Key points
• Augmentation, feminization, masculinization, and other adjustments to the cranium can be done with implants, osteotomies, hydroxyapatite bone cements, and autogenous bone grafts. Soft tissue can further be augmented with fat grafting, filler materials, or free tissue transfer.
• Virtual surgical planning has enabled the surgeon to engage in the custom fabrication of implants.
• Polymethylmethacrylate (PMMA) can be used as a low-cost material to fabricate custom prostheses intraoperatively.
• To minimize the risk of exposure, the incision sites should not overlap the implant directly.
• Implants should be fitted without gaps to the bone surface on which they are to be placed, and they should be secured with screws in the subperiosteal plane to prevent migration.
Introduction
Patients may present to craniofacial surgeons with aesthetic complaints regarding cranial deformities. Cranial defects have multiple possible etiologies: congenital, traumatic, iatrogenic, and cosmetic. It is important to consider both skeletal tissue and soft tissue in developing a treatment plan. Fortunately, the surgeon has many options at his or her disposal. This chapter discusses the various options for correcting cranial deformities.
Anatomy
The three layers that make up the cranium in the grown adult are the hard internal and external laminae and the middle layer, or diploe. The average thickness ranges from 5 to 7 mm but varies considerably, depending on location, patient age, and other individual factors, with the thickest area being the occipital region and the thinnest being the temporal region. The external and internal laminae have a layer of periosteum on their surfaces. The internal periosteum is the dura’s outermost layer, and in adults, it has very little ability to produce new bone.
The appearance of the upper third of the face is secondary to the frontal and temporal bones, which are posteriorly circumscribed by the coronal suture. The cranial bones behind the coronal suture are less likely to be noticed by the patient from an aesthetic standpoint and are usually only addressed in the case of infection, osteoradionecrosis, or the syndrome of the trephined (SoT).
The forehead portion of the frontal bone constitutes its vertical portions; it is typically convex in appearance. Located just on either side of the midline are the frontal eminences, above the supraorbital rims and the overlying superciliary arches, which represent the two sections of frontal bone that were previously separated by the metopic suture. The superciliary arches join at the midline at the glabella. These elevations correspond to the location of the frontal sinuses.
The supraorbital rim, which serves to protect the globe, forms the upper boundary of the orbit. Laterally, it borders the zygoma at the zygomatic arch. The supraorbital nerve exits via the supraorbital foramen, located one third of the distance from the medial-to-lateral supraorbital rim.
The frontalis muscle, supplied by the facial nerve, has no attachments to bone and is responsible for the raising of eyebrows. The orbicularis oculi muscle consists of both orbital and palpebral portions and originates from the nasal process of the frontal bone, frontal process of the maxilla, and anterior surface of the medial canthal tendon. It is supplied by the facial nerve and responsible for closing the eye. Smaller muscles include the corrugator supercilii, a small triangular-shaped muscle originating from the superciliary arch that draws the eyebrow downward through its connection to the skin of the medial eyebrow. The procerus originates from the nasal bones and inserts onto the medial eyebrow to draw the medial angle downward. Both the corrugator and the procerus muscles are supplied by the facial nerve. Finally, the temporalis muscle is a fan-shaped muscle arising from the temporal fossa deep to the temporal fascia and inserting onto the coronoid process of the mandible. Supplied by the mandibular division of the mandibular nerve, it assists in closing the jaw.
Patient evaluation
History
A thorough history should be taken to screen for cranial abnormalities. Most often, cranial abnormalities are secondary to congenital, traumatic, or oncologic causes. In the case of congenital abnormalities, it is important to discern any prior surgeries that have been performed and, if applicable, whether autologous or nonautologous reconstruction was done with either permanent or resorbable plates. If the patient has a history of reconstruction, it is important to know what donor sites were used. In the case of a traumatic etiology, it is important to determine the degree of injury, prior infections, and the method of treatment. If an infection was present, the physician should discover which pathogens were implicated and the details of antibiotic therapy (duration and type). When evaluating postoncologic defects, it is important to discern the patient’s remission status, need for prior radiation therapy and chemotherapy, and which method of reconstruction was utilized. It is vitally important to make sure that the patient is receiving continued cancer-specific screening and is free of malignancy before repeat reconstruction is performed. If active malignancy exists, negative margins should be achieved before reconstruction is considered.
