’s final tension vector may cause undesirable pulling on free margins such as the eyelid, alar rim, and mouth.