Creating pleasing breast aesthetics after an explantation can be challenging, especially when performed with a total capsulectomy. The necessity of a capsulectomy for a variety of breast issues is not completely agreed on and can increase adverse events, patient morbidity, and cost of the operation and often impairs the final aesthetic result. In this article, an algorithm for the management of explantation patients provides a framework for treatment of the capsule and the breast to optimize outcomes.
Key points
- •
In explantation, a primary decision is on determining whether the capsule is friend or foe.
- •
A total capsulectomy easily is accomplished with subglandular breast implants but imposes more significant risks when attempted in an adherent capsule along the chest wall in submuscularly placed implants.
- •
A total capsulectomy is the gold standard for the treatment of capsular contracture, but partial capsulectomies or capsulotomies are performed in many patients because reduction in evidence-based recurrence of capsular contracture appears mostly to be associated with implant exchange and pocket exchange, not total capsulectomy.
- •
En bloc capsulectomy is solely indicated for breast implant associated–anaplastic large cell lymphoma and not breast implant illness.
- •
Preservation of the capsule simplifies the surgical procedure, provides support and circulation for the overlying breast and additional coverage anteriorly, and improves the ability to perform substantial fat grafting during the explantation procedure.
Introduction
Whereas breast implants had little regulation during the 1960s, the following decade was characterized by growing public concerns of the risks associated with breast implants, such as rupture and removal, resulting in increased Food and Drug Administration (FDA) regulation. In the early 1990s, with concerns over implant safety and an association with autoimmune diseases, a 14-year moratorium on the use of silicone gel implants ensued and saline breast implants became the standard in the United States. This resulted in numerous studies demonstrating safety and efficacy of silicone gel breast implants, with subsequent FDA approval of silicone breast implants again for both cosmetic and reconstructive purposes in 2006. ,