To address the patient’s complaint, particularly for aesthetic correction of morphology already considered to be within normal range, it is vital to elucidate the patient’s understanding of his or her appearance so that the clinician’s goals are in alignment with the patient’s goals. The patient should be asked about the perceived aesthetic and nonaesthetic components of his or her face. Part of this discussion should delve into the patient’s cultural background, if relevant.
The surgeon should uncover what factors the patient feels are contributing to any deficiency in appearance. This includes questions about both soft tissues and the facial skeleton. Asymmetries should be noted during visits with the surgeon.
Differential diagnosis
It is important to know what medical factors are contributing to the patient’s condition and appearance. Continued osteomyelitis can lead to recurrent infection and sinus tracts. Hypersensitivity reactions to prior implants can result in inflammation, swelling, and discoloration. Prior graft resorption can lead to deficiency in the appearance of the skull. Prior radiation therapy can distort the cranial skeleton and also cause microvascular thrombosis. Other comorbidities may also alter the appearance of the face. Human immunodeficiency virus (HIV) medication–associated lipodystrophy can accelerate temporal wasting and buccal fat pad atrophy. Other conditions affecting the skull include facial nerve palsy, Parry-Romberg syndrome, and chronic steroid use.
Physical examination
The physical examination is most vital for the surgeon’s preoperative planning. It is important to examine the patient’s skeletal cranium, soft tissue quality, prior scars, and areas of previous trauma.
High-quality digital images help guide the discussion of the patient’s goals and expectations. Standardized preoperative photographs (at a minimum, six views: anteroposterior, right lateral, right oblique, left lateral, left oblique, and vertex views) are useful for treatment planning and medical documentation. Newer three-dimensional imaging software with graphic manipulation, such as Vectra, can allow for an informative discussion and facilitate agreement between the surgeon and the patient on realistic goals. Standardized preoperative photos also aid in legal documentation for all involved parties.
Imaging and other preoperative diagnostic evaluation
Preoperative imaging with computed tomography (CT) is almost always necessary before developing a treatment plan. This allows the surgeon to visualize bone quality and quantity, as well as the presence of foreign bodies, such as previously placed implants or fixation. These images also are necessary to fabricate custom implants using virtual surgical planning (VSP).
Indications and contraindications
Goals for cranioplasty include protection for the brain and achieving an aesthetic contour. Certain neurologic defects secondary to direct exposure of the brain resulting from pressure from the overlying soft tissues can also be reversed. Skull defects, particularly in the frontal and parietal areas, even small ones that are 2 to 3 cm in length, should be considered for repair because they can lead to obvious deformity; however, defects of the temporal and parietal areas may not need reconstruction.
Full-thickness cranial defects
Most frequently, cranioplasty is required after the loss of a craniotomy bone flap as a result of infection. Usually, the loss is caused by a combination of dehiscence and skin necrosis, resulting in exposure of the flap directly under the incision. This is exacerbated by the presence of dead space under the incision or foreign bodies—which include plates and screws often used in operative fixation. Not much is required to disrupt the incision. Even pressure necrosis due to lack of offloading can exert significant stress on the closure site. A possible solution here is to design large skin flaps so that incisions are located away from bone flaps and hardware to the extent possible.
