• Centrifuged fat has three layers: (1) the upper supernatant, containing lipid; (2) the middle stromal vascular fraction, containing adipose-derived stem cells, stromal cells, vascular cells, and mural cells; and (3) the lower layer, containing blood, local anesthetic, and tissue fluid.
• The donor site has not been shown to influence fat graft survival.
• Centrifuge and rolling techniques are the preferred processing techniques for fat preparation. Higher centrifuge speeds are advantageous with 1200 ( g) (gravitational force, or g-force) being the ideal speed.
• Adipose-derived stem cells found in the stromal vascular fraction are advantageous to fat graft survival.
• The fat graft needs to be within 2 mm of a vascularized recipient bed to allow plasmatic imbibition. Therefore injection in small aliquots utilizing a fanning pattern is recommended.
• Shear stress during injection has a negative effect on fat graft survival. It is recommended to inject slowly (0.5–1 cc/second) to minimize shear stress and optimize graft survival.
Introduction
Adipose tissue is a biologic entity composed of various cell types that play a multitude of functions in the body, including insulation, protection and padding, energy storage, and endocrine regulation. Adipose tissue comprises approximately 15% of nonobese female total body mass and 22% of nonobese male total body mass and is distributed all over the body. Liposuction was developed in the 1970s, and it has opened avenues not only to remove excess fat but also to transfer fat from one part of the body to another. Fat grafting was first described in 1893 by Neuber, but it was Coleman who popularized free fat transfer in a minimally traumatic fashion. The so-called Coleman technique has allowed surgeons to obtain a consistently higher graft retention compared with prior techniques. This process produces three layers ( Fig. 14.1 ): the top supernatant layer containing lipid and the lower layer containing blood, tissue fluid, and local anesthetic are both discarded, leaving the middle layer, which contains adipose tissue, adipose-derived stem cells (ASCs), stromal cells, vascular cells, endothelial cells, and mural cells, termed the stromal vascular fraction (SVF). The large number of cells present in adipose tissue highlights what a complex and functional organ it is.
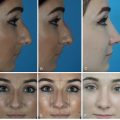
Fat grafting has become one of the most commonly performed procedures by plastic surgeons because of its unique versatility in soft tissue remodeling and its potential in reconstructive and aesthetic surgery. A recent survey by our group showed that approximately 80% of plastic surgeons have used fat grafting in their practice and that last year, a total of 80,000 cosmetic procedures and 30,000 reconstructive procedures involved fat grafting. Over the past decade, fat grafting has been increasingly used to improve facial aesthetics or to correct craniofacial contour abnormalities, ranging from traumatic to congenital deformities, in both the pediatric and adult populations.
Anatomy of a fat graft
Fat graft can be defined as particles composed of adipocytes and interconnecting mesenchyme, including connective tissues, fragments of vessels, and ASCs. Particles do not occur in native fat but, rather, are formed during the harvesting procedure of fat grafting. Fat particles are composed of three sections during healing: the surviving outer layer (1–300 μm), in which all adipocytes survive; the regenerating middle layer (600–1200 μm), where adipocytes undergo necrosis but are replaced by proliferating stem cells; and a necrotizing central core, characterized by oil cysts, necrosis, and fibrosis ( Fig. 14.2 ). ,
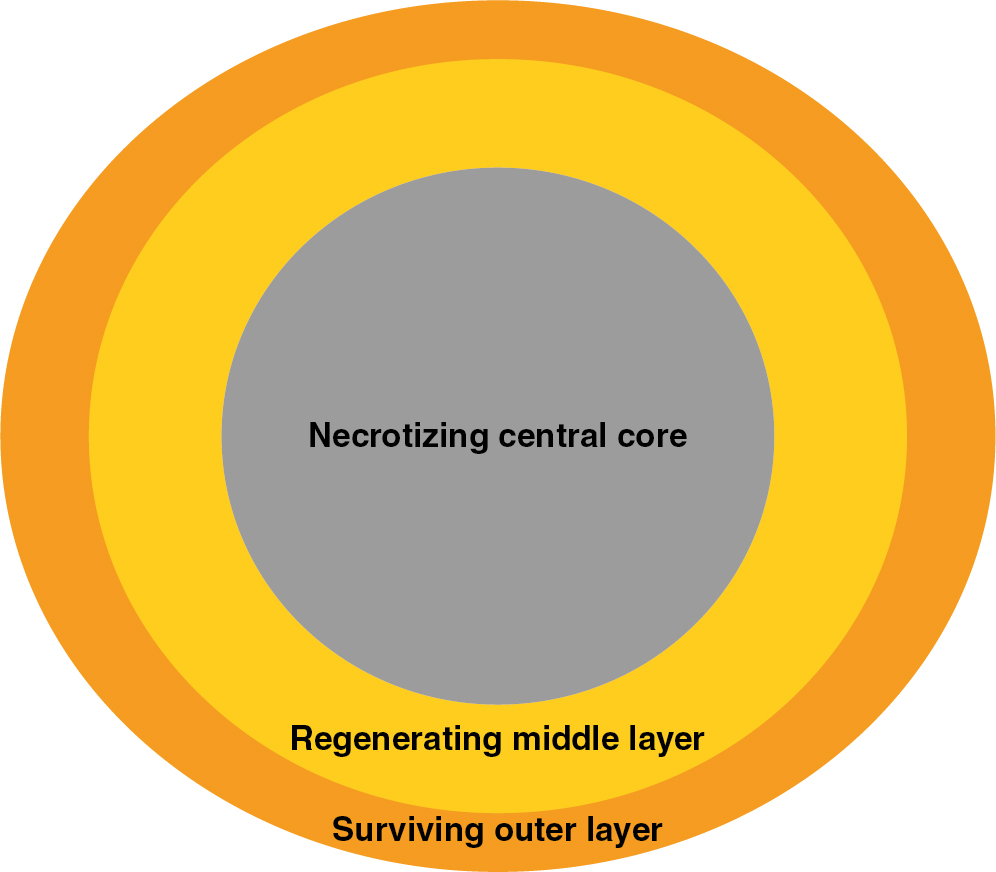