Whether for breast reconstruction or cosmetic breast augmentation, it is well known that breast implants are not lifetime devices. Common reasons for explantation include implant rupture and capsular contracture; however, recent discussions pertaining to the safety of silicone breast implants are fueling increasing demand for explantation. Rates of explantation have increased dramatically over the past few years, making explantation 1 of the top 10 most commonly performed plastic surgery procedures in the United States. , Given that these procedures are becoming increasingly popular, this review serves as a comprehensive look at the indications for explantation, capsulectomies, and the correction of the breast aesthetics without the use of implants.
Indications for explantation with or without capsulectomy
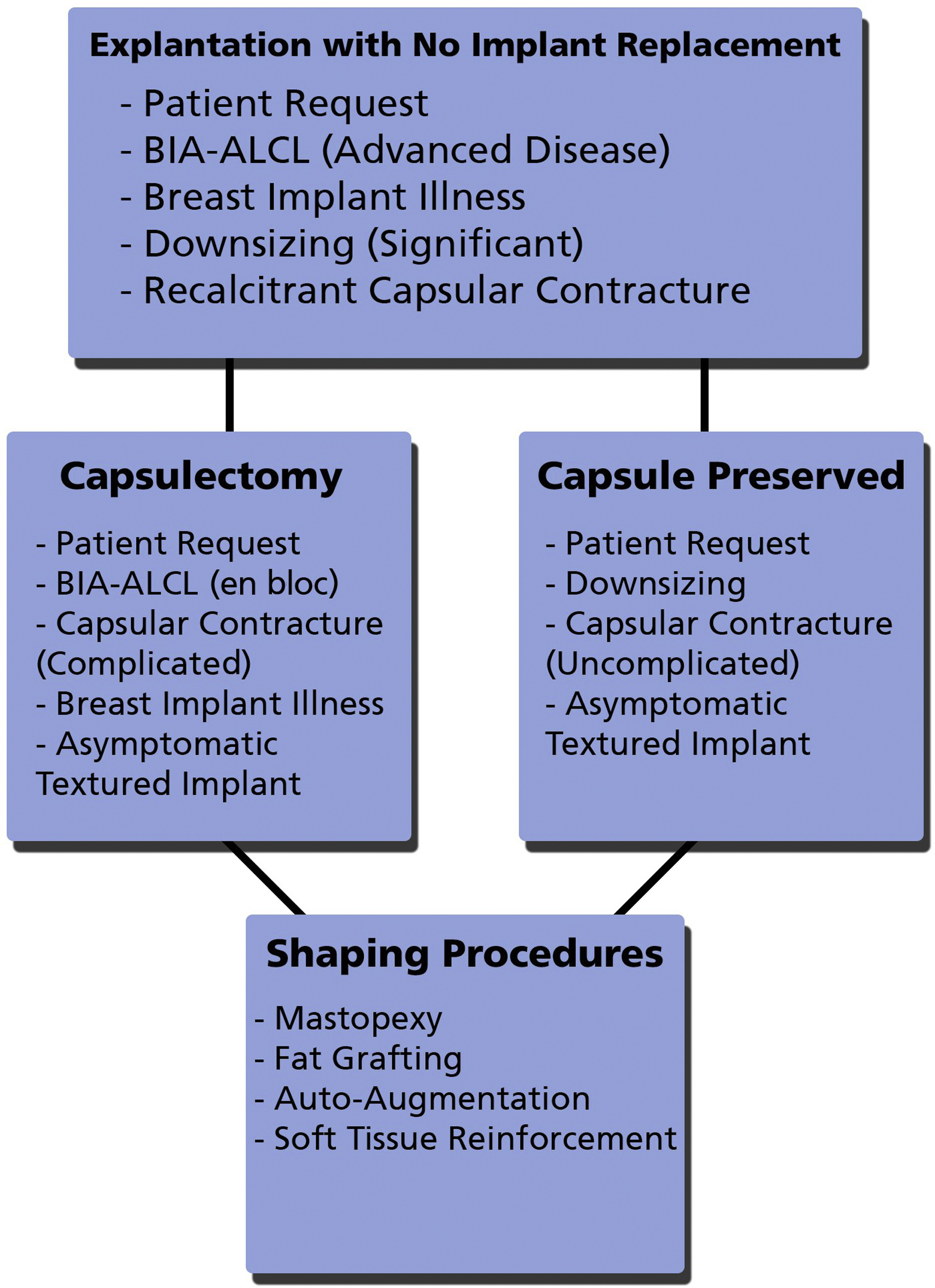
The indications for implant removal are numerous and have included not only complications associated with breast implants, such as implant rupture, capsular contracture, and patient desire for an aesthetic change, but also more recently, issues, such as concerns of breast implant illness (BII), treatment of breast implant associated (BIA)–anaplastic large cell lymphoma (ALCL), and risk reduction by explantation of textured devices. When performing an explantation, a decision then must be made as the necessity or appropriateness of capsule removal, whether total, partial or en bloc. This ultimately requires a significant amount of discernment by the plastic surgeon because the ultimate surgical approach is dependent on a variety of factors designed to ensure the surgery addresses not only the aspects related to the disease process but also adequately the issues related to the patient’s concerns. An algorithm for the management of explantation, capsulectomy decisions, and reconstruction provides a framework for the decision making in these challenging cases ( Fig. 1 ).
Capsular Contracture
Capsular contracture has for decades been the most common complication associated with aesthetic and reconstructive breast surgery. , Although the exact mechanism or etiology still is unclear, many etiologies have been implicated; the most feasible include the hypertrophic scarring, the infectious theory, or potentially both. The hypertrophic theory implicates either blood or seroma fluid surrounding the breast implant that induces the capsular contracture, and the role of a myofibroblast in this process has been implicated by some authors. , The infectious theory has the most supporting evidence and has become the predominant theory, implicating low-level bacterial contamination and the formation of a biofilm around the