Syndrome of the trephined
SoT is believed to be triggered by exposure of intracranial contents to the open atmosphere. It can present after craniectomy, manifest as a sunken calvarial skin flap, and is suspected to arise secondary to a dysregulated balance of intracerebral and external pressures. Patients present with reduced cerebral perfusion, impaired venous return, altered cerebrospinal fluid (CSF) flow, brain herniation, and other deformations, with subsequent metabolic changes and cortical dysfunction. , Neurologic sequelae include nonspecific symptoms, such as headache, as well as motor weakness and cognitive deficits affecting executive function and memory. Early cranioplasty within 6 to 12 months of craniectomy is an effective treatment for SoT and is associated with normalized CSF hydrodynamics and cerebral blood flow. ,
Revision cranioplasty
Delayed hypersensitivity reactions and even death have occurred years after alloplastic cranioplasty. , Patients with a history of alloplastic calvarial reconstruction presenting with subacute–chronic nonspecific neurologic and dermatologic symptoms, such as suboccipital headaches, swelling of the head, and diffuse rash, should be suspected and tested with wide-range patch testing for common alloplastic materials, including bone cement.
If revision cranioplasty is being sought for aesthetic reasons, the surgeon needs to ensure that the recipient site is well-vascularized, has adequate soft tissue, and is free of infection. Although autologous bone can be used, due to variable resorption over time, it is less ideal for purely aesthetic cranioplasty. The primary approach to aesthetic cranioplasty is to use calcium hydroxyapatite (HA) bone paste or a prefabricated custom implant. If cost is a concern, polymethylmethacrylate (PMMA) is an inexpensive, safe material that the surgeon can use intraoperatively to make a custom implant. These PMMA kits are used frequently in orthopedic surgery and are inexpensive. PMMA is a safe, biocompatible material for cranioplasty and can even be mixed with antibiotic powders or solutions to prevent infection.
Congenital abnormalities
Many congenital abnormalities caused by premature suture fusion, irregularities in branchial arch development, or genetic abnormalities require skeletal work, often earlier in the patient’s life. Some patients with fibroosseous diseases, such as fibrous dysplasia, will benefit from cranioplasty. Acquired deformities can contribute to the problem or may involve prior procedures, making repeat exposure slightly more difficult because of the presence of scar tissue. It is our preference to use autologous bone with resorbable plates, if possible, in the pediatric population and address any aesthetic concerns once growth is complete. In the growing patient, nonresorbable plates should be avoided because these plates will migrate from the outer lamina to the inner lamina with skull growth.
Preoperative planning
The surgeon should strive to achieve three goals: cerebral protection, cosmesis, and normalization of cerebral hydrodynamics and metabolism. However, complex cranial defects with a history of infection, radiation, and/or prior surgery require careful selection of the reconstructive timing and approach (alloplastic versus autologous versus combined) and means of soft tissue coverage.
Timing of surgery
Final cranioplasty is indicated when any infection has been cleared and risk factors for recurrence of infection have been minimized. Data show that waiting 1 year after clearance of acute infection reduces the risk of recurrence, although many practitioners feel that a period of 6 months is adequate. It is common for surgeons to perform serial debridement and other preparatory wound care during this period. Additionally, the patient’s condition, with regard to comorbidities, should be optimized for surgery. Communications from any sinus tracts to the planned graft or device should be eliminated, and the frontal sinus can be ablated or cranialized in this instance. A galea frontalis flap, or temporalis flap, or a free tissue transfer may be required at the time of cranioplasty to achieve adequate coverage. Tissue expansion may be needed to increase the amount of soft tissue available for coverage over the final reconstruction. Incisions should be planned away from the sites of grafting and hardware.
Virtual surgical planning with computer-aided design/computer-aided manufacturing
VSP has become an important tool in the aesthetic facial surgeon’s arsenal. VSP analyzes more data points during computer assessment of the skull and provides more accurate information to the surgeon compared with a steriolithographic cranial replica. As a result, VSP allows for objective, reproducible outcomes. The surgeon is able to better visualize intraoperative moves as a result of the 3D skull–dental model and to achieve cephalometric analytical accuracy. If a microvascular tissue transfer is necessary to cover a prosthesis that is anatomically ideal, the overall aesthetic result may be compromised. In these patients, intentionally designing a smaller prosthesis that could be placed under hair-bearing scalp skin may be aesthetically preferable to an anatomically correct implant covered by a hairless flap.
