Introduction
Severe burn injury is a dramatic event: dramatic in its inception in the flaming home, in an industrial accident, or in the hands and actions on a child of an abusive adult. Extraction, transport, and emergency room care are imbued with purpose, speed, sometimes panic, and sometimes efficiency. The arrival of the patient in the burn center signals a change from the fast-paced immediacy of the emergency trauma situation to the deliberate careful and calculated approach to a serious problem that has many aspects both acute and chronic.
Burn care has improved in the past 20 years, perhaps more than any other aspect of medicine in the institution of the multidisciplinary team approach that begins with the initial evaluation of the injury and continues throughout hospitalization and after discharge. Each member of the burn team (nurse, physical therapist, occupational therapist, dietitian, psychologist, social worker, pharmacist, etc.) led by the burn surgeon participates in the initial evaluation of the patient and the therapeutic plan. The plan and its execution must not be considered a sequential or step-by-step process but an integrated approach synthesizing all present and future problems, real or potential, and addressing them prospectively. Even as the total body surface area (TBSA) and depth of the burn are being assessed to calculate resuscitation, prediction of functional and aesthetic problems is considered, and these are mitigated either by preventative actions or by a direct medical or surgical approach. The reconstructive aspects of burn care begin at the same time as those actions directed at survival itself.
This integrated approach to the burn must, at its inception, address the enormous emotional and psychological trauma that this injury carries with it. The patient and family must be informed and educated for best results. The elements of fear, guilt, or denial make early informed consent difficult, and the surgeon and other team members must be patient and constant in their support of the patient and family—reassuring them that though many steps may be necessary in the process, they all move toward the ultimate goal. Because most of the public looks at surgery as a single definitive and hopefully curative event, it is important that the surgeon and team gently assure the patient and family that the burn is a multifaceted injury that requires time, effort, and often multiple operations to overcome and that the initial plan, no matter how detailed or comprehensive, will likely require significant modification. They must be prepared for a prolonged course that may change and one in which each operation or the patient’s response to it may alter the course. Such an initial approach combined with frequent updates to the patient and family will help create an environment of trust and hopefully minimize the depression, despair, and even anger that so frequently accompany the situation of a burn injury. The early incorporation of a social worker and psychiatrist or psychologist may be useful in facilitating this aspect of care.
As in any disease or injury, there is a hierarchy of priorities in the care of the burn patient. Clearly, survival is foremost. Protection of, restoration of, and maintenance of function is second. Appearance is a critical factor in allowing the patient to return to function in society. Because of the disfiguring and deforming nature of the burn injury and its therapy for children and adults alike, the restoration or preservation of sexuality is also important to consider. Interwoven throughout is the necessity of minimizing the pain inflicted upon a patient and maintaining as stable an emotional milieu as possible throughout the course of therapy ( Table 42.1 ).
Table 42.1
Hierarchy of Priorities in Burn Care
|
As previously stated, reconstruction is not a second or third step in burn care but an integral part of the process. In the initial evaluation, immediate assessment of the function of the eyelids and eyes is critical to prevent cicatricial ectropion and subsequent damage to the globe, particularly the cornea. The hands, particularly on adults, must be splinted in the position of function (wrist slightly extended, metacarpophalangeal joints at 90 degrees, interphalangeal joints at 0 degrees) to prevent shortening of the collateral ligaments and subsequent joint stiffness and even arthritis ( Fig. 42.1 ). Joints over which there are burns must be enrolled in an immediate passive range-of-motion protocol, and the feet and toes should be splinted in the neutral position (ankle 90 degrees, toes 0 degrees) ( Table 42.2 and Fig. 42.2 ).
Splinting of the hand both in the acute injury and in the rehabilitative period should be in the position of function with dorsiflexion, thumb extensor abduction, the metacarpophalangeal (MP) joints flexed at 90 degrees, and the interphalangeal joints extended 0 degrees. This keeps the MP collateral ligaments at full length so that they will not shorten during inactivity resulting in decreased excursion of the joint.
Table 42.2
Hierarchy of Priorities in Burn Reconstruction
|
(A) Joints that need special attention in splinting a burn patient. (B) In large burns, the sequence of coverage should support the priorities in burn care (see Table 42.2 ).
The very initiation of fluid resuscitation has enormous ramifications on reconstruction. The burn injury is a dynamic situation particularly in patients with large burns with a component of partial thickness. Within the zone of injury, there is a zone of coagulation or cell death, a zone of stasis or moderate to impending ischemia, and a zone of hyperemia ( Fig. 42.3 ). Inadequate fluid resuscitation results in hypoperfusion of the burn causing coagulative necrosis in the zone of stasis and the zone of hyperemia resulting in conversion of portions of the partial-thickness burn to full thickness. Such conversion increases the total body surface area ultimately requiring reconstruction. Excessive fluid resuscitation may cause persistent edema in burned and nonburned areas. This edema may result in conjunctival or corneal edema, inability to flex and extend involved joints, and increased interstitial pressure aggravating the damage done to the underlying tissues by full-thickness burns. Thus, although survival is clearly the first priority in fluid resuscitation and a major part of affecting it, reconstructive considerations must be incorporated in its delivery.