The initial survival of a fat particle relies on simple diffusion of nutrients until blood supply is established. Therefore the larger the diameter of the fat particle, the greater is the necrotizing central core due to diffusional limitations. Fat particle size therefore has a significant influence on the amount of grafted material survival. However, Del Vecchio and Rohrich believe that smaller fat particles may lack stromal components, such a blood vessels, fibroblasts, and connective tissue, for structural support of proliferating stem cells and adipocytes. Therefore one might argue that the ideal fat particle size must be large enough to contain adequate mesenchyme but not too thick to preclude diffusion of nutrients.
Over the past decades, techniques have evolved to produce smaller fat particles. Trepsat described, in 2009, the “microfat” grafting technique, where fat is harvested with a 2 mm cannula with 1 mm side holes. Subsequently, Tonnard et al. described, in 2013, the “nanofat” grafting technique, where fat is harvested with a 3 mm cannula with 1 mm side holes, rinsed with saline, mechanically emulsified by shifting the fat between two 10 cc syringes connected to each other by a female-to-female Luer-Lok connector, and filtered by using a nylon cloth. Nanofat was shown to lack adipocytes, while maintaining a rich supply of ASCs comparable with macro- and microfat in terms of stem cell proliferation and differentiation.
Variables impacting graft survival
The mechanism by which fat survives after being grafted remains unclear, and various theories have been brought forward. The cell survival theory, introduced by Peer in the early 1950s, hypothesizes that fat graft survival is determined by the successful transfer of viable adipocytes and the development of a vascular network from the recipient site. Furthermore, initial nutrition and survival are through plasmatic imbibition, which requires the graft to be within 2 mm from the edge of vascularized tissues. Therefore fat-grafted cells receiving early and adequate blood supply survive, whereas other cells degenerate and are gradually removed. This theory highlights the importance of atraumatic processing and injection techniques to enable the transfer of viable adipocytes. More recently, the host replacement theory, introduced by Suga et al., hypothesizes that ischemic conditions lead to early death of grafted adipocytes, which, in turn, leads to the activation and regeneration of ASCs promoting the replacement of adipocytes.
Harvest site
Fat can be harvested from anywhere in the body, but often the harvest site is selected according to the preference of the surgeon, the patient, or both. However, practical issues have to be considered. For starters, the donor site needs to have enough fat to provide sufficient volume to the desired recipient area. Although donor sites may vary in the amount of fat, the site has no impact on how much of that harvested fat is obtained after processing. Another consideration is how the donor site influences graft survival, but both animal and human studies have shown no differences. Rohrich et al. found no statistically significant differences in adipocyte viability among four donor sites (abdomen, flank, thigh, knee) determined on an in vitro colorimetric assay of cell proliferation. Similar results were confirmed by Li et al., who found no differences in weight, volume, and histologic parameters of fat harvested from five donor sites (upper and lower abdomen, flank, lateral and inner thigh) and grafted into nude mice. Similarly, Small et al. demonstrated that regardless of donor site location (thigh or abdomen), volume retention is comparable, based on retrospective examination of three-dimensional scans of the grafted areas. Given the current evidence, the decision of harvest site should therefore be based on safety, ease of access, and preference of the patient.