breast prosthesis, introduced either at the time of implantation or during seeding of the implant space from a transient bactermia. , The exact etiology most certainly is multifactorial and may be a combination of the prevailing theories in some patients. The significance of this pathologic process cannot be overstated because capsular contracture consistently has been the leading indication for revision after placement of a breast implants, ranging from 15% to 30%, and accounts for approximately 50,000 reoperations annually. , ,
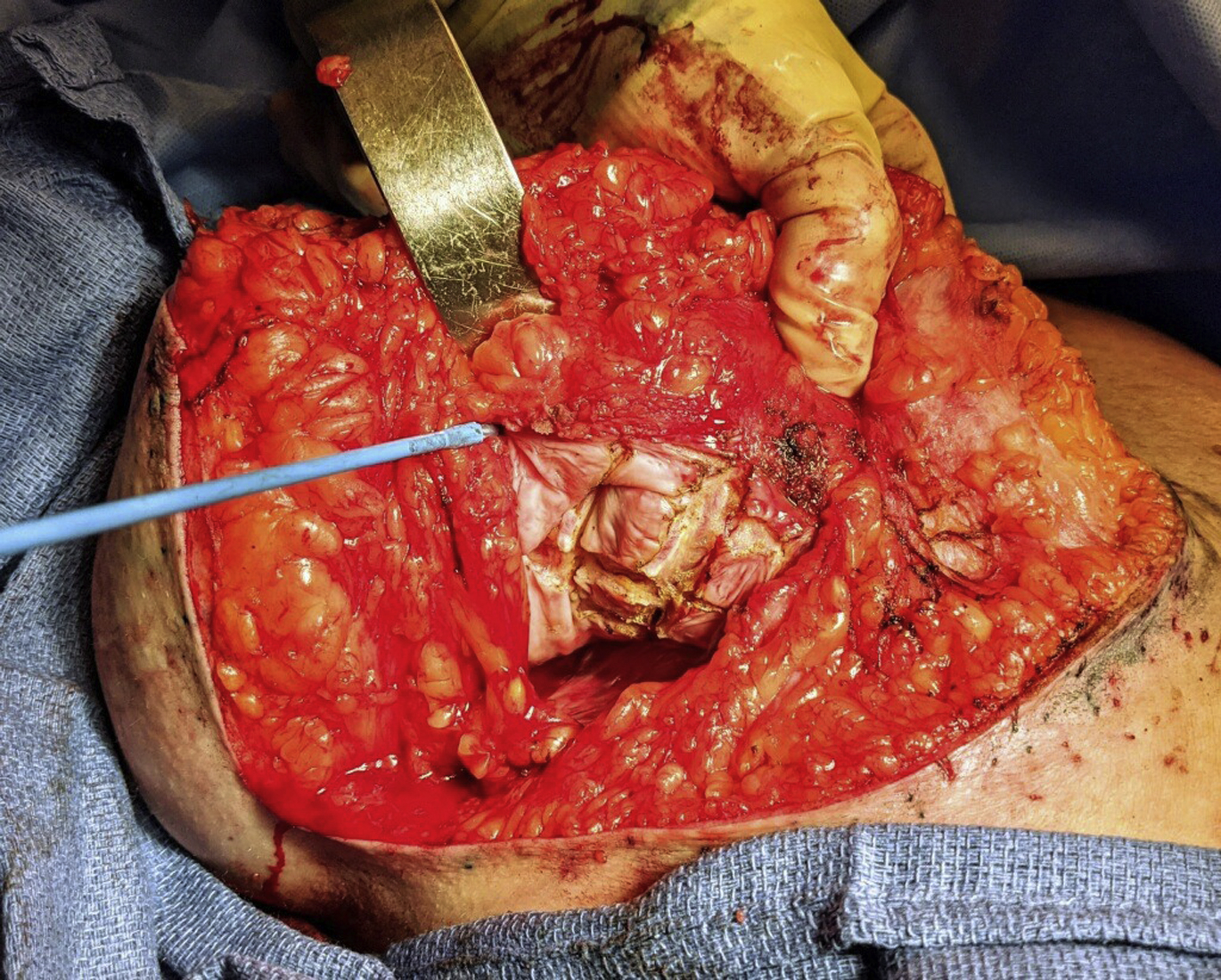
For implant patients presenting with breast pain and deformity constituting Baker grade III or IV capsular contracture, treatment is indicated. It has long been accepted as conventional wisdom that total capsulectomy is the most effective treatment of capsular contracture, demonstrating superiority over partial capsulectomy and capsulotomy. The concept of a total capsulectomy stems from the infectious etiology theory, reasoning that if bacteria and biofilm on the capsule are the etiology of the capsular contracture, complete removal of the capsule and its contamination is advisable. Likewise, it was demonstrated in 1 study that a partial capsulectomy was associated with 50% of patients having recurrence with a partial capsulectomy versus 11% of patients with a total capsulectomy. A recent systematic review concerning the evidenced-based treatment of capsular contracture concluded, however, that actually site change, implant exchange, and addition of acellular dermal matrix (ADM) were the only aspects of treatment most definitely associated with lower capsular contracture recurrence rates and that evidence to definitively demonstrate the benefit of a total over a partial capsulectomy was lacking. Thus, in the face of a capsular contracture, explantation or implant exchange with pocket exchange is the best treatment option in nearly every case. When capsular contractures are recalcitrant and have failed previous attempts at correction, an explantation or implant exchange with concomitant ADM may be the most prudent approach. In patients with subpectoral implants, a complete capsulectomy is associated with an increased risk of chest wall injury and pneumothorax because the capsule is dissected off the chest wall. In some cases, the capsule is excised anteriorly but retained posteriorly to minimize soft tissue trauma with the new implant placed anterior to the dissected capsule ( Fig. 2 ).