The computer-aided design/computer-aided manufacturing (CAD/CAM) technology was first introduced in the mid-1960s and reintroduced in 2001 by Mommaerts et al. for treatment of metopic synostosis and unicoronal synostosis. CAD/CAM has made it possible to reconstruct most defects, including larger ones, with nearly all alloplastic materials. Printed models of preoperative skulls and the desired postoperative shape can be used ahead of time to design complex structures of the final implant. The treatment of Gulf War veterans led to rapid advances in VSP with CAD/CAM technology. Excellent results of complex skeletal defects were achieved, leading to broad adoption and decreased cost for civilian patients.
Primary operative approach
During the surgery, the patient is positioned such that the entire skull and upper face are exposed so that the surgeon can use the contralateral normal side as a reference, when present. If that is not feasible, preoperative photographs and radiologic imaging are particularly useful.
Incisions
Coronal incision
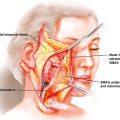
Coronal incisions can be placed at or posterior to the hairline, depending on the goals. If hairline lowering is desired, an incision at the hairline maximizes the amount of hair-bearing scalp that is removed. The incision can be extended to the ear and can even continue as a preauricular extension, if necessary. A maneuver that facilitates coronal flap reflection without a preauricular extension is an 8 to 10 mm cutback, which can be made to allow the coronal flap to be turned anteriorly while minimizing tension. Unless the goal is to advance the hair-bearing scalp, the incision should be made at least 3 to 5 cm posterior to the hairline. The appropriate dissection plane is under the galea to facilitate speed and ease of dissection in an avascular plane. The galea is continuous with the superficial temporal fascia, so this fascia will be raised in continuity with the galea. If the temporalis muscle is released from the parietal bone, it should be resuspended before closure. The coronal incision can be reused for any future surgeries.
Materials
Adhering to the principle of replacing “like with like,” calvarial reconstruction with autologous bone was historically considered the ideal reconstructive material for long-term success. Bone is attractive for its ability to revascularize, a property that alloplastic materials do not possess. For these reasons, vascularized bone is preferred for hostile wound beds, such as those previously infected or irradiated. However, some factors, such as a large defect size, or donor site availability, may preclude autologous reconstruction. Additionally, the osteoclast-mediated remodeling may result in up to 40% resorption and unpredictable final contours. , Contemporary alloplastic materials used for aesthetic cranioplasty have been shown to be nontoxic and durable. Alloplastic implants offer predictable structure and can be used for coverage of large defects that are not amenable to autologous reconstruction. Operative morbidity is also decreased with alloplastic augmentation because donor site harvest is obviated. However, alloplastic materials may elicit immune reactions, such as delayed hypersensitivity years after placement, and even death. As foreign bodies, they are at risk of transforming into infective nidi and perpetuating infection.