Harvesting technique
Various aspects of the harvesting technique can affect fat graft survival, including the cannula size, particle size, and aspiration method. Since the description of the Coleman technique, the use of single-bore, blunt-tip cannulas has been the standard practice.
There are very few studies examining the impact of cannula type and size, and most of the evidence comes from in vitro and animal research. Erdim et al. showed that 6 mm blunt cannulas produce higher in vitro adipocyte survival compared with both 2 and 4 mm cannulas. Alharbi et al. showed no difference in cell viability between fat harvested using a Coleman 3 mm single-bore blunt cannula and a 2 mm multihole blunt tip st’RIM cannula. Kirkham et al. demonstrated that 5 mm Coleman cannulas lead to higher graft retention and improved adipocyte survival compared with 3 mm cannulas in a murine model. Therefore these studies suggest that larger bore cannulas produce more viable adipocytes leading to better fat graft retention. However, we must balance their results with data on particle size discussed previously.
Various aspiration techniques have been developed over the years. The decision of performing hand or machine-assisted liposuction is a balance between the ease and efficiency of harvesting and the risk of adipocyte damage by the increased pressure from the machine. In fact, Pu et al. showed that hand liposuction produced a greater number of viable adipocytes compared with machine-assisted aspiration at no greater than 20 cm H 2 O. However, Lee et al. found no difference in fat graft retention or histology between -15 mm Hg hand and -25 mm Hg machine-assisted harvested fat.
Machine-assisted liposuction can employ the assistance of vacuum pressure, ultrasound, or waterjet to facilitate the harvesting of fat. Our group showed similar graft retention and histology between ultrasound-assisted liposuction and traditional liposuction at 430 mm Hg in a murine model. Yin et al. showed better fat graft retention and adipocyte viability with waterjet-assisted liposuction compared with traditional liposuction at -0.5 bar in BALB/c nude mice. Current evidence suggests that machine-assisted liposuction does not impact graft survival, although it may affect adipocyte viability; waterjet-assisted liposuction may improve fat graft retention and cell viability, whereas ultrasound-assisted liposuction does not appear to have an impact on graft survival.
Processing technique
Four primary processing methods have been used clinically to process fat graft: centrifugation, filtration, rolling, and decanting techniques.
Most of the studies have investigated centrifuging as a processing technique because of the consistent results shown by Sydney Coleman with his technique, which involves centrifugation at 3000 rpm (1200 g ) for 3 minutes in 10-mL syringes. As mentioned previously, this process produces three layers, allowing the surgeon to discard the undesirable top supernatant layer and the lower blood and anesthetic layer, leaving the middle layer, which contains adipose tissue and SVF (see Fig. 14.1 ). There is clear evidence demonstrating the superiority of centrifugation compared with nonprocessed fat in terms of retention and cell viability. However, the speed of centrifugation has been debated. Ferraro et al. compared resorption rates in patients grafted in the gluteal region with fat centrifuged at 3000 rpm (1500 g ) × 3 minutes, 1300 rpm (250 g ) × 5 minutes, or uncentrifuged and decanted. They confirmed that no centrifugation leads to the highest resorption rate and found the slower centrifuge speeds to cause the lowest resorption rates. Hoareau et al. looked at the effect of speed on adipocyte survival and showed that although speeds up to 400 g had little impact on adipocytes, speeds of 900 g caused adipocyte damage. Finally, Kurita et al. compared grafts that were not centrifuged or were centrifuged at 400 g , 700 g , 1200 g , 3000 g , or 4200 g for 3 minutes and found statistically significant improvement in retention up to 3000 g , but when balanced with ASC concentration and volume compaction, 1200 g was suggested to be the ideal speed. Thus in determining the ideal speed, cell damage and concentrating power must be balanced to produce optimal graft survival.