The burn is usually a dynamic process. Areas of definite cell loss (zone of coagulation) are surrounded by areas of marginal profusion (zone of stasis), which are in turn surrounded by areas of hyperemia resulting from acute inflammatory response. Adequate tissue perfusion and good wound care will sometimes allow the zone of stasis to decrease in size, making the extent of injury small. Inadequate fluid resuscitation and poor wound care will expand the zone of coagulative necrosis and make the injury more extensive.
Most burn centers today employ various techniques of early burn excision and coverage to prevent the persistence of negative physiologic influences by the burn, to decrease the risk of burn wound sepsis, and to restore, as quickly as possible, the numerous homeostatic mechanisms to the body that the skin, its largest organ, provides. If not enough autologous skin is available to accomplish this with one procedure, a skin substitute or temporary covering is necessary to prevent numerous problems attendant on a large open wound. Cadaver homograft is superior because it engrafts temporarily even when full-thickness loss of skin is present. Although this may be only a temporary coverage, careful placement and fixation to the wound will decrease the risk of excessive or prolonged granulation tissue, which may predispose toward hypertrophic scarring when definitive wound coverage with autograft is accomplished. Complete coverage with homograft or autograft will also facilitate the ongoing preservation of function by allowing early range of motion exercises.
Wound healing and reconstruction
Reconstruction of the burn wound has three basic goals: replacement of burned dysfunctional skin with autologous functional tissue, prevention of scarring, and removal or minimization of scarring and its effects. Partial-thickness wounds heal by reepithelialization of the epidermis by migration of basal cells or keratinocytes up from the depth of the hair follicles and sebaceous glands in the burned skin and migration centrifugally across the remaining dermal infrastructure until they meet other cells upon which their migration is stopped by contact inhibition ( Fig. 42.4 ). The provision of a moist environment for this process and the prevention of invasive infection facilitates this process. Intermittent hydrotherapy and subsequent burn coverage with an occlusive dressing and topical antibiotics will ordinarily provide such an environment. In full-thickness injury, or when partial-thickness injury converts to full thickness, the process of acute inflammation proceeds. Inflammatory cytokines attract neutrophils, monocytes, and fibroblasts. If the wound persists in the inflammatory stage, the normal progression to the proliferative phase and subsequent remodeling of collagen is thwarted. Granulation tissue represents persistence of the inflammatory phase and, because of the prolonged abundance of the population of fibroblasts and other factors, predisposes to hypertrophic scarring. Prevention of persistent granulation tissue by meticulous application of homograft and subsequent autografting will minimize granulation tissue and thus, to some degree, hypertrophic scarring.
After the edema and other aspects of the acute inflammatory stage subside and the eschar or other damage to epithelial cells is removed by debridement or topical wound care, this skin heals by the migration of basal cells (keratinocytes) from the sweat glands, sebaceous glands, and hair follicles up onto the surface of the burn wound. They then move centrifugally until they come in contact with the intact epidermis or other migrating keratinocytes when they stop, chemically signaled by contact inhibition. This process occurs in partial-thickness burns only. Full-thickness burns, where there are no surviving skin appendages, are healed by contraction and epithelialization or migration of keratinocytes into the defect from the edges. Both of these processes occur more efficiently in a warm, moist environment with minimal bacterial colonization.
Contraction of the wound is an attempt to minimize the surface area and promote normal healing by secondary intention. This does not necessarily lead to scarring. It is the persistence of the inflammatory stage with its overabundance of fibroblasts, mast cells, and edema that results in hypertrophic scarring and ultimately to contracture or scar-shortening of normal tissue relationships. Prevention of this abnormal extension of the healing process is the most efficient way to deal with it, but when contracture is present, it is much better treated either medically or surgically. Medical therapy with steroid topical applications and intralesional injection of steroids has been attempted. Although there may be some decrease in the size of scars, particularly with injection, this is usually only partially effective for scars of limited size. Injection of steroids into adjacent normal skin or areas without hypertrophy may result in atrophy of subcutaneous tissue and discoloration of surrounding skin. Surgical therapy usually involves resection of the contracture and coverage of the underlying tissue with more normal tissue.