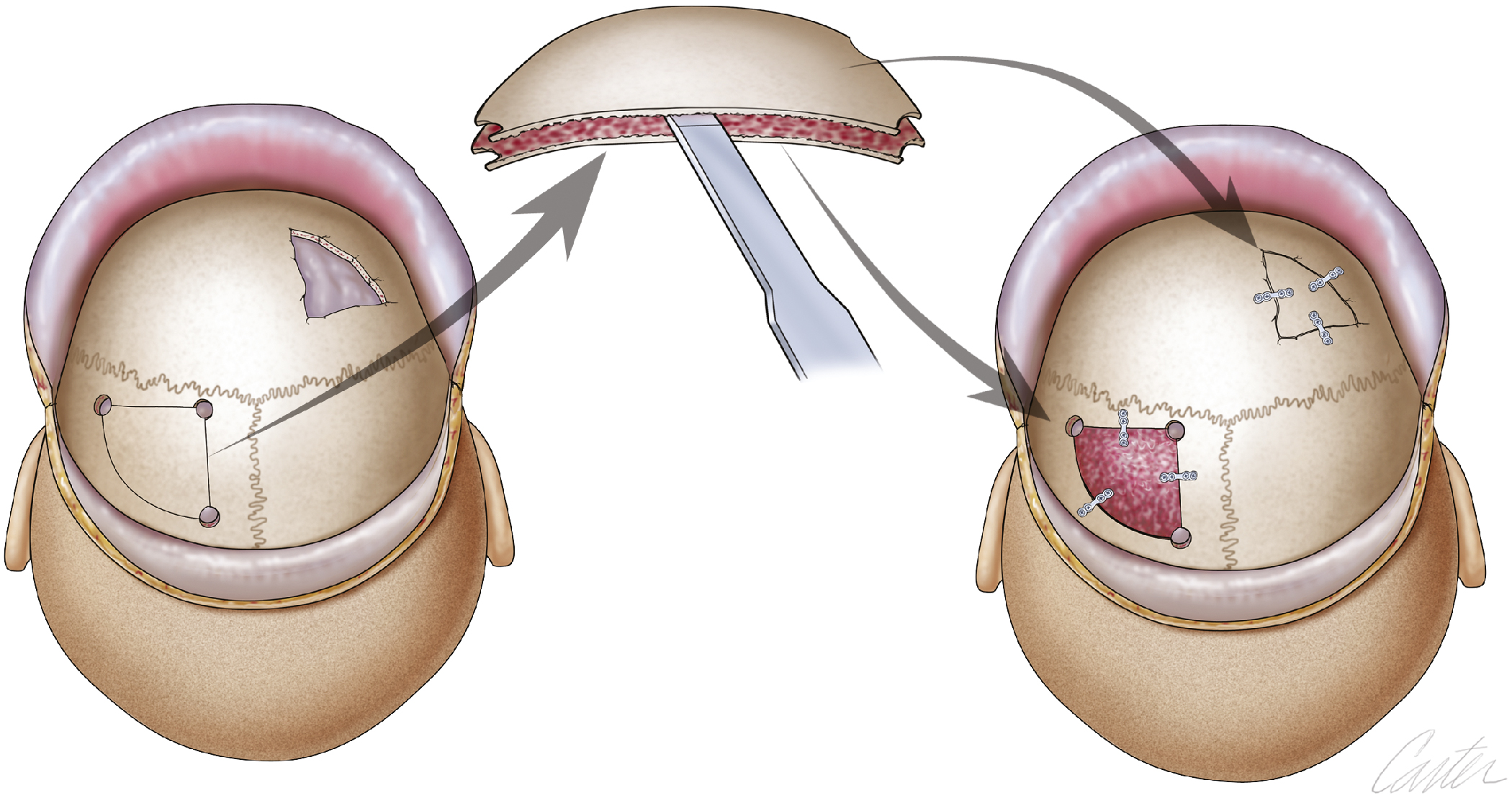
Bone
Split-calvarium and rib are bone graft options for autologous cranioplasty; the iliac crest is often inadequate for grafting. Calvarial bone grafting is safe when performed by an experienced surgeon, but major morbidity resulting from dural tears, meningitis, and bleeding has been reported. When planning internal orbit and nasal reconstructions or midface skeletal augmentation, a calvarial graft is usually taken from an intact skull and usually from the parietal bone, making it possible to conceal the donor site. The alternative approach is to perform craniotomy and split a resected bone segment so that the inner table and outer table can be then fitted to the donor and defect sites, respectively ( Fig. 30.1 ). Splitting can be done with a C-1 bit and a high-speed drill ( Fig. 30.2 ). Naturally, calvarial donor sites are limited. Split-rib may be an option in these circumstances and for larger reconstructions, although its use can create an uneven appearance, depending on the ribs’ prominence through soft tissues.