Our group compared the centrifugation, rolling, and filtration techniques by transferring processed human fat in our nude mouse model. Although all of these techniques demonstrated adequate graft healing, intact adipose architecture, and equivalent vascularity, gauze-rolled fat produced the highest SVF cell count and graft volume retention. Similarly, Canizares et al. showed that Telfa rolling, compared with unprocessed and centrifuged lipoaspirate, produced superior adipocyte viability as well as improved graft retention in a murine model. However, Minn et al. compared centrifuging, gauze rolling, and sieve filtering in a murine model and found no statistically significant differences in fat graft retention or necrosis but noted a statistically significant lower in vitro cell viability when fat was processed with a metal sieve.
Another processing technique that has emerged recently is the “nanofat” grafting technique. As mentioned previously, the fat is harvested with a 3 mm cannula with 1 mm side holes, rinsed with saline, mechanically emulsified by shifting the fat between two 10 cc syringes connected to each other by a female-to-female Luer-Lok connector, and filtered by using a nylon cloth. Nanofat was shown to lack adipocytes while maintaining a rich supply of ASCs comparable with macro- and microfat in terms of stem cell proliferation and differentiation. These authors also reported several clinical cases using nanofat grafting, demonstrating improved skin quality and no complications 6 months postoperatively.
The current evidence confirms that fat graft should be processed by using either centrifugation or rolling techniques. Centrifugation with higher speeds and densities appears to be beneficial, most likely because it concentrates adipocytes, ASCs, and angiogenic growth factors, which all play a key role in the survival of the graft. However, from a practical perspective, the fat processing technique must allow feasible workflow in the operating room. For this reason, a surgeon might need to tailor the processing technique based on the volume of fat that needs to be grafted. For instance, smaller fat volumes can be easily processed with either centrifugation or rolling techniques, whereas larger volumes may be more efficiently performed with filtering or decantation.
Intrinsic patient variability in fat tissue
The unpredictability of fat graft retention remains a major issue, and intrinsic patient factors are likely to play a role. Unfortunately, very few studies directly examined patient variables as predictors to successful fat graft survival. Geissler et al. examined fat in 24 female patients harvested from the lower abdomen, inner thigh, and flank using dry liposuction. These authors showed that younger patients had greater adipocyte viability in the lower abdomen than in the flank, whereas older patients did not show any differences. Lower abdominal fat in younger patients showed superior adipocyte viability compared with that in older patients. However, flank fat had higher adipocyte viability in older patients. Patient weight did not appear to affect adipocyte viability. Denadai et al. performed a prospective analysis of 142 patients who had unilateral craniofacial contour deformities and underwent fat grafting. These authors identified older age, Parry-Romberg syndrome, previous craniofacial bone surgery, grafted volume, and forehead unit as independently negative predictors of fat graft retention, whereas injection in the cheek unit was an independently positive predictor. It therefore appears that age and underlying pathology of the recipient play a role in adipocyte viability and retention. Another factor that has been studied is exposure to radiation. Fat grafting has been implicated in the improvement of radiation-induced fibrosis, but the role of radiation in fat graft survival remains uncertain. Retrospective studies by Choi et al. and Small et al. showed that prior radiation exposure does not affect fat graft retention over time in breast reconstruction. However, Garza et al. did not agree with these findings because their case-control immunocompromised mouse model showed that fat graft retention rates are significantly lower in irradiated tissue compared with nonirradiated tissue, but the quality of grafted fat is comparable.
The role of other factors, such as gender, diabetes, hypertension, smoking, and obesity, on the impact of fat graft retention and adipocyte viability remains uncertain. Only few studies have confirmed their impact, but the evidence is minimal and weak. For instance, Özalp and akmakoğlu attempted to describe the impact of smoking on facial fat grafting in their 18-patient retrospective cohort study and highlighted a low rate of fat graft survival and low impact on skin quality improvement. However, no controls were included, and this highlights the need for larger prospective studies focusing on patient factors as predictors for fat grafting results.
Our search for intrinsic factors has led us to consider ASCs as a key correlating factor in graft volume retention, given their capacity for differentiation and growth factor release. We compared lipoaspirate from eight human subjects, and after standardized centrifugation, lipoaspirate was injected into athymic nude mice. A strong correlation was found between SVF percentage of CD34+ progenitor cells (e.g., endothelial mature, endothelial progenitor, pericyte, and preadipocyte/supraadventitial ASCs) and high graft retention, thus making the concentration of CD34+ progenitor cells within SVF, a potential fat grafting biomarker, to accurately predict fat graft retention in patients.
Injection technique
Injection of the processed fat into the recipient site requires significant care to produce long-lasting desirable outcomes. Coleman described the use of Luer-Lok syringes connected to a 17G blunt cannula to inject the processed lipoaspirate. The cannula is withdrawn as lipoaspirate is injected to allow the grafted fat to fall into the natural tissue planes as the cannula is removed.