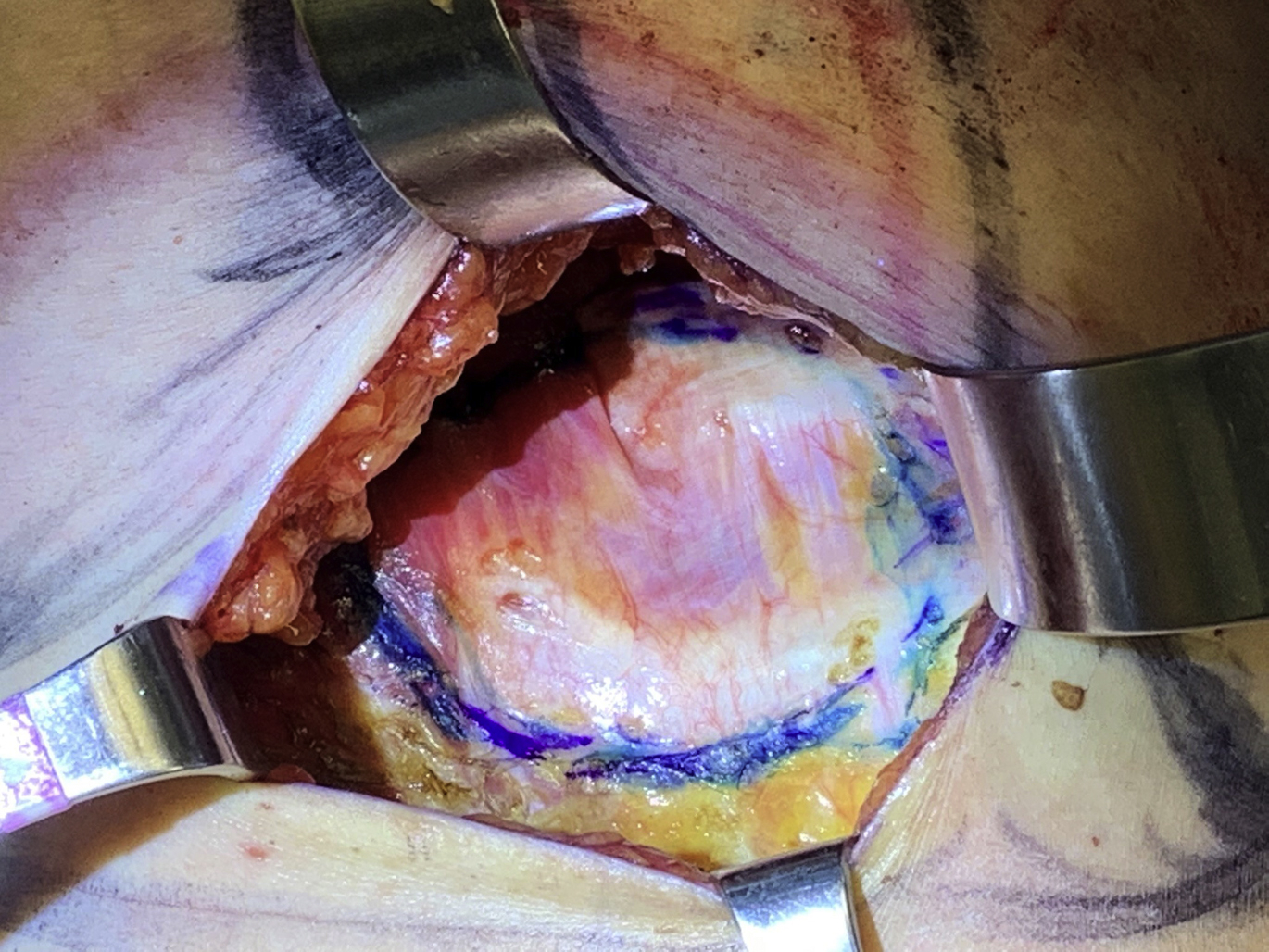
There also has been good evidence that the use of a neosubpectoral pocket with placement of ADM significantly reduced the risk of capsular contracture recurrence, but this requires maintaining the capsule. Finally, a retrospective review by Swanson concluded that capsulotomy alone for capsular contracture demonstrated good results with low rates of recurrence, although this recurrence rate was reported at 22.7%. If a capsulotomy is done only to treat the capsular contracture, the logic for implant exchange is less clear.