Skin grafting involves removal of an area of skin from its underlying tissues, the donor site, and transporting it without its blood supply to an area without any skin or without the full thickness of skin, the recipient site. The thickness of the graft and the thickness of the skin at the donor site determine the amount of normal skin components and appendages in the graft and thus, assuming complete take of the graft, the similarity of the engrafted skin at the recipient site to normal skin. Split-thickness skin grafts take with them the epidermis and some of the dermis, depending on their thickness ( Fig. 42.5 ). Full-thickness skin grafts, as the name implies, contain all of the skin down to the subcutaneous fat, including hair follicles, sebaceous glands, elastin fibers, and all other components of normal skin ( Fig. 42.6 ). A thin split-thickness skin graft may include only the epidermis and a thin layer of dermis, whereas a thicker graft may include most of the dermis. The process of engraftment is a multistage event. When a skin graft of any thickness is placed on its recipient bed, fibrin from the underlying wound bonds it to the surface initially. For the first 24 to 48 hours, the keratinocytes, melanocytes, and other cells of the graft receive their nutrition from and discharge their metabolites into the interface between recipient bed and graft by simple osmotic diffusion—a process termed plasmatic imbibition. During this period, the beginning of a blood supply to the graft develops as the blood vessels in the underlying tissue create anastomoses to the cut ends of the blood vessels remaining in the graft; the second phase of graft take is known as inosculation. Over 24 to 36 hours, a new circulation is established within the grafts so, if pressure is applied, the graft will blanch. Over the next several weeks, the deposition and remodeling of collagen and the maturation of the vascular network between the recipient bed and overlying grafts stabilize the tissue, although it is unclear whether the remnants of the blood vessels in the graft persist or are replaced by the process of angiogenesis. Either way, the graft is now nourished in the normal fashion by arterioles, venules, and capillaries.
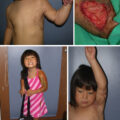
(A) Dorsal scar contracture after excision and grafting of foot for deep partial-thickness burn. (B–C) Appearance of regrafted foot showing stable grafts after resection of scar contracture.
Cross-section of normal skin showing the epidermis, consisting of basal cell (keratinocytes), melanocytes, and the basement membrane. Keratinocytes progress through various growth stages and ultimately become keratinized epithelium, which enables the many barrier functions of the skin. The subjacent dermis contains the skin adnexae, sweat glands, sebaceous glands, and hair follicles. It also contains the arborizations of the blood supply ascending from any axial cutaneous vessels, the perforating vessels, and the subdermal plexus.
Interference with any of these steps will prevent graft take. The bed must be adequately vascularized to allow metabolite diffusion into and out of the grafts, therefore bone denuded of periosteum or tendon stripped of paratenon will not accept a graft. Any mechanical force that prevents the juxtaposition of the tissues will interfere with both plasmatic imbibition and inosculation; thus seroma, hematoma, and shearing will result in the loss of the graft. Enzymatic activity from microbes, such as bacteria and fungi, will interfere with the protein synthesis necessary for the establishment of a stable vascular supply to the graft. Toxicity effects of free hemoglobin below the graft and certain chemicals sometimes used in dressings may interfere with cellular metabolism and thus impair graft take.
The anatomy of the graft itself predicts its ability to become engrafted. A thin split-thickness skin graft has fewer cells in its vertical dimension and thus a shorter distance for diffusion of metabolites during the phase of plasmatic imbibition. It is therefore more likely that all the cells will be adequately nourished. Furthermore, the thinner the dermal layer, the farther out the cells are from the arborization of blood supply from the subcutaneous perforators to the subdermal plexus to the dermal vascular system, and thus there are more recipient vessels for the blood vessels in the bed to join in the phase of inosculation ( Fig. 42.7 ). The thicker the split-thickness skin graft, the longer the distance for diffusion, and the fewer the blood vessel ends remaining in the graft, so the higher the risk in both phases resulting either in no take, slower revascularization, or epidermolysis (loss of the superficial layers of skin). This is true specifically with full-thickness skin grafts. Healing of the donor site of the split-thickness skin graft occurs as keratinocytes migrate out of the bases of the transected hair follicles and sebaceous glands, traveling centrifugally over the denuded surface until coming in contact with other keratinocytes similarly moving. The thicker the split-thickness skin graft taken, the fewer residual hair follicles and sebaceous glands there are left at the donor site and thus the longer the distance the keratinocytes need to migrate before completely covering the wound. Thus the thicker the split-thickness skin graft taken, the longer the time to complete healing of the donor site. This migration of keratinocytes, known as epithelialization , proceeds most quickly and efficiently in a sterile, moist environment at body temperature. Dried eschar of blood and fibrin on the donor site serves as a mechanical and chemical barrier to this movement and should be minimized by covering the donor site with a sterile occlusive dressing, such as gauze impregnated with antibiotic ointment. Because full-thickness skin grafts remove all the skin appendages leaving only subcutaneous fat or muscle as the bed, the donor sites must be closed preferably by direct approximation and healing by primary intention or by coverage with a split-thickness skin graft.