Most surgeons believe a single bolus injection of a large volume of fat is counterproductive because it leads to poor outcomes and complications, such as fat necrosis. As mentioned previously, the fat graft needs to be within 2 mm from the edge of vascularized tissues to survive through plasmatic imbibition. For this reason, injections in small aliquots in a fanning pattern is recommended to optimize survival.
Similar to harvesting technique, cannula size has been thoroughly investigated, and various studies have shown no differences in adipocyte count or graft retention. Erdim et al. compared fat injected through 14G, 16G, and 20G cannulae and showed no statistically significant difference in cell viability. Nguyen et al. also showed no statistically significant differences in retention or histology by injecting 0.7 mL of human fat in nude mice through 17G, 20G, 23G, and 25G cannulae. Despite this evidence, other studies have shown that fat injected through narrower, longer, and bent cannulas exhibited more shear thinning with diminished quality. In fact, Luan et al. showed that fat injected through a 14G and 8 cm long cannula significantly improved the in vivo histologic structure compared with fat injected through longer or narrower cannulas and that it also improved fat graft retention compared with 18G and 8 cm long cannulae. The reason for these differences in findings among studies is uncertain. A possible theory is that cannula size and length affect the shear stress during injection. In fact, Lee et al. found that the decrease in shear stress with slow injection rate (0.5–1 cc/sec) compared with fast injection rate (3–5 cc/sec) was the cause of the increased weight of lobules, improved graft survival, and healthier adipocytes with less vacuoles, infiltrate, and fibrosis.
We can therefore conclude that although not enough evidence exists to support a specific type of cannula, shear stress has proven to have a significant impact on adipocytes. Because shear stress is proportional to flow rate, lipoaspirate should be injected slowly (0.5–1 cc/sec) to optimize graft survival.
Recipient site
How can the recipient site affect graft survival? Fat graft retention depends on early revascularization, among many things, and this means that injection of lipoaspirate into a vascularized bed is crucial. To assess for the optimal recipient bed, Karacaoglu et al. injected lipoaspirate into the subcutaneous, supramuscular, and submuscular layers of the buccomandibular area in rabbits. These authors showed that fat graft survived more in the supramuscular layer than in the subcutaneous or the submuscular layer. Shi et al. similarly showed that lipoaspirate was retained longer in fat pads than under the dorsal dermis or intramuscularly. However, angiogenesis and vascularized connective tissue were more prominent in fat injected in the intramuscular layer, likely being the reason for the highest survival rates among the three recipient sites. The facial anatomy, with its intricate musculature, poses a challenge for the surgeon because areas with increased mobility, such as the glabella and the lips, are less amenable to correction, compared with less mobile areas, such as the malar and lateral cheek. More objectively, Denadai et al. demonstrated radiologically that fat graft retention was better in the cheek unit at 12 months compared with the forehead or the chin units. As the face ages, it progressively loses fullness, leading to increased prominence in the underlying soft tissue and skeletal structures; as such, facial physiognomy can be improved by infiltrating fat into selective fat compartments to accurately control facial contouring. Aging is also associated with deterioration in graft revascularization, which may be associated with lower volume retention of fat grafts.
Manipulation of the recipient site has been greatly studied, and it offers the chance to improve graft survival and predictability. Much of the research has focused on preconditioning and preexpansion of the recipient site by using devices that induce mechanotransduction, by which mechanical tension is converted into growth-promoting signals. An example is the use of Brava (Brava, LLC, Miami, FL), an external soft tissue expander used in breast surgery to produce sustained and low-level negative pressure leading to outward distractive force. This stimulates breast tissue enlargement and produces a large fibrovascular scaffold, which is an ideal recipient for fat graft. The benefit of this technology was demonstrated in a prospective multicenter study highlighting the significant improvement in weighted mean graft retention rate and the ability to inject much larger volumes of fat graft.