A capsular contracture itself is not necessarily an indication for a capsulectomy if no implant is to be replaced and sometimes can be used advantageously, as in the simultaneous implant exchange with fat grafting (SIEF) procedure. The capsule can provide support for the soft tissue envelope, provide enhanced circulation to the overlying breast, and create an inner lamellae for fat grafting.
Implant Rupture
Rupture of a silicone implant is an indication for implant removal. In many cases, rupture of the device can occur with no other outward sign that the integrity of the shell has been lost. Such silent ruptures of silicone gel devices often remain intracapsular and often are asymptomatic and detected only incidentally on mammogram or magnetic resonance imaging (MRI). Implant rupture rates in primary breast augmentation vary widely based on surgeon and manufacturer, but recent data indicate a range from 8.7% to 24.2% at 10 years. Diagnosis of silicone gel implant rupture usually is made using MRI or ultrasound. Due to its lesser expense, there is increasing interest in the use of high-resolution ultrasound (HRUS), in particular in making it available in the physician’s office, providing convenience for the patient, and assisting the surgeon in providing a more complete evaluation of the implant. This is an emerging area of interest that will have more clarity in the near future but has for many surgeons become the preferred approach over MRI for the initial evaluation of a possible rupture, with HRUS sensitivity and specificity approaching or exceeding those of MRI (Please see Bradley P. Bengtson’s article, “ Instructional Course: Officed Based High Resolution Ultrasound for the Plastic Surgeon ,” in this issue). Importantly, a high concordance has been demonstrated between ultrasound and MRI for the detection of implant rupture, potentially obviating MRI evaluation for implants if ultrasound demonstrates a rupture. The new FDA guidelines include a high-definition ultrasound at 5 years to 6 years for asymptomatic patients every 2 years thereafter and an MRI for symptomatic patients or if results of ultrasound in asymptomatic patient are equivocal. ,