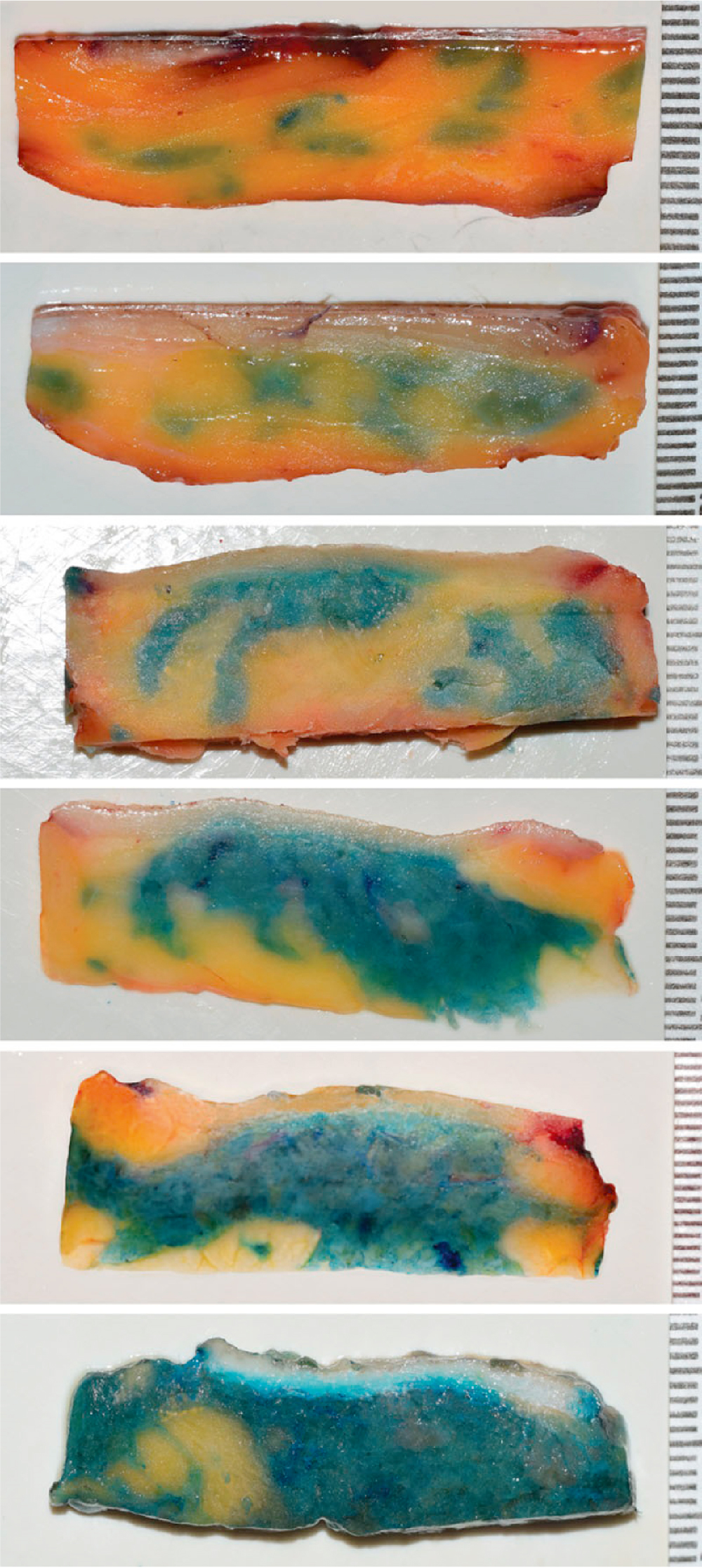
Khouri et al. hypothesized that two fundamental principles influence the results of large-volume fat grafting: graft-to-recipient interface and interstitial fluid pressure limit. The graft-to-recipient interface principle suggests that grafted adipocytes should be no more than 2 mm away from their recipient capillary network to survive. Thus only “microribbons” should be injected to ensure survival of fat graft because larger injections are likely to suffer central necrosis. This leads to a theoretical model where fat is injected through a cannula with an internal diameter of 4 mm to deliver no more than 1 mL of fat along a 10 cm pass. Multiple passes should be used throughout the recipient tissue so that the ribbons of fat are not confluent. The accidental creation of cavities or pools of fat can lead to failure to vascularize because of the distance from the center of the graft to the vessels. Although this theory is reasonable, in practice, it is unrealistic because not only it would be challenging to precisely inject only 1 mL of fat in a 10 cm pass, but it would also be difficult to distribute it in perfect microribbons without spreading or pooling. Our group confirmed this by examining the architecture of fat deposits following fat grafting stained with methylene blue. Our study showed that as the total transferred fat volume increases, grafted deposits coalesce to form larger globules, particularly at ratios beyond 1:4 ( Fig. 14.3 ), thus increasing the likelihood of central necrosis and subsequent graft loss.