Although recommendations concerning the appropriate usage of ultrasound, MRI, and mammography for rupture detection at present are still not fully agreed on, there is agreement that a ruptured implant is an indication for removal, with or without exchange to a new device. Whereas the capsule generally does not need to be removed for a rupture, a significant rupture with silicone imbedded in the capsule preventing complete removal with washings mandates removal of the capsule as well.
Silicone Implant Illness or Breast Implant Illness
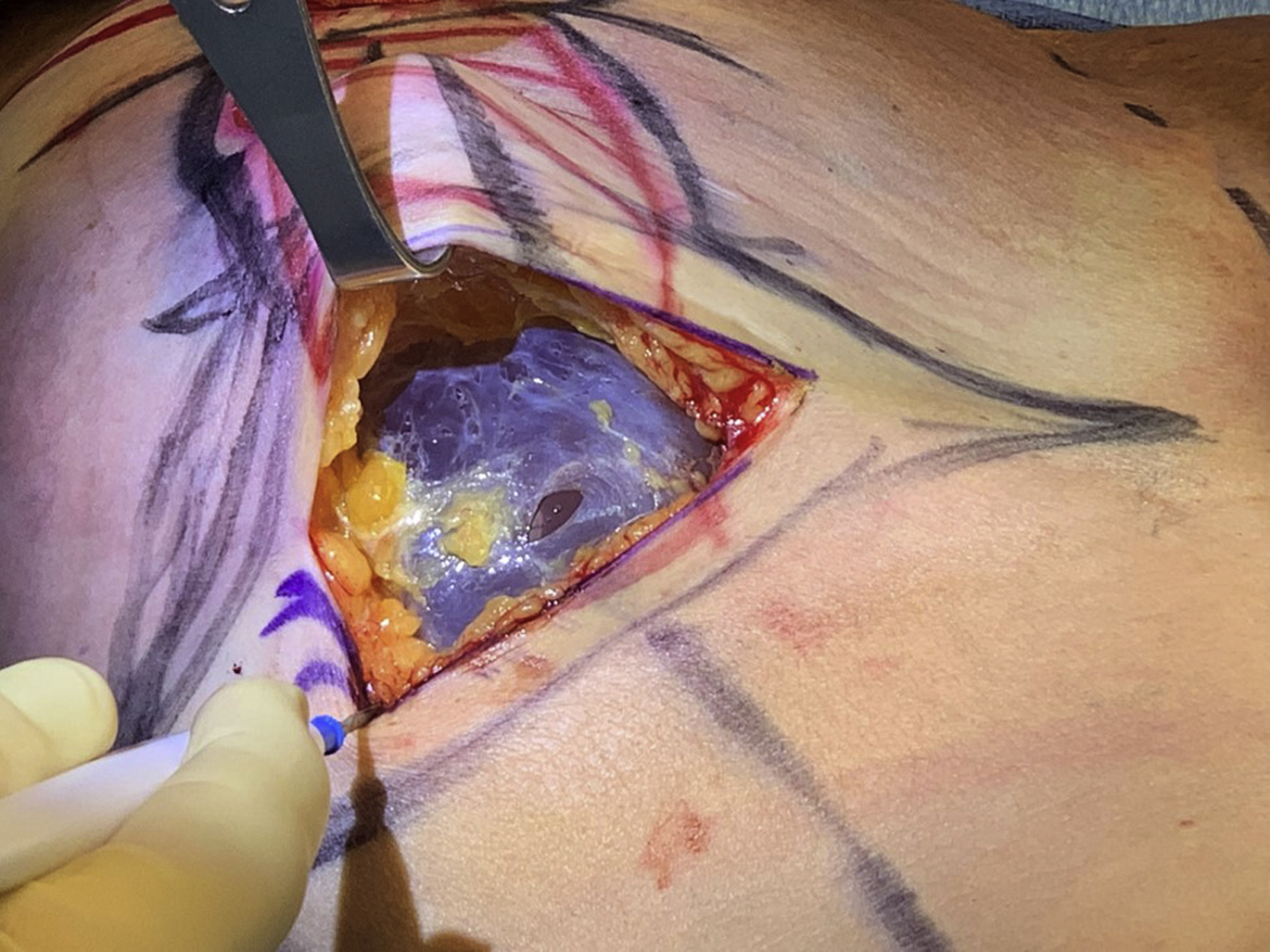
Increasingly, some patients have concerns that their implants are the cause of myriad symptoms referred to in the news and social media by the term, breast implant illness . This implant illness is characterized by a nonspecific and generalized constellation of symptoms that includes malaise, fatigue, fibromyalgia, headache, and other patient-reported complaints. In 1997, Peters and colleagues reported on 100 patients presenting for explantation during the silicone moratorium. These patients were divided into 3 groups. Group 1 did not meet diagnostic criteria for rheumatic or autoimmune disease and demonstrated a greater than 80% improvement in physical symptoms and 93% improvement in psychological well-being after explantation. Group 2 had rheumatic but not autoimmune disease and had an initial improvement (placebo effect) but with recurrence in symptoms at 6 months to 12 months. Group 3 had a diagnosed autoimmune disease and showed no improvement of symptoms or autoantibody levels and went on to suffer from the autoimmune disease. A review of 240 explantation patients by Melmed in 1998 found that patients reported a decrease in flulike symptoms, depression, and fatigue after implant removal. Rohrich and colleagues, in 2000, demonstrated that in patients who underwent explantation, improvements were documented for musculoskeletal symptoms, general pain, and mental health. De Boer and colleagues conducted a review of 23 case reports and cohort studies dating from 1994 to 2014, in which 75% of a combined 622-patient sample noted improvements in their overall health and systemic illnesses after the removal of their breast implants. Explantation alone, however, helped alleviate symptoms in only 16% of patients who were definitively diagnosed with an autoimmune disorder.
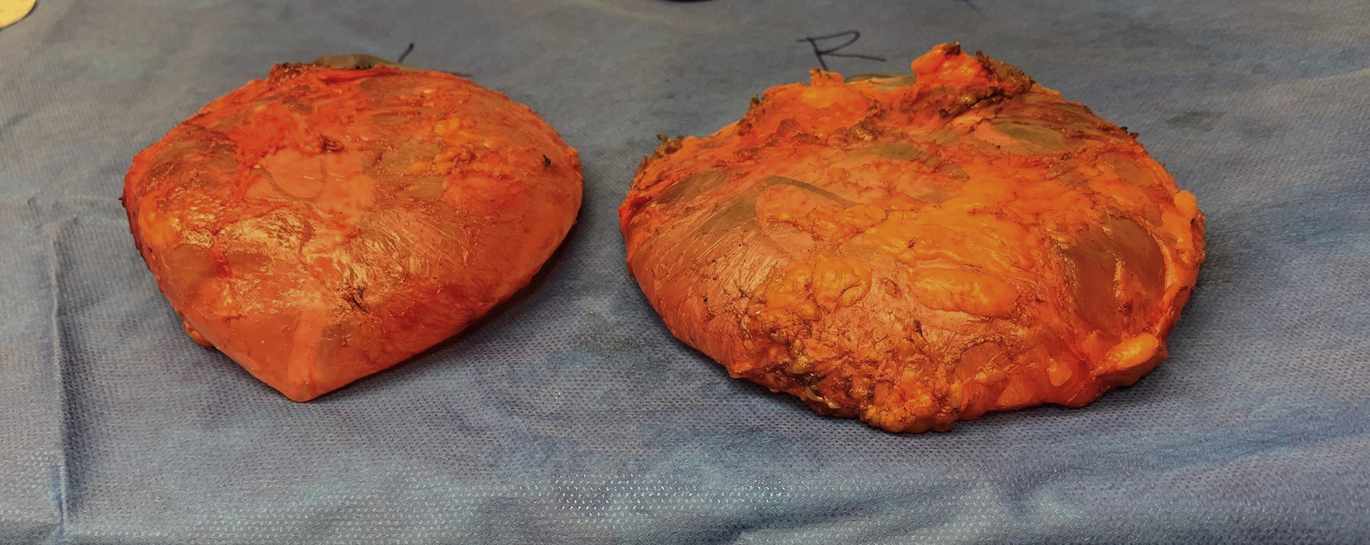
Improvement in symptoms after explantation is variable, and breast implants alone may not be the cause of any specific autoimmune or systemic illness. Currently, there are no dependable studies demonstrating a cause-effect relationship between silicone implants to what is now termed BII. No test for BII currently exists and it may be that BII is a manifestation of the hyperstimulation of the immune system that represents an allergic-type response to breast implants, but this remains conjecture. Patients seeking explantation for BII have saline-filled implants as often as silicone-filled implants, which seems to exonerate the fill substance as the cause but potentially not the shell because both types of implants are silicone-based. Nevertheless, patient complaints about BII should be taken seriously, and symptomatic patients should be given the option of implant removal with or without capsulectomy ( Fig. 3 ). In BII-concerned patients, whereas a near-total capsulectomy or total capsulectomy may be a reasonable option based on the clinical scenario, there is no indication for an en bloc capsulectomy, and the use of this term and this type of treatment should be discouraged and reserved for confirmed malignancy. If referring to the removal of the capsule and implants as an entire unit without entering the capsule, although often impossible in many clinical situations, the more appropriate term is, en masse removal ( Fig. 4 ). There are no evidenced-based data that support this approach as providing any additional benefit when treating patients concerned with BII, but this type of radical treatment may pose greater potential risk for complications, such as pneumothorax, in some patients.
Breast Implant–Associated Anaplastic Large Cell Lymphoma
BIA-ALCL is associated with textured breast implants and has been the focus of research and discussion in the plastic surgery community. The most common presentations include seroma, pain and/or swelling in the breast, capsular contracture, and a mass or lump around the implant. The current incidence of BIA-ALCL varies based on the type of implant. All patients should be counseled on the potential for BIA-ALCL when using textured devices and it should be included as a part of the informed consent. Patients should be counseled that BIA-ALCL is not breast cancer and often follows an indolent course and that removing the implants and surrounding capsules often is sufficient to achieve complete remission. The capsule, periprosthetic effusion, and any associated masses should be removed completely with the implants through an en bloc resection, which results in better overall survival and event-free survival outcomes compared with partial capsulectomy or no capsulectomy. , Whereas leaving a part of the capsule when performing a capsulectomy may be appropriate for other indications, such as capsular contracture or BII, when treating BIA-ALCL, a complete and total capsulectomy surrounded by a contiguous rim of healthy margin ( en bloc capsulectomy) is warranted. Incomplete removal and persistent margins are associated with recurrence and disease progression.
Asymptomatic Textured Implant Patients
Recently, textured implants have come under increased scrutiny due to their association with the development of BIA-ALCL, as discussed previously. This included a worldwide ban on the use of Biocell textured implants (Allergan Corporation, Dublin, Ireland) in 2019 due to their association with approximately 90% of the worldwide cases of BIA-ALCL. Thus, plastic surgeons increasingly are seeing patients with many questions concerning their textured implants and the appropriate management of their implants and capsules. This can be challenging, because a surgeon often is faced with trying to balance the concerns of the patient with the available evidenced-based information to support the management of these cases. When treating patients with textured implants who desire removal for risk reduction from BIA-ALCL in the future, there currently are no evidenced-based data to support a capsulectomy in an asymptomatic textured implant patient with a normal-appearing capsule at surgery, no matter the type of textured device implanted. When it can be removed easily, such as in a subglandular augmentation, removal of the capsule adds little to the morbidity and may prove beneficial. Likewise, if performing an explantation and a worrisome capsule is encountered with a mass, fluid, or abnormal thickening, the surgeon may deem it appropriate to remove the capsule with appropriate pathologic evaluation. Currently, without supporting data for a prophylactic capsulectomy in an asymptomatic textured implant patient, each surgeon ultimately needs to counsel patients on the best course of treatment with the understanding that a prophylactic capsulectomy may or may not reduce a patient’s risk of future BIA-ALCL, is associated with increased operative complexity and morbidity, often can impair the final aesthetic result, and is associated with a greater financial burden. In asymptomatic patients, simply proceeding with ongoing observation and evaluation, explantation alone, or implant exchange to a smooth implant without a capsulectomy each may be an appropriate treatment option, is less costly to the patient, and has a much lower risk profile. In addition, if capsule is left behind and a patient develops BIA-ALCL, these would include signs and symptoms of developing disease and, in this rare event, total capsulectomy or en bloc resection may be performed at this time. The true incidence of these situation requires additional research and, because of the low incidence of this disease, may be difficult to document.
Patient Preference
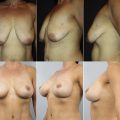
Unsatisfactory breast aesthetics due to age-related changes, pregnancy, weight gain or loss, or changes in public opinion concerning breast implants may result in a nonmedically related desire for explantation. As the body acceptance movement gains popularity on social media platforms and concern regarding the safety of silicone implants persists, an increase in the number of patients who seek breast implant removal is anticipated , (see Fig. 17 ).
Surgical management of the capsule
The surgical approach appropriate for each patient requires a significant amount of discernment by the plastic surgeon because myriad factors must be considered to ensure the surgery addresses not only the issues related to the disease process but also issues related to the patient’s concerns, whether evidence-based or based solely on personal perception.
The management of the capsule requires a thorough understanding of the indications for the explantation. The capsule may be considered friend or foe. The appropriateness as to whether capsule removal is indicated not always is clear and can be controversial based on the various indications. This has significant implications because a total capsulectomy can be challenging and can have a significant impact on surgical morbidity, with significant adverse events of severe hematoma and pneumothorax, financial burden, and ultimate aesthetic outcome reported.
If the capsule is calcified, has a mass present, or is embedded with silicone product, removal of the capsule is warranted. Additionally, patients concerned with BII desire that any retained silicone left behind by rupture, particle shedding, or gel bleed be removed as completely as possible and, therefore, are served best with removal of the entire capsule to ease concerns with inadequate treatment. A confirmed diagnosis of BIA-ALCL requires an en bloc resection.
Capsulotomy
Capsulotomy is the simplest, and safest, of the procedures used to manage the capsule in patients, although not considered the first-line treatment by most surgeons. Capsulotomies are performed by scoring the capsule radially and sometimes in a checkboard pattern to achieve expansion of the breast pocket. The dissection sometimes is carried into the breast tissue as well to allow for greater expansion ( Fig. 5 ). There are many advantages of performing a capsulotomy, including preservation of support for the overlying skin envelope when soft tissue coverage is thin, improved pocket control and support for the implant if an implant replacement is planned, and improved preservation of vascularity for the overlying breast skin soft tissue envelope, especially when a mastopexy is planned. Capsulotomies are not adequate in cases of BIA-ALCL, patients concerned with illness from breast implants, or complicated capsular contractures. These complicated capsular contractures may be calcified or have embedded silicone in the capsules or an associated capsular mass present. These require some form of capsulectomy ( Fig. 